Advanced Stainless Steel Production: From EAF to Modern Oxygen‑Based Techniques
Stainless Steel Manufacturing Processes
Stainless steels contain from 10 % to 30 % chromium. These steels also contain varying amounts of nickel, molybdenum, copper, sulphur, titanium, and niobium etc.
The majority of production of stainless steel was through the electric arc furnace (EAF) till around 1970. With the use of tonnage oxygen in steel production, the EAF stainless steel making practice changed. Oxygen gas could be used for improving the decarburization rate. This could be achieved by injecting high oxygen potential but it was accompanied by the adverse reaction of extensive oxidation of chromium to the slag. This necessitated a well defined reduction period in which ferro silicon was used to reduce the oxidized chromium from the slag.
Production of stainless steel started by duplex process with the successful development of argon oxygen decarburization (AOD) converter process. Though duplex process with AOD converter is the prominent one, there are several duplex processes are being used today for making stainless steels. In these processes there is an EAF or similar furnace that melts down scrap, ferroalloys and other raw materials to produce the liquid steel. This liquid steel, which contains most of chromium and nickel as well as some other alloying elements, is the charge of the converters. The converters are used to achieve low carbon stainless steels. The versatility of the EAF-AOD duplex process led steelmakers to re-examine the use of different converters for melting of stainless steels. This led to the development of several other converters for duplex processes.
The development work to make stainless steels using conventional BOF (basic oxygen furnace) had begun in the late 1950s and early 1960s. By the mid 1960s, some steelmakers were using existing BOF converters for a partial decarburization followed by decarburization in a ladle under vacuum to make the low carbon stainless steels. These processes are known as triplex processes because three process units, such as EAF, a converter for pre-blowing and a vacuum decarburization unit for final refining, are involved. The steels undergo treatment for final decarburization, final trimming, homogenization and flotation of inclusions before the ladle is taken to the teeming operation. In almost all of these triplex processes, vacuum processing steels in the teeming ladle is the final step before casting. Fig 1 shows comparison of duplex and triplex process routes.
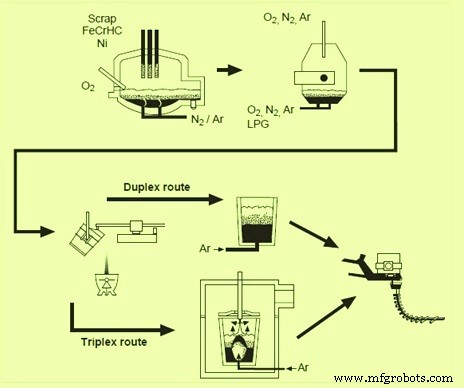
Fig 1 Comparison of duplex and triplex process routes
With all these developments presently there are available different processes to make stainless steels. The available processes are divided into three groups. They are the converter processes, converter with vacuum processes, and vacuum processes.
General principles of producing stainless steels
Stainless steel decarburization must minimize the oxidation of chromium. It is generally accepted that when oxygen is injected into stainless steel, a mixture of chromium and iron is oxidized. Decarburization occurs when dissolved carbon reduces the chromium and iron oxides that form. The decarburization sequence is thus
3O? (g) + 4Cr = 2Cr?O?
Cr?O? + 3C = 2Cr + 3CO (g)
Decarburization occurs on the surface of rising bubbles that form from either the inert gas that is injected or on the surface of chromium oxide particles that are being reduced and generating CO.
Decarburization involves techniques to minimize chromium oxidation. There are three basic techniques which are temperature, dilution, and vacuum.
The temperature technique was used by EAF stainless steel making before the development of duplex processes. As the temperature increases, the equilibrium content at particular chromium content decreases. However, this leads to operational difficulties and high costs.
The dilution technique is that used by the AOD and all converter processes. The injection of inert gas (argon or nitrogen) lowers the partial pressure of CO gas in the bath, thus allowing higher chromium contents to be in equilibrium with lower carbon contents.
Applying a vacuum to the metal bath also removes CO, allowing high chromium contents to be in equilibrium with low carbon contents. It is especially effective when the carbon content is low.
Careful control of the slag, as it is produced in the reaction, is important. Any chromium oxide not reduced by carbon ends up in the slag, which can form a complex spinel. Subsequent processing (called reduction) is required to recover oxidized elements such as chromium, iron, manganese etc. The effectiveness of the reduction step is dependent on many factors including slag basicity and temperature, mixing conditions in the converter and solid addition dissolution kinetics.
