Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
Abstract
Modern medicine continually seeks therapies that combine high efficacy with cost‑effectiveness, driving research toward novel delivery platforms that target diseased tissues while sparing healthy cells. Metallic nanoparticles—particularly superparamagnetic core‑shell constructs—offer a unique solution: their magnetic core enables external steering, while the functional shell can carry therapeutic cargo or imaging agents. This review surveys the latest advances in single‑metal, functionalized, and Fe3O4‑based core‑shell nanoparticles, detailing synthesis strategies, biomedical applications, and the challenges that remain before clinical translation.
Introduction
Metal nanomaterials have emerged as a cornerstone of next‑generation medicine. While long‑term safety profiles are still being refined [1], their versatility is evident in applications ranging from in‑vivo imaging [2–4] to targeted therapy for cancer [5–10], neurodegeneration [11–13], HIV/AIDS [14–16], ocular [17–19], and respiratory diseases [20–21]. Figure 1 illustrates this breadth. Yet, practical hurdles—such as reproducible synthesis, size and shape control [22–28], and scalable production [29]—continue to limit widespread adoption. Moreover, the potential for chronic toxicity remains largely unexplored [30–32]. In this context, metal nanoparticles present an attractive platform for drug delivery [33–34] owing to their high surface area and tunable surface chemistry [35–36]. Incorporating a magnetic core further enhances control over particle localization, albeit with new synthesis challenges [37–38].
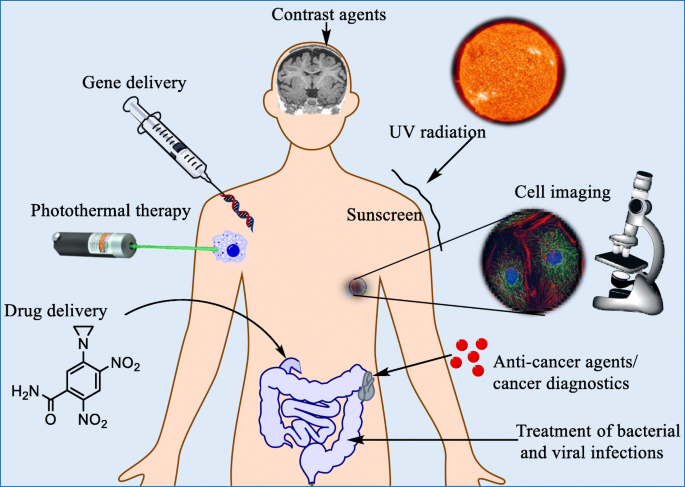
Current uses of nanoparticles in medicine
This review first examines core‑shell synthesis methods—particularly Fe3O4 cores with gold or silver shells—before exploring their biomedical roles, limitations, and future prospects.
Synthesis of Core‑Shell Nanoparticles
Metal nanoparticle synthesis spans classical routes such as gas deposition [40], sol‑gel [41], and aerosol/vapor phase [42] to more contemporary approaches. A persistent challenge arises when creating core‑shell architectures: ensuring a uniform, controllable shell over a chemically distinct core, particularly when the core (e.g., Fe3O4) is prone to oxidation and the shell precursor (e.g., HAuCl4) is a strong oxidant [38]. Two dominant strategies exist: direct metal deposition onto the core [44] and the use of an intermediary “glue” layer [45]. The following subsections detail representative techniques.
Reverse Micelle Synthesis
Reverse micelles—self‑assembled structures where hydrophilic heads face an aqueous core surrounded by a hydrophobic tail—provide a confined reaction environment ideal for core‑shell formation [46]. The method originated in the 1980s with colloidal rhodium, platinum, and palladium nanoparticles [47]. Two common emulsion geometries are water‑in‑oil (w/o) and water‑in‑supercritical CO2 (w/sc‑CO2) [53–56]. The latter offers a greener alternative by eliminating toxic organic solvents [57]. In the seminal work on Au‑Fe3O4 NPs [59], a H2O/CTAB system generated micelles that, upon reduction with NaBH4, coated iron cores with gold, yielding an average diameter of 12 nm. Subsequent refinements [60, 61, 62, 63, 64] varied surfactants, co‑surfactants, and reductants (e.g., hydrazine) to tailor size (5–35 nm) and optical properties (UV/vis shift 526 nm → 555 nm).
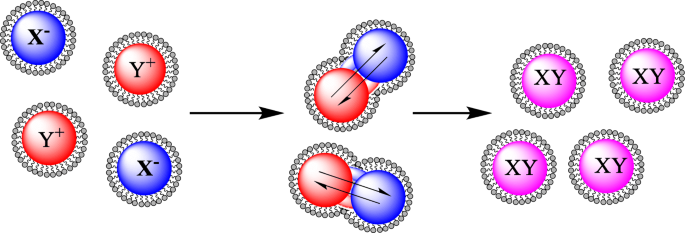
A schematic of reverse micelle‑mediated core‑shell formation
Alternative systems—such as cyclohexane/Brij‑97 with FeSO4 and FeCl3 [65]—have produced 13 nm particles coated with silver or gold [46, 58]. Tamer et al. demonstrated a co‑precipitation approach that yielded 23.5 nm Au‑Fe3O4 NPs, which were subsequently functionalized with a self‑assembled monolayer for bacterial detection [62]. Zhang et al. introduced laser‑initiated reduction within CTAB micelles, achieving 18 nm cores with ±36 nm dispersion [66].
