Polyurethane: Chemistry, Applications, and Production Overview
Polyurethanes are linear macromolecules featuring carbamate (urethane) linkages (–NHCO2) along their backbone. These linkages form when a diisocyanate reacts with a polyol—a reaction first explored in the late 1930s. Today, polyurethanes are prized for their versatility, finding roles in building insulation, surface coatings, adhesives, solid plastics, and athletic apparel.
Background
Polyurethanes, also known as polycarbamates, belong to the broader class of polymers—macromolecules composed of repeating monomer units. Their defining feature is the presence of carbamate groups (–NHCO2) within the backbone.
Production of synthetic polyurethanes begins with a condensation reaction between a diisocyanate (OCN–R–NCO) and a diol (HO–R–OH). The initial step links the two molecules, leaving a reactive alcohol (OH) on one end and an isocyanate (NCO) on the other. These reactive termini then continue to react with additional monomers, rapidly generating high‑molecular‑weight chains even at ambient temperature. Commercially useful polyurethanes often incorporate other functional groups—esters, ethers, amides, or urea moieties—to tailor performance.
History
German chemist Friedrich Bayer first investigated polyurethane chemistry in 1937, reacting toluene diisocyanate with dihydric alcohols. This work led to Perlon U, one of the earliest crystalline polyurethane fibers. During World II, the search for rubber alternatives spurred the development of elastic polyurethanes; by 1940, the first polyurethane elastomers were produced, offering millable gums suitable as rubber substitutes. The discovery that polyurethanes could be spun into fine threads enabled their combination with nylon, producing lightweight, stretchable garments.
In 1953, the United States began commercial production of flexible polyurethane foam, a material that quickly became essential for insulation. Three years later, cheaper, more flexible foams entered the market, and by the late 1950s moldable polyurethanes were available. Subsequent decades saw the advent of Spandex fibers, polyurethane coatings, and thermoplastic elastomers.
Raw Materials
Polyurethane synthesis employs a range of raw materials: monomers, prepolymers, stabilizers, and colorants.
Isocyanates
Diisocyanates are the key reactive agents. They contain highly reactive isocyanate (NCO) groups. The most common isocyanates are toluene diisocyanate (TDI) and polymeric isocyanate (PMDI). TDI is produced by nitrating toluene, reducing the product to a diamine, and separating the isomers. PMDI results from phosgenation of aniline‑formaldehyde polyamines. Other, higher‑performance isocyanates—such as 1,5‑naphthalene diisocyanate and bitolylene diisocyanate—offer greater hardness and higher melting points for elastomers.
Polyols
Polyols provide the alcohol groups (–OH) that react with isocyanates. Polyether polyols, derived from cyclic ethers via alkylene oxide polymerization, are frequently used. Common examples include polyethylene glycol, polypropylene glycol, and polytetramethylene glycol, and they are chosen for flexible foams and thermoset elastomers. Polyester polyols, often by‑products of terephthalic acid production, are also employed, especially for foams and coatings. While polyester polyols were once dominant, polyether polyols have largely supplanted them due to lower cost and comparable performance.
Additives
Polyurethane formulations are protected from heat, light, atmospheric contaminants, and chlorine by adding stabilizers. Hydroxybenzotriazole serves as a UV screen, antioxidants mitigate oxidation, and tertiary‑amine compounds counteract discoloration from nitrogen oxides. Antimildew agents are added for specific applications. After polymerization, the material is naturally white; colorants—typically dispersed or acid dyes—are incorporated to achieve desired aesthetics.
Design
Polyurethanes are produced in four principal forms: elastomers, coatings, flexible foams, and cross‑linked foams. Elastomers stretch and return to shape, making them ideal for applications requiring strength, flexibility, abrasion resistance, and shock absorption. Thermoplastic polyurethane elastomers can be molded into complex parts, serving as the base for automotive components, ski boots, roller‑skate wheels, cable jackets, and more. When spun into fibers, they become Spandex—used in socks, bras, support hose, swimsuits, and athletic apparel.
