PEG‑Coated Gold Nanostars: A Safe, High‑Contrast Agent for CT Imaging with Rapid Renal Clearance
Abstract
Gold nanoprobes are emerging as transformative tools for disease diagnostics in computed tomography (CT). Compared to small‑molecule iodine agents, nanoparticles offer superior in‑vivo detection due to their larger surface area and tunable physicochemical properties. Here we report the synthesis of polyethylene glycol (PEG)‑functionalized gold nanostars (AuNS@PEG) that exhibit a high X‑ray mass‑absorption coefficient, ultrasmall size, efficient metabolic clearance, and excellent biocompatibility. In vivo CT scans confirm that AuNS@PEG provide robust contrast enhancement, positioning them as a promising next‑generation contrast agent.
Background
The last decade has seen rapid advances in nanoparticle (NP) technology for biomedical applications, driven by diverse materials and high surface‑to‑volume ratios. Gold (Au) NPs, in particular, are prized for their inert chemistry, biocompatibility, and high atomic number, making them ideal for X‑ray attenuation. Conventional iodinated contrast media—small organic molecules such as diatrizoic acid (DTA) and iohexol (Omnipaque)—provide short‑lived imaging windows and carry a risk of renal toxicity, especially in patients with compromised kidney function. Nanoparticle‑based agents can extend imaging time, reduce nephrotoxicity, and improve contrast efficiency, as demonstrated by Au and Ag nanomaterials in recent studies.
Nanostructuring gold into a star shape increases the effective surface area, amplifying X‑ray attenuation per unit mass. Functionalization with PEG further improves aqueous dispersibility, reduces protein adsorption, and facilitates renal clearance, addressing key challenges of nanoparticle toxicity and biodistribution.
Methods
All experimental protocols were approved by the Regional Ethics Committee, Jinzhou Medical University, Liaoning Province, China.
Materials and Instruments
Reagents were obtained from Sigma‑Aldrich (St. Louis, MO) and used as received unless stated otherwise. AuNS@PEG were characterized by transmission electron microscopy (TEM), energy‑dispersive X‑ray spectroscopy (EDX), X‑ray photoelectron spectroscopy (XPS), UV‑vis spectroscopy, dynamic light scattering (DLS), and MTT viability assays.
Synthesis of Au Nanostars/PEG (AuNS@PEG) Nanoparticles
Seed‑mediated growth produced 10‑nm Au seeds via reduction of HAuCl4 in water, followed by oleylamine injection and subsequent purification. Au nanostars (≈50 nm) were grown by mixing AgNO3 and ascorbic acid with a gold seed solution, then passivated with thiolated PEG (6 kDa) in excess. The final AuNS@PEG were purified by repeated centrifugation/redispersion and stored in water.
Cell Culture and Exposure
Primary rat spinal cord neuroglia were cultured in DMEM supplemented with 10% FBS, 100 U mL−1 penicillin, and 100 µg mL−1 streptomycin. Cells were exposed to AuNS@PEG (50–1000 ppm) for 2 h; controls received media only.
Animals and Treatment
Male Sprague‑Dawley rats (180–200 g) were housed under standard conditions. Two groups were established: control (tail‑vein injection of PBS) and test (200 µg mL−1 AuNS@PEG). After injection, rats were monitored and sacrificed at specified time points for tissue collection.
Cell Viability Assay
Neuroglia were seeded at 1 × 104 cells mL−1 in 96‑well plates and treated with AuNS@PEG (0–1000 ppm). After 24 h, an MTT assay quantified metabolic activity; absorbance was read at 490 nm.
Flow Cytometry
Apoptosis was assessed using annexin V‑APC/7‑AAD staining after 2 h exposure to AuNS@PEG, followed by BD FACSCanto II analysis.
CT Imaging
In vivo CT was performed on a 128‑row GE scanner (120 kV, 100 mA, slice thickness 0.625 mm). Images were acquired pre‑injection and at 0.5, 1, 2, 6, and 24 h post‑injection.
