Aluminum: Production, Properties, and Environmental Impact
Aluminum is the third‑most abundant element in the Earth's crust, constituting about 8% of soil and rock. Oxygen and silicon dominate the rest, at 47% and 28% respectively. In nature, aluminum exists only in compounds with elements such as sulfur, silicon, and oxygen. Pure metallic aluminum can be extracted only from aluminum‑oxide ores.
Metallic aluminum is prized for its lightweight, high strength, non‑magnetic, and nontoxic nature. It conducts heat and electricity, reflects light, and maintains strength at extreme temperatures without becoming brittle. A thin, invisible oxide layer forms rapidly on the surface, providing natural corrosion resistance. Moreover, aluminum is one of the most recyclable materials worldwide, with high economic returns from recycled feedstock.
Background
Aluminum compounds have been utilized for millennia. Around 5000 B.C., Persian potters created the toughest clay vessels by adding alumina. Ancient Egyptians and Babylonians used aluminum salts in dyes, cosmetics, and medicines. It was not until the early nineteenth century that the element was isolated in pure form. Because of the difficulty of extracting it, aluminum remained as rare and valuable as silver for many decades.
In 1886, two young scientists—Hall in the United States and Heroult in France—independently developed the Hall–Heroult electrolytic smelting process, making large‑scale production feasible. The Bayer process, patented in 1888 by Austrian chemist Karl Bayer, refined the ore and became essential to the industry’s growth. By 1884, the U.S. produced 125 lb (60 kg) of aluminum, priced roughly equal to silver. In 1995, U.S. plants produced 7.8 billion lb (3.6 million t) of aluminum, while silver was still seventy‑five times more expensive per unit.
Raw Materials
The most valuable ore for producing pure aluminum is bauxite, which contains 45–60% aluminum oxide along with impurities such as sand, iron, and trace metals. While some deposits are hard rock, most are soft, easily mined via open‑pit methods. Australia supplies over one‑third of the world’s bauxite. Roughly 4 lb (2 kg) of bauxite is required to produce 1 lb (0.5 kg) of aluminum.
Caustic soda (sodium hydroxide) dissolves the aluminum compounds in bauxite, separating them from impurities. Depending on ore composition, small amounts of starch, lime, or sodium sulfide may also be added.
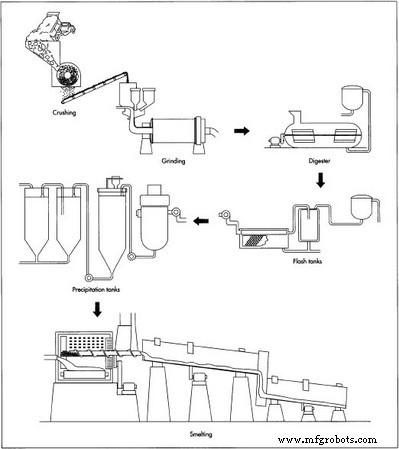
Aluminum production involves two main stages: the Bayer process, which refines bauxite into alumina (Al₂O₃), and the Hall–Heroult process, which electrolytically smelts alumina into pure metal.
Cryolite (Na₃AlF₆) serves as the electrolyte in the smelting step. Historically mined in Greenland, it is now synthesized. Aluminum fluoride is added to lower the electrolyte’s melting point.
Carbon electrodes conduct the electric current through the electrolyte. During smelting, carbon reacts with oxygen to form CO₂. Approximately 0.2 kg of carbon is consumed per 2.2 kg of aluminum produced. Some carbon originates from oil refining by‑products, with the remainder derived from coal.
Because the Hall–Heroult process requires large currents, it consumes significant electrical energy. Producing 1 kg of aluminum typically demands 15 kWh of electricity, which accounts for about one‑third of the smelting cost.
The Manufacturing Process
Aluminum production is divided into the Bayer process and the Hall–Heroult process.
The Bayer Process
- 1. Bauxite is mechanically crushed and mixed with caustic soda, then ground to produce a fine slurry.