Polyurethane coatings offer resistance to solvents, excellent impact durability, and fast curing. They are applied to surfaces that demand abrasion resistance, flexibility, adhesion, and chemical resilience—such as bowling alleys and dance floors. Water‑based polyurethane coatings are popular for aircraft, automobiles, and industrial equipment.
Flexible foams represent the largest polyurethane market, prized for high impact strength and widespread use in furniture cushioning, mattresses, and seat cushions. Rigid, or cross‑linked, foams provide insulation in boards or laminates, extensively used in commercial roofing and building spray‑foam applications.
Semiflexible 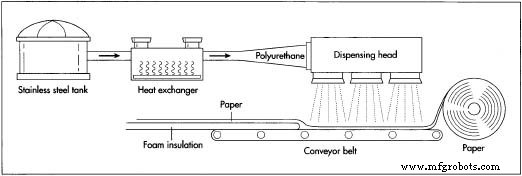 polyurethane foams are employed for car dashboard and door liners, carpet underlay, packaging, sponges, squeegees, and interior padding.
polyurethane foams are employed for car dashboard and door liners, carpet underlay, packaging, sponges, squeegees, and interior padding.
The Manufacturing Process
Polyurethane production can be divided into three stages: bulk polymer synthesis, processing, and final product transformation and shipment. The continuous production of polyurethane foams exemplifies this workflow.
Polymer reactions
- Raw materials—diisocyanates and polyols—are stored as liquids in large, stainless‑steel tanks equipped with agitators to keep them fluid. Metering devices ensure precise dosing; the typical polyol‑to‑diisocyanate ratio is 1:2, a ratio that directly influences polymer properties.
- Materials pass through a heat exchanger before entering reaction pipes. The exchanger adjusts temperature to optimal levels. Polymerization occurs within the pipes, producing polyurethane before the stream reaches the dispensing head.
Processing
- The dispensing head connects to the processing line. For rigid foam insulation, a roll of baking paper is spooled at the start of the line, moved along a conveyor, and positioned beneath the dispensing head.
- Polyurethane is dispensed onto the paper and immediately mixed with carbon dioxide, triggering expansion. The material rises along the conveyor, forming a “bun” that expands like dough.
- As the expansion begins, a second paper layer and optional side papers are added. Each layer encapsulates the foam, giving it shape. The foam bun passes through panels that set its width and height, then dries along the line.
- At the line’s end, an automatic saw cuts the insulation to the desired length. The finished bun is then conveyed to final processing steps—packaging, stacking, and shipping.
Quality Control
Quality assurance starts with incoming raw‑material inspection. Chemists measure pH, specific gravity, viscosity, appearance, color, and odor. Strict control of these parameters ensures a consistent finished product.
After production, the polyurethane product undergoes further testing. Coatings are evaluated for dry time, film thickness, and hardness. Fibers are tested for elasticity, resilience, and absorbency, while foams are checked for density, resistance, and flexibility.
The Future
Polyurethane performance has continually improved since its inception. Ongoing research explores prepolymer modifications to enhance stretchability, the use of advanced fillers, improved catalysts, and optimized prepolymer ratios. Production trends point toward faster, cheaper, and greener processes—replacing volatile toluene diisocyanates with polymeric isocyanates and eliminating chlorinated fluorocarbon blowing agents.
Manufacturing process
- Enhancing Product Durability: Mastering Polyurethane Impact Resistance
- Expert Guide to Machining Polyurethane: Techniques & Best Practices
- Enhancing Polyurethane Conductivity with Conductive Additives
- Polyurethane vs Metal: Why Thermoset Polyurethane Outperforms Traditional Metals
- Polyurethane: The Leading Choice for Superior Abrasion Resistance
- Polyurethane Foam: Composition, Properties, and Diverse Applications
- Polyurethane: Advanced Chemical Resistance for Durable Applications
- Is Polyurethane Foam Safe? Understanding Toxicity & Environmental Impact
- Elastomeric Polyurethanes: High-Performance, Versatile Thermoset Materials
- Polyurethane vs Silicone: Which Material Is Best for Your Design?



