Artificial Heart Valves: Types, History, and Future Innovations
Background
Heart valves act as check valves, opening and closing millions of times each year to regulate blood flow. Natural valves can develop stenosis (narrow opening) or regurgitation (incomplete closure), compromising cardiac output and increasing pulmonary pressure. In the United States alone, over 80,000 adults undergo valve repair or replacement annually.
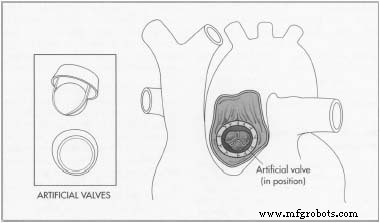
Artificial heart valves comprise an orifice and a mechanical or biological mechanism that opens and closes the passage. There are two main categories:
- Mechanical valves – made from synthetic alloys and polymers. They are durable but require lifelong anticoagulation.
- Biological (tissue) valves – derived from porcine or bovine pericardium, or xenografts. They are preferred for patients over 65 or those who cannot tolerate anticoagulants.
Mechanical valves are further subdivided by their motion mechanisms: ball‑in‑cage, tilting‑disk, and bileaflet (two hinged leaflets). The ball‑in‑cage design, introduced in the 1960s, has seen over 200,000 implantations. Tilting‑disk valves have reached approximately 360,000 procedures, while bileaflet valves – the most common today – exceed 600,000 implantations worldwide.
History
The first documented valve operation dates to 1913, but widespread replacement began only in 1962 with the first successful biological valve using donor human tissue. Mechanical valves followed shortly, with the ball valve pioneered by American Edwards Laboratories in the 1950s. Disk valves entered clinical practice in 1969, and bileaflet designs were introduced in 1977, gaining popularity in the 1980s.
Material advances played a pivotal role. In 1965, Dr. J. C. Bokros discovered that pyrolytic carbon—originally used in nuclear fuel—offers exceptional durability and blood compatibility, leading to its widespread adoption in mechanical valves. Today, roughly 90 % of mechanical valves contain at least one pyrolytic carbon component.
Regulatory oversight intensified in 1976 when the FDA brought prosthetic valves under its jurisdiction, issuing pre‑market approval (PMA) guidelines that set a minimum of 800 valve‑years of follow‑up for clinical studies. Subsequent FDA guidance in 1993 established objective performance criteria, ensuring robust post‑market surveillance.
Mechanical valves have a proven safety record. Over 30 years, the ball valve has experienced fewer than a dozen structural failures. The tilting‑disk shows <1 % failure after 15 years, and bileaflet valves have reported only a handful of issues to the FDA. In the early 2000s, a single manufacturer recalled silver‑coated valves due to a 2 % leakage rate; overall, less than 50 failures have occurred among nearly one million valves in service.
Today, roughly 265,000 prosthetic valves are implanted worldwide each year, generating a market valued at over $700 million. Mechanical valves constitute about 60 % of this volume, with a market worth of $400 million. Over two million mechanical valves have been implanted globally in recent decades.
Raw Materials
Typical mechanical valves are constructed from titanium, graphite, pyrolytic carbon, and polyester. Titanium forms the robust outer housing and lock rings. Graphite coated with pyrolytic carbon creates the bileaflet leaflets, while the inner ring is 100 % pyrolytic carbon, often impregnated with tungsten for radiopacity. The sewing cuff, attaching the valve to the heart, is made from double‑velour polyester.
Titanium is chosen for its strength and biocompatibility. Outer rings are machined from bar stock by third‑party suppliers. Polyester tubes are deburred to remove surface imperfections. The pyrolytic carbon layer is deposited by vaporizing hydrocarbons (usually methane) at 3,272–4,172 °F (1,800–2,300 °C) in a high‑temperature chamber, producing a dense, crack‑resistant film.
Manufacturing Process
- Majority of components are sourced from specialized suppliers; the polyester cuffs are sewn in‑house.
- Assembly occurs in ISO‑clean rooms to prevent contamination. Leaflets are secured to the inner ring, then placed into the housing.
- Concurrent cuff production uses a pressurized heating step that shapes the cuff around the valve at several hundred degrees. The finished valve is mounted on a surgical rotator assembly.
Assembly
- Clean‑room assembly ensures sterility. Leaflets are attached to the inner ring, which is then seated in the outer housing.
- During cuff fabrication, the valve is formed within a heated chamber. The completed valve is fitted to a rotator handle for surgical delivery.
Sterilization and Packaging
- Post‑assembly, valves undergo steam sterilization at 270 °F (132 °C) for at least 15 minutes. A biological indicator confirms sterility by verifying absence of viable organisms.
- Each valve is individually encased in a double plastic container, then boxed for shipping.
Quality Control
Every component undergoes visual, dimensional, and functional inspection before assembly. Rings are measured and matched to leaflets to ensure a perfect fit. Microscopic analysis detects scratches or micro‑defects, with up to 50 inspections during the assembly line.
Proof testing applies controlled pressure to assess structural integrity. Acoustic emission monitoring captures subtle cracks, enabling rejection of sub‑standard valves. Final packaging is verified for accurate labeling.
Byproducts and Waste
Stringent QC minimizes scrap. Recyclable materials are reclaimed where possible, and defective parts are returned to suppliers. Chemical waste from cleaning solutions is disposed of according to safety regulations.
The Future
Ongoing research aims to eliminate thromboembolic risk inherent to mechanical valves. Advanced computational modeling and surgical techniques are refining orifice geometry to reduce turbulence and shear stress. Material innovations—such as ultra‑strong, thin‑wall alloys—allow for reduced valve height and improved hemodynamics.
Minimally invasive approaches are being developed, shrinking incisions from 12 in (30 cm) to 3–4 in (8–10 cm). Manufacturing efficiencies are expected to rise with automation and additive manufacturing.
Emerging concepts include flexible polymer valves that mimic natural dynamics without requiring anticoagulation, and patient‑specific valves engineered from autologous cells. While promising, these technologies may take decades before clinical deployment.
Manufacturing process
- Artificial Heart: Technology, History, and Future Prospects
- Understanding Pneumatic Valve Operation: A Practical Guide
- Maximize Valve Longevity: The Ultimate Guide to Preventative Maintenance
- Key Qualities of High-Performance Pneumatic Valves
- Essential Air Control Valve Maintenance for Optimal System Performance
- What Is a Dump Valve? How They Work and Why They’re Essential
- Understanding Air Cylinder Valves: Key to Pneumatic Systems
- Motorized Ball Valves: Function, Benefits, and Applications
- Mastering the Automobile Valvetrain: Functions, Layout, and Components
- Understanding Servo Valves: How They Control Fluid Flow