Histological Analysis
Heart, liver, kidney, spleen, lung, and intestine were fixed, sectioned, and stained with H&E for morphological evaluation.
Assessment of Renal Function
Serum BUN, creatinine, β2-MG, and CO2 were measured before and 24 h after injection to evaluate nephrotoxicity.
Statistical Analysis
Data are presented as mean ± SD. One‑way ANOVA with Tukey’s post‑hoc test (p < 0.05) was used to assess significance.
Results and Discussion
Synthesis and Characterization of AuNS@PEG Nanoparticles
TEM images (Fig. 1a) confirm a uniform star‑shaped morphology with an average diameter of ~50 nm. EDX (Fig. 1c) and XPS (Fig. 1b) spectra verify the presence of Au and PEG on the nanoparticle surface, confirming successful functionalization.
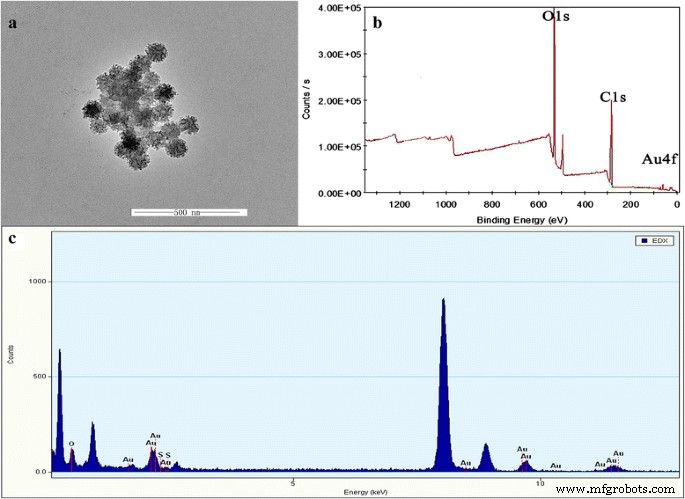
Transmission electron micrographs of AuNS@PEG nanoparticles (a), XPS (b), and EDX (c)
CT Value of the AuNS@PEG Nanoparticles
AuNS@PEG exhibit higher CT attenuation than iodine at equivalent concentrations (Fig. 2a). The linear relationship between CT value (HU) and Au concentration (Fig. 2b) demonstrates predictable imaging contrast.
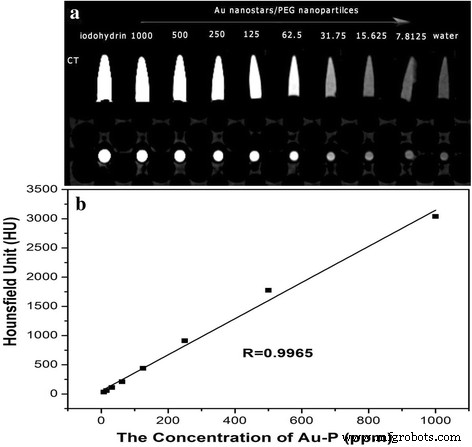
CT attenuation vs. Au concentration (a) and images at varying concentrations (b)
Cytotoxicity Assay
MTT assays show >90% cell viability at 1000 ppm, with no significant cytotoxicity up to 200 ppm (Fig. 3a). Flow cytometry confirms negligible apoptosis across all tested concentrations (Fig. 3b).