- 2. The slurry is pumped into a digester—essentially a pressure cooker—heated to 110–270 °C under 340 kPa for 30 minutes to several hours. Additional caustic soda ensures complete dissolution of aluminum compounds.
- 3. The hot slurry, now a sodium aluminate solution, passes through flash tanks that lower pressure and recover heat for reuse.
- 4. In a settling tank, undissolved impurities settle to the bottom as “red mud.” The soluble aluminum remains in the liquid.
- 5. The clear liquid is filtered through cloth to trap any remaining fine particles. The filter wash yields reusable alumina and caustic soda.
- 6. Seed crystals of alumina hydrate are introduced into a series of six‑story precipitation tanks. As the solution cools, crystals grow and settle to the bottom.
- 7. The crystals are washed, then calcined in a 1,100 °C kiln to remove bonded water, producing anhydrous alumina. A cooler follows the kiln to bring the product to ambient temperature.
The Hall–Heroult Process
Smelting occurs in steel reduction pots lined with carbon. The top electrodes are carbon rods suspended just above the molten aluminum. Pots are arranged in lines (potlines) of 50–200 units, each line producing 60,000–100,000 t of aluminum annually. A typical plant contains two or three potlines.
- 8. Inside each pot, alumina dissolves in molten cryolite at 960–970 °C, forming an electrolyte. A direct current of 4–6 V and 100,000–230,000 A passes through, breaking Al–O bonds. Oxygen reacts with carbon rods to form CO₂; molten aluminum collects at the bottom.
- 9. The process is continuous: alumina is added as it decomposes, while a steady current and heat keep the mixture molten. Periodically, the crust that forms on top is broken to allow more alumina. Siphoned aluminum is collected in a crucible that moves down the potline, accumulating 4,000 kg of 99.8% pure metal. The melt is then cast into long horizontal molds, cooled with water, and cut into ingots.
Byproducts and Waste
Alumina, the intermediate product of the Bayer process, is itself valuable. It is a white, powdery material used in detergents, toothpaste, fluorescent lamps, ceramics (false teeth, spark plugs, military windshield glass), polishing compounds, catalytic converters, explosives, and even rocket fuel—each shuttle launch consumes 180,000 kg of alumina. About 10% of alumina produced annually is sold for these alternative uses.
The primary waste from bauxite refining is “red mud.” For each tonne of alumina produced, roughly a tonne of red mud is generated. Although it contains iron, titanium, soda, and residual alumina, no cost‑effective recovery method exists. It is usually stored in ponds until it dries, then spread over land.
Smelting also generates greenhouse gases. U.S. plants emit approximately 5.5 million t of CO₂ and 3,300 t of perfluorocarbons annually. Additionally, about 120,000 t of spent potlining (SPL)—classified as hazardous by the EPA—is removed each year. The first SPL recycling facility opened in 1996, converting the material into glass frit used in ceramic tiles, glass fibers, and asphalt shingles.
The Future
Most U.S. aluminum producers belong to the Voluntary Aluminum Industrial Partnership (VAIP), collaborating with the EPA to tackle environmental challenges. Current research focuses on developing inert electrode materials to replace carbon. A promising titanium‑diboride‑graphite composite could eliminate greenhouse emissions and cut smelting energy by 25% once commercialized.
Manufacturing process
- Aluminum Foil: Premium, Versatile Material for Thermal Insulation & Industrial Applications
- Aluminum vs. Standard PCBs: Which Is Best for Your Electronics Design
- Aluminum vs. Steel Tooling: Choosing the Right Material for Your Production
- 14 Fascinating Facts About Aluminum: History, Uses & More
- Aluminum Sustainability: Why It’s a Greener Choice
- 6061 vs 7075 Aluminum Alloys: Strength, Versatility, and Application Guide
- Preventing Aluminum Corrosion: Proven Strategies for Long-Lasting Durability
- Aluminum Cladding Explained: Benefits, Uses, and Applications
- EN AW-5005 Aluminum Alloy: Low Density, High Thermal Conductivity & Corrosion Resistance
- AA 7075 T7: Premium High‑Strength Aluminum Alloy for Aerospace