Choice of process routes
There are many different process routes available for stainless steel manufacturing process. One has to choose from these routes based on many factors which determine capital and operating costs. The choice of process route is influenced by raw material availability, desired product, downstream processing, existing shop logistics, and capital economics. Also some degree of flexibility in process route is desirable, as these factors can change in due course. In general, stainless steel making process flow can be classified as duplex or triplex. Duplex process, where EAF steel making is followed by refining in a converter, tends to be flexible with respect to raw material selection. Triplex process, where EAF steel making and converter refining is followed by refining with a vacuum system, is often desirable when the final product has very low carbon and nitrogen specifications. Triplex process tends to have overall cycle times longer than duplex process because there is an extra transfer from process converter to vacuum unit. It also tends to have slightly higher refractory costs because there are two furnaces performing decarburization.
For maximum flexibility, a shop should have a process flow that can incorporate both duplex and triplex routes. This offers the most flexibility in raw materials, production capability, and process flow. In this case, only products that require vacuum refining for economic and/or quality reasons will use a triplex route.
Dilution refining processes
In a converter, decarburization is carried out using the dilution principle. There are different converters based on the gases used. Converters are also either side or bottom-blown. Users of side blown converters claim that side blowing results in higher carbon removal efficiencies (amount of oxygen reacting with carbon divided by the total amount of gases blown) in the range of 0.1 to 0.005 % carbon, due to longer inert gas bubble residence time, and improved desulphurization in the range of 0.005 % to less than 0.001 % sulphur due to improved mixing. The most popular converters dilution processes are described below
- AOD converter process – The process is described in a separate article at link http://www.ispatguru.com/argon-oxygen-decarburization-process/
- K-BOP and K-OBM-S – Kawasaki Steel Corporation’s K-BOP process began as conventional top oxygen blown BOF. It was modified to have seven bottom tuyeres of the OBM (Q-BOP) type. These tuyeres could blow oxygen with propane for tuyere cooling. Powdered lime could also be injected through these tuyeres. The K-OBM-S process developed by Voest Alpine Industrieanlagenbau (now Siemens VAI) and evolved from Kawasaki’s K-BOP process. The K-OBM-S process initiated with tuyeres in the converter bottom. However, some installations are side-blown reactors. Thus a K-OBM-S converter is a top blown with a lance and with bottom or side blown with tuyeres. It is very similar to a modern AOD. However, in the K-OBM-S process, hydrocarbons, such as natural gas or propane, are used for tuyere protection and this can be helpful in increasing the refractory life.
- Metal refining process (MRP) converter -The MRP converter was developed by Mannesmann Demag Huttentechnik (now SMS Siemag). Molten liquid steel, which contains chromium and nickel, is charged to the MRP converter. Decarburization is carried out using oxygen and inert gases. In early stages of development, the gases were alternately blown through the tuyeres in the bottom of the converter. The oxygen is blown into the melt without dilution with any inert gas. The desired oxygen blow is followed by blowing with inert gas only. The cycle of oxygen blow followed by the inert blow is called cyclic refining or pulsing and the developers claim that flushing with pure inert gas can lead to achieving low CO partial pressure and faster decarburization and thus lower chromium oxidation and consumption of silicon for reduction. The original version of the converter has now evolved into the MRP-L process in which all oxygen is top-blown and inert gas is injected through the porous elements in the bottom. The bottom tuyeres can be replaced easily through the use of an exchangeable bottom. With bottom tuyeres, there is less likely to be erosion on the sidewalls of the converter. In recent years, the MRP-L units have been coupled with a vacuum unit as part of the triplex process for making stainless steels, especially those requiring lower carbon and nitrogen levels.
- Creusot-Loire-Uddeholm (CLU) converter – The CLU process is similar to the AOD process for making stainless steels. It also uses liquid steel from an EAF. The major impetus for its development was the idea to substitute steam as the diluting gas rather than argon. The converter is bottom-blown thus differentiating it from the side-blown AOD converter. The first commercial plant using the CLU process was built in 1973. The decarburization period consists of injecting an oxygen-steam mixture. The process is energy inefficient as the reaction of steam with the molten steel bath is endothermic. Chromium oxidation is higher than in the AOD process when decarburization is continued below about 0.18 % carbon. Although the original goal of reducing argon consumption can be met, the increased silicon requirement for the reduction step does not necessarily lead to overall cost savings. Further, the use of steam throughout the entire period was found to lead to undesirable hydrogen contents in the refined steel. Therefore, practices have evolved which use various amounts of steam, argon, and nitrogen in the process.