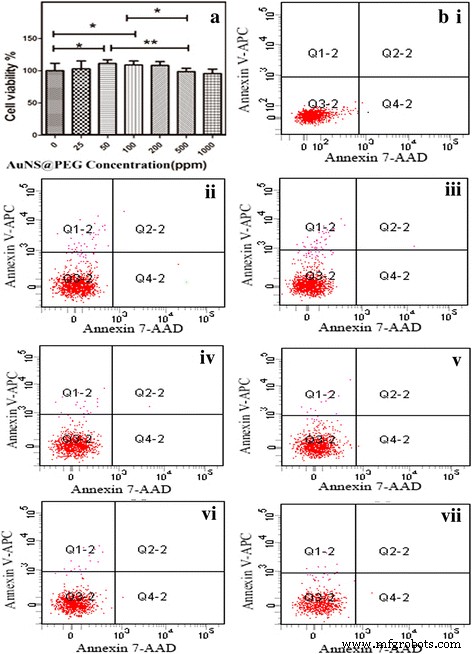
Cell viability (a) and apoptosis profiles (b) across AuNS@PEG concentrations
In Vivo CT Imaging and Biodistribution
Intravenous injection (200 ppm) produced clear renal enhancement, with peak kidney HU values rising from 95 to 464 HU within 2 h (Fig. 4). Subsequent CT scans show progressive clearance, with complete bladder visualization at 24 h, confirming efficient renal excretion.
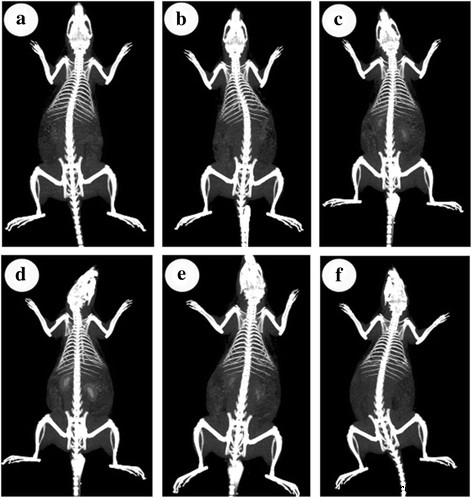
CT images pre‑injection (a) and at 0.5, 1, 2, 6, and 24 h post‑injection (b–f)
H&E Staining
Histology of heart, liver, spleen, lung, kidney, and intestine after 24 h revealed no discernible pathology or residual nanoparticles (Fig. 5).
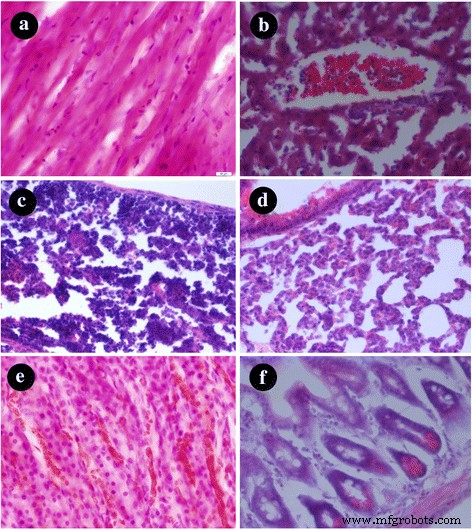
Tissue sections stained with H&E: a heart, b liver, c spleen, d lung, e kidney, f intestine (scale bar 100 µm)
Renal Function Study of AuNS@PEG Nanoparticles
Serum markers (BUN, creatinine, β2-MG, CO2) measured 24 h post‑injection showed no significant changes compared to baseline, indicating no renal impairment (Table 1).
Conclusions
PEG‑coated gold nanostars of ~50 nm diameter exhibit high X‑ray attenuation, excellent biocompatibility, and rapid renal clearance. Their favorable safety profile and superior contrast performance make them strong candidates for clinical CT imaging, offering a safer alternative to iodinated agents.
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Redox‑Responsive Dual‑Targeting Nanoparticles Co‑Deliver Curcumin to CD44‑Positive Tumor Mitochondria
- Cost‑Effective PDMS Nanocone Cluster for Ultra‑Low Solar‑Cell Reflectance and Superhydrophobic Self‑Cleaning
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Highly Biocompatible Au Nanocage@PEG Nanoparticles: A Novel CT Contrast Agent for Enhanced In Vivo Imaging
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy
- Ultrasound-Enhanced Multifunctional Nanoparticles: A Novel Platform for Imaging and Synergistic Therapy of Metastatic Breast Cancer