Thermal Synthesis
Thermal routes involve heating reaction mixtures above their boiling point, often under reflux [67–69]. Hydrothermal (water‑based) and solvothermal (organic‑based) methods [70–72] typically require separate core synthesis and shell deposition steps [68, 69, 72, 74, 77, 79–81]. Rudakovskaya et al. added Fe3O4 nanoparticles to boiling HAuCl4, producing 30 nm spherical Au‑Fe3O4 NPs (Fig. 3) [76].
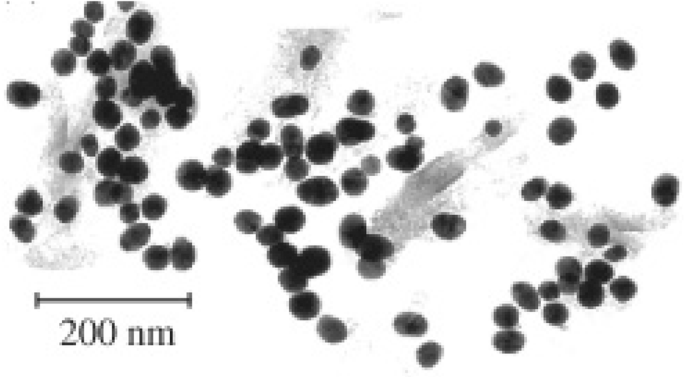
Hydrothermal Au‑Fe3O4 nanoparticles (average 30 nm)
Colloidal Synthesis
Colloidal methods—mixing metal ions with a reducing agent in an aqueous medium at controlled temperatures—offer simplicity and solvent‑free synthesis [82–84]. Advantages include room‑temperature operation and avoidance of toxic solvents, though size polydispersity and shape control remain challenges [85–86]. For gold‑coated iron oxide, colloidal routes employ various reductants: ascorbic acid [84], gold acetate [95], or ethanol‑mediated reduction [83]. The latter yields 30–100 nm particles with diverse morphologies (Fig. 4), indicating rapid but less selective growth [83].
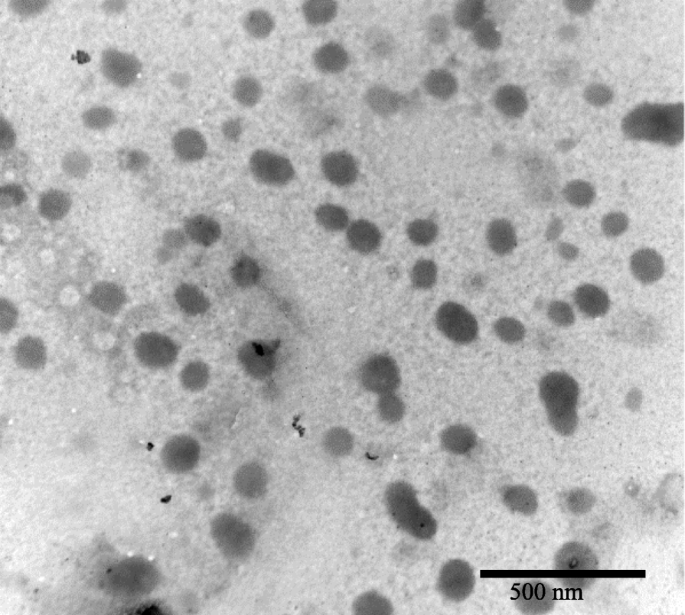
Colloidal Au‑Fe3O4 nanoparticles (20–100 nm)
Seeding approaches—binding pre‑formed gold seeds to the iron core—improve shell uniformity [91, 92, 97]. For example, Goon et al. employed polyethyleneimine to achieve complete coverage, albeit with high polydispersity (40–110 nm) [91]. Levin et al. used organosilane‑functionalized cores to narrow the size range to 50–70 nm [92].
Bio‑Medical Applications of Metal Nanoparticles
Antimicrobial Agents
Metal nanoparticles have revitalized antimicrobial strategies, offering alternatives to conventional antibiotics that face rising resistance. Silver nanoparticles (AgNPs) consistently demonstrate broad‑spectrum activity: a study incorporating AgNPs into antimicrobial fibers achieved 94.3% inhibition of E. coli [102], while silver‑decorated vanadate nanowires outperformed oxacillin against MRSA [105]. Verma et al. reported AgNPs with a minimum inhibitory concentration of 5.83 µg/mL against Pseudomonas fluorescens, E. coli, and Candida albicans, outperforming ampicillin and neomycin [106]. Gold nanoparticles, especially when functionalized (e.g., thioguanine‑capped), also exhibit potent antibacterial effects against E. coli, Aspergillus fumigatus, and Pseudomonas aeruginosa [107], and naked AuNPs inhibit growth of S. aureus, Klebsiella pneumoniae, and Bacillus subtilis at sub‑µg/mL doses [109].