- Krupp combined blowing-stainless (KCB-S) process -The production of stainless steels in the BOF converter using the top lance was being practiced prior to the advent of the AOD Process. After introduction of the AOD process, Krupp Stahl AG modified the converter so that combined blowing through the lance and tuyeres could be practiced for refining stainless steels. The process was named Krupp combined blowing-stainless or KCB-S. The simultaneous introduction of process gases helped them increase the decarburization rate. The blowing through a top lance and through the tuyeres below the bath surface helped achieve very high decarburization rates. The increased decarburization rate led to a reduction of up to 30 % in the refining times compared to a conventional AOD alone. Liquid steel from EAF is charged to the converter. At the start of the blow, pure oxygen is injected simultaneously through the lance and sidewall tuyeres. After a desired process temperature is reached, various additions are made during the blow. The additions consist of lime, ferroalloys, and scrap. After a critical carbon level is reached, the oxygen content of the process gas is reduced by using inert gases such as nitrogen or argon. Oxygen to inert gas ratios of 4:1, 2:1, 1:1, 1:2 and 1:4 are used as decarburization to lower levels is pursued. When the carbon content of 0.15 % is reached, the use of the lance is discontinued and the process gases are introduced only through the tuyeres. When the desired aim carbon level is reached the oxygen blow is discontinued and silicon is added as ferrosilicon to reduce chromium oxide in the slag and to achieve the required silicon specification. The addition of lime and other fluxing agents with the ferrosilicon leads to lowering of the dissolved oxygen content and enhances the desulfurization.
- Argon secondary melting (ASM) converter – This process was developed by MAN GHH in Germany. It is similar to the AOD process, except that the tuyeres are in the bottom of the vessel. When using top-blown oxygen, it is identified as the ASM-L process.
- Sumitomo top and bottom blowing process (STB) converter – Sumitomo Metal Industries developed the Sumitomo top and bottom blowing process (STB). It was developed to overcome the disadvantages of a pure top or pure bottom blowing process by combining the two concepts into one process. It also tried to overcome two disadvantages of the AOD process at the time: tuyere erosion and limited oxygen flow rate. The additional supply of oxygen rich gases from the top lance led to shorter decarburization times.
- Top mixed bottom inert (TMBI) converter – In this process converter is equipped with bottom tuyeres to inject only inert gases such as argon or nitrogen. The majority of the process gas is introduced through the top lance. The top lance can be used to introduce the desired mixture of gases. The process is called top mixed bottom inert (TMBI). The process is similar to the other processes that use combined gas blowing in a converter. The plant operated by Allegheny Ludlum with this process has coreless induction furnaces that melt carbon steel scrap and supply chromium-free hot metal to the BOF converters.
- Combined converter and vacuum units – The converter processes discussed above have one disadvantage in that stainless steels with very low carbon and nitrogen residuals become difficult to produce. The decarburization period becomes longer while chromium oxidation and argon consumption increase as the desired carbon and nitrogen levels decrease. Some steelmakers have tried to overcome this disadvantage by applying vacuum to the converter at the very late stages of the decarburization process. The concept was also promoted by Leybould-Heraus as an alternative to AOD or VOD. The concept of applying vacuum to a converter is being pursued by the installation of AOD/VCR by Daido at Shibukawa and Chita plants and by Nippon Steel at its Hikari works. The AOD-VCR operates as a conventional AOD down to 0.08 % – 0.10 % C. The process is stopped for sampling and a vacuum lid is put into place. The lid is sealed to a flange located about half way up the conical section of the converter. A vacuum is pulled and used for the remainder of decarburization and reduction. Desulphurization is carried out in the transfer ladle prior to AOD charge. The major advantages of this process relative to converter processes are decreased argon and silicon consumption. Disadvantages include higher refractory consumption, decreased ability to melt scrap and added maintenance and costs associated with steam production. When compared to separate converter and VOD units, the AOD-VCR has higher operating costs (silicon, refractory, and argon), lower productivity and higher nitrogen contents. Capital costs may be somewhat lower than having two separate units.
