Brass: Composition, History, and Modern Manufacturing
Background
Brass is a copper‑zinc alloy that ranges from a dark reddish‑brown to a light silvery‑yellow, depending on zinc content. With 55–95 % copper and 5–40 % zinc, it is stronger and harder than pure copper but softer than steel. Its excellent thermal conductivity and resistance to salt‑water corrosion make it ideal for pipes, tubes, weather stripping, architectural trim, screws, radiators, musical instruments, and firearm cartridge casings.
History
Metalworkers in the region of modern Syria and eastern Turkey first combined copper and tin around 3000 B.C. to produce bronze. In the centuries that followed, accidental mixing of tin and zinc ores sometimes yielded early brass. By 20 B.C.–A.D. 20, craftsmen could distinguish zinc ore from tin and deliberately alloyed zinc with copper to mint coins and manufacture other items. The process was refined in Germany and the Netherlands from about 300 A.D., but the chemical nature of zinc remained unknown until 1746, when Andreas Sigismund Marggraf identified it as a distinct metal. The first industrial brass alloying process was patented in England in 1781.
In 1852 the first brass cartridge casings were introduced. Brass’s ability to expand and seal under firing pressure, then contract for extraction, enabled the development of rapid‑fire automatic weapons.
Raw Materials
In a typical brass alloy, copper constitutes 55–95 % by weight. High‑copper grades use electrically refined copper of at least 99.3 % purity to limit impurities. Lower‑copper grades often use recycled copper alloy scrap, with copper and alloy percentages carefully measured to achieve the target composition.
Zinc makes up 5–40 % of the alloy. Higher zinc content yields a stronger, harder metal but reduces corrosion resistance and formability. Commercial grade zinc, sometimes called spelter, is commonly used.
Minor alloying elements are added to tailor properties: up to 3.8 % lead improves machinability; tin enhances corrosion resistance; iron hardens the metal and refines the grain structure; arsenic and antimony inhibit corrosion in >20 % zinc alloys; trace amounts of manganese, silicon, and phosphorus may also be present.
Design
Traditional brass names reflected color or application—red brass (≈15 % zinc), yellow brass (≈35 % zinc), cartridge brass (≈30 % zinc), naval brass (up to 39.7 % zinc). Misnomers such as “commercial bronze” (10 % zinc) and “architectural bronze” (40 % zinc + 3.8 % lead) led to the adoption of the Unified Numbering System (UNS). In the UNS, brass alloys start with “C” for copper, followed by five digits: the first digit indicates whether the alloy is wrought (1–7) or cast (8–9).
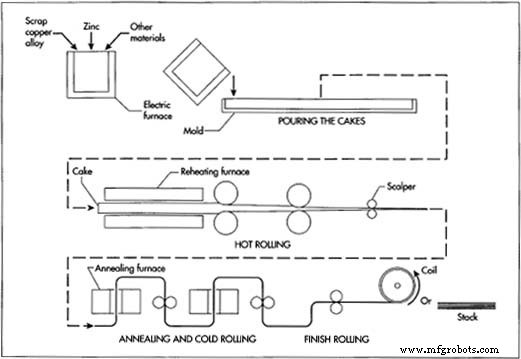
The Manufacturing Process
The production of brass involves a series of controlled steps that transform raw materials into finished stock in various forms—plate, sheet, strip, foil, rod, bar, wire, and billet.
Melting
- Weigh suitable copper alloy scrap and melt it in an electric furnace at ~1920 °F (1050 °C). Adjust for the zinc content in the scrap and add the necessary zinc, plus a 50 % zinc reserve to offset vapor loss.
- Pour the molten alloy into 8 in × 18 in × 10 ft molds to form slabs (cakes). Some operations produce semi‑continuous long slabs.
- Once cooled, transfer the cakes to the rolling area for storage.
Hot Rolling
- Reheat the cakes in a furnace to the target temperature for the desired final shape.
- Pass the heated cakes through opposing steel rollers, gradually reducing thickness to ~0.5 in (13 mm) while increasing width—a process known as breakdown rolling.
- Use a scalper to remove surface oxides from the freshly rolled brass.
Annealing and Cold Rolling
- Anneal the hot‑rolled brass to relieve hardness and restore ductility. Batch or continuous furnaces with a nitrogen atmosphere prevent oxidation.
- Cold‑roll the annealed brass through a second set of rollers to reduce thickness to ~0.1 in (2.5 mm). Cold rolling refines the grain structure, increasing strength and hardness.
- Repeat annealing and cold‑rolling cycles as needed to achieve the target thickness and mechanical properties.
- Convert wide sheets into strips by slitting, then clean with an acid bath and rinse.
Finish Rolling
- Apply a final cold roll to tighten tolerances and provide a smooth surface finish.
- Cut to specification, stack or coil, and store for distribution.
Quality Control
Throughout production, every batch undergoes rigorous inspection. Raw material composition is verified before melting. Process parameters—temperature, time, cooling rate—are tightly controlled. Thickness is measured at each stage, and finished samples are tested for hardness, tensile strength, and dimensional accuracy to ensure compliance with industry standards.
The Future
Brass’s blend of strength, corrosion resistance, and workability secures its role in future applications—from aerospace components to sustainable plumbing. Its high recyclability—most brass products are re‑processed rather than landfilled—ensures a reliable supply chain and aligns with circular‑economy goals.
Manufacturing process
- The Accidental Discovery of Cadmium: From Zinc Ore to Toxic Heavy Metal
- Zinc: Properties, Applications, and Production Process
- 7 Unexpected Uses of Brass That Showcase Its Versatility
- Steel Galvanization: Metallurgical Principles & Protective Strategies
- Professional Steel Galvanizing for Long-Lasting Corrosion Protection
- Bronze vs. Brass: Key Differences & How to Identify Them
- Zinc Phosphate Coating: Edmonton's Premier Corrosion Protection Solution
- Why Brass CNC Machining Delivers Superior Precision & Durability
- Copper vs. Brass: Key Material Properties for Electrical Conductors
- Premium Zinc Plating Services in Melbourne – Protect Your Metal Parts from Corrosion



