Vermiculite: Properties, Production, and Applications
Background
Vermiculite is a group of hydrated laminar magnesium‑aluminum‑iron silicates that expand into long, worm‑like strands when heated. This expansion, known as exfoliation, produces a material with exceptionally low density and thermal conductivity. These traits make expanded vermiculite valuable as a lightweight construction aggregate, thermal insulation filler, soil amendment, and chemical absorbent. When ground into a fine powder, it serves as a filler in inks, paints, plastics, and other products.
History
Thomas H. Webb first documented vermiculite’s unique properties in 1824, noting its worm‑like appearance when heated. The mineral remained a scientific curiosity until the early 20th century, when practical applications prompted commercial interest. The first mining venture began in 1915 in Colorado, but it closed shortly after. Success came in 1923 when the Zonolite Company opened a mine in Libby, Montana, operating until 1990. Today, the world’s largest vermiculite operation is in the Phalabowra district of South Africa. Other major producers include the United States, China, Russia, Brazil, Japan, Zimbabwe, and Australia.
By 1999, the United States operated three active mines—two in South Carolina and one in Virginia—shipping concentrated ore to exfoliation plants nationwide. These facilities also imported approximately 77,000 tons (70,000 metric tons) of concentrated vermiculite from South Africa.
Raw Materials
Vermiculite’s distinctive properties stem from its layered crystal structure and internal water content. The layers hinge like an accordion; when heated, the trapped water flashes to steam, forcing the layers apart. Commercial vermiculite is typically extracted from rocks rich in biotite and iron‑bearing phlogopite. Weathering of these rocks allows water infiltration and chemical reactions that produce vermiculite.
A typical commercial vermiculite composition is 38–46 % silicon dioxide (SiO₂), 16–35 % magnesium oxide (MgO), 10–16 % aluminum oxide (Al₂O₃), and 8–16 % water, with smaller amounts of other oxides. When expanded, the granules are gold‑brown and have a bulk density of 4–10 lb/cu ft (64–160 kg/cu m), depending on particle size.
The Manufacturing Process
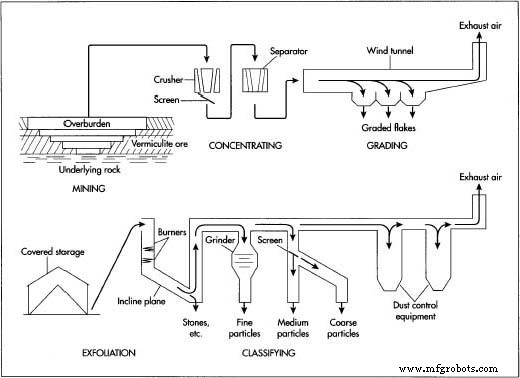
Mining
- Rocks are excavated from open‑pit mines. Overburden is removed with shovels or scrapers, and rock layers are drilled and blasted with explosives.
- The resulting ore is hauled by trucks or rail to a processing facility.
Concentrating
- Ore is crushed and screened to reduce size, then separated from surrounding material using froth flotation, gravity separation, winnowing, or electrostatic methods, yielding about 90 % pure vermiculite flakes.
- Flakes from different sections may be blended to ensure uniform quality.
Grading
- Flakes are sorted by size using screens or an enclosed air‑tunnel. Heavier, larger flakes fall out first; the process can produce grades from 0.63 in (16 mm) down to 0.02 in (0.8 mm).
- Large flakes may be lightly crushed (debooking) to accelerate exfoliation.
- Sorted flakes are packaged in bulk containers for transport to exfoliation plants.
Exfoliating
- Flakes are stored in covered facilities to keep them dry. Moisture absorption would increase energy required for expansion.
- They are loaded onto conveyor belts and dropped into a 20–25 ft (6.1–7.6 m) vertical furnace lined with ceramic bricks. Natural‑gas burners raise the temperature to 1,000–1,500 °F (540–810 °C), causing the internal water to vaporize and the flakes to expand into worm‑like particles.
- Particles slide down an inclined chute, allowing further heating to achieve full expansion.
Classifying
- Hot expanded vermiculite is drawn upward by a vacuum. Heavier contaminants fall out, while the fine particles are cooled by the airflow.
- If a specific particle size is required, the vermiculite may be ground and screened accordingly.
- Classified granules are stored in hoppers and then packed into 4–6 cu ft (0.10–0.15 cu m) paper or plastic bags for retail, or 50 cu ft (1.3 cu m) bags for commercial use. Bags are sealed, labeled, and shipped from warehouses.
Health Aspects
Vermiculite ore can contain mica, quartz, feldspar, and, in some deposits, trace amounts of asbestos. These constituents are regulated under Material Safety Data Sheets (MSDS). The most significant occupational hazard is crystalline silica (quartz). Fine quartz dust can cause silicosis, so dust control and personal protective equipment are mandatory during grinding and screening operations. Consumer exposure to silica dust from finished vermiculite products is negligible and poses no health risk.
Major producers certify that ore bodies used in their operations pose no asbestos risk when processed according to MSDS guidelines. An EPA report in 2000 concluded that vermiculite sold as a soil amendment presents little or no asbestos risk to consumers.
The Future
Despite the availability of alternative materials, vermiculite’s ultra‑low density and thermal conductivity keep it competitive in many markets. Global reserves are estimated at roughly 55 million tons (50 million metric tons). Continued innovation in processing and application will sustain its relevance.
Manufacturing process
- Seismograph: History, Design, and the Future of Earthquake Detection
- Keeping Production Running Efficiently When Business Slows Down
- When a Repair Isn't a Real Fix: How to Identify Genuine Repairs
- Mastering Casting Defects: When to Return or Rework a Casting
- Choosing Urethane Casting: Ideal Applications & Benefits
- Guidelines for Selecting Polypropylene Applications
- Thermoplastics Explained: Definition, Properties, and Recycling Benefits
- Understanding Pilot Lamps: Function, Types, and Safety Applications
- The Critical Role of Surface Lay in Machining Performance
- When to Upgrade Beyond Raspberry Pi for Your Product's Next Phase