Vacuum Refining Processes
The use of vacuum for decarburization of steels was developed in Germany. The early processes included RH degassing, DH degassing, and the Allegheny vacuum refining (AVR) as a second step in the duplex process. These processes involve lowering the pressure above the steel bath to promote evolution of carbon monoxide gas. The liquid stainless steels going into the vacuum process generally contain carbon of about 0.5 % or lower. Most vacuum processes are performed in a chamber with a ladle full of metal as opposed to a separate refining vessel used in the dilution/converter processes.
In mid 1960s, the vacuum decarburization concept was used by Allegheny Ludlum Steel Corporation to develop the Allegheny vacuum refining process. This process became non-competitive with the introduction of AOD process and was discontinued.
Early duplex processes where vacuum processing was used as the second step were too slow and had very limited flexibility with respect to raw materials that could be used. The vacuum processes could not keep up with the improving productivity of EAFs and the operating costs were high. Hence, later developments focused on the use of converters to decarburize the liquid steel from EAFs as a second stage followed by vacuum degassing for the finishing stage. Such processes are known as triplex processes for making stainless steels as they use three processes to achieve the desired finished chemistry.
Nippon Steel Corporation introduced the RH-OB process for making of stainless steels. Hot metal from a blast furnace was fed to a BOF converter where the metal was alloyed with chromium and blown down to a 0.5 % – 0.6 % carbon level. The final decarburization was conducted using the RH-OB process. Nippon Steel converted an existing RH degasser, which was used for carbon steels, so that oxygen could be injected under vacuum.
Thyssen in Germany developed the vacuum oxygen decarburization (VOD) process in the mid-1960s. In the early stages of its development, VOD was used to decarburize molten alloy from the EAF. Later, preliminary decarburization was done in a BOF and the EAF-BOF-VOD triplex process became more productive. In early 1970s, Kawasaki Steel Corporation modified the VOD process using multiple porous plug bubblers in the ladle. They called this the SS-VOD (strong stirring VOD) process. In 1988, ALZ in Belgium modified the facilities to make stainless steels by a triplex process consisting of EAF melting, MRP-L converter, and VOD process.
The major advantages of the vacuum processes include low consumption of argon and low oxidation of chromium during the final decarburization to low carbon levels. The latter has led to lower consumption of reduction elements for the recovery chromium from slag. The teeming of steel from the ladle used in the vacuum processes eliminates the pickup of nitrogen and oxygen from air that is associated with tapping of the converters. The SS-VOD process, because of the strong stirring achieved using multiple bubblers in the ladle, further enhanced the ability to produce even lower levels of carbon, nitrogen and hydrogen at higher chromium levels.
A major disadvantage of VOD processing is that it is less flexible than an AOD or other converter process with respect to raw materials usage.
Many steelmakers have also realized that vacuum processes often have high operating costs and cannot compete with the ease of operating a converter process at atmospheric pressure. However, the vacuum processes, especially the SS-VOD process, have the unique ability of achieving lower carbon and nitrogen levels in stainless steels which cannot be easily achieved by the AOD process or other converter processes
Direct stainless steelmaking
In recent years, there have been efforts to use chromium and nickel ores for stainless steel making in lieu of the ferroalloys. In Japan, a number of companies have developed and are using such processes commercially.
The direct stainless steel making processes require less scrap and ferro alloys and may be considered when the stainless steel scrap and ferro alloys are not readily available in sufficient quantities. However, chromium and nickel ores are needed and can be used in an integrated steel plant where the hot metal is available. The processes are more capital intensive than the other processes. Therefore, their implementation in other plants and locations is likely to be limited.
Manufacturing process
- Stainless Steel Passivation: Enhancing Corrosion Resistance & Performance
- How Stainless Steel Is Produced: From Smelting to Finished Products
- Stainless Steel: Properties, Manufacturing, and Future Applications
- Carbon Steel vs Stainless Steel: Choosing the Right Material for Your Project
- Effective Stainless Steel Cleaning & Passivation: Protect Against Rust & Corrosion
- Carbon Steel vs Stainless Steel: Choosing the Right Alloy for Your Project
- Steel vs. Stainless Steel: Choosing the Right Metal for Your Project
- Mild Steel vs Stainless Steel: Key Differences Explained
- Copper-Infused Antibacterial Stainless Steel: Enhanced Safety & Corrosion Resistance
- Stainless Steel Grades Explained: How Sulfur & Phosphorus Levels Determine Quality