Antiviral
AgNPs have shown efficacy against a spectrum of viruses. Small (10–50 nm) AgNPs inhibit Hepatitis B by binding dsDNA and suppressing viral RNA [113]. Polysaccharide‑coated AgNPs reduce monkeypox plaque formation across concentrations of 12.5–100 µg/mL [114]. For HIV, AgNPs (30–50 nm) block gp120–cell receptor interaction with IC50 of 0.44 mg/mL [116]; PVP‑coated AgNPs prevent transfection in cervical tissue models while stimulating lymphocyte proliferation [115]. Gold nanoparticles—2 nm Au‑MES—target HIV‑1 gp120 and neutralize infection [118], while copper‑iodide NPs inactivate feline calicivirus and H1N1 via ROS generation [119, 120].
Imaging
Superparamagnetic core‑shell particles enhance contrast in magnetic resonance imaging (MRI). Gold‑coated iron nanoparticles successfully improve T1 and T2 weighted imaging, and their PEGylated dendrimer conjugates outperform iodinated agents like iopamidal in CT contrast [134–135]. AuNPs have also been employed for photoacoustic imaging, providing high spatial resolution due to efficient light absorption and heat conversion [137–142]. Darkfield microscopy demonstrates clear intracellular localization of AuNPs targeting EGFR in A431 cells (Fig. 6) [144].
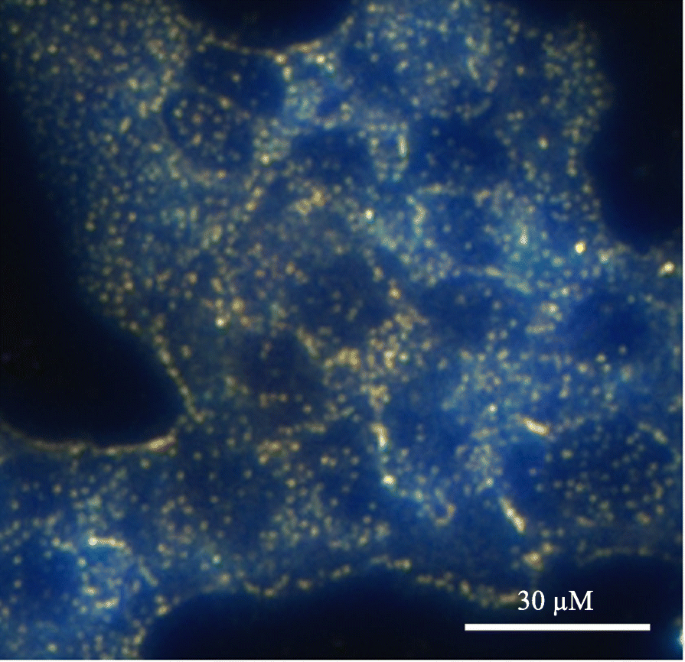
Darkfield imaging of A431 cells treated with AuNPs; bright spots indicate nanoparticle uptake
Biomedical Cargo Delivery
Nanoparticles’ high surface‑to‑volume ratio facilitates drug loading and targeted delivery. Gold nanoparticles have been engineered for trans‑mucosal insulin delivery, achieving significant hypoglycaemia in diabetic rats after nasal and oral administration [149]. Smart AuNPs—citraconic amide‑functionalized particles—display dual charge switching, enabling rapid aggregation in tumour tissue while remaining dispersed in normal tissue, thereby enhancing photothermal therapy selectivity [138, 150]. Nucleic‑acid conjugation (DNA, siRNA) is also well‑established: Yonezawa et al. produced DNA‑bound AuNPs forming wire‑like hybrids [155], while Rosi et al. achieved antisense oligonucleotide functionalization on 13 nm AuNPs [152]. Such constructs open avenues for gene‑targeted therapies [156–157].
Anticancer Drug Delivery
Gold nanoparticles serve as versatile carriers for chemotherapeutics. PEGylated AuNPs loaded with tumor necrosis factor (TNF) preferentially accumulate in MC‑38 colon carcinoma cells, reducing tumour mass more effectively than free TNF [158]. AuNP‑doxorubicin conjugates mitigate cardiotoxicity while maintaining anticancer efficacy [160]. Folic acid‑coated AuNPs demonstrate selective uptake by FR‑positive tumour cells, illustrating ligand‑mediated targeting [159].
Limitations of Single Metal Nanoparticles and Overcoming Them
The principal hurdle is precise in‑vivo targeting. Conventional ligands—antibodies [164–166] and homing peptides [167–168]—often face biological barriers such as the blood–brain barrier [169]. Magnetic guidance offers a compelling alternative: superparamagnetic particles can be steered with external fields, enhancing localization while reducing off‑target effects [170, 171]. Nonetheless, many magnetic cores raise toxicity concerns [31, 174, 175]; iron oxide, while FDA‑approved for imaging, can elicit adverse responses in cell‑labeling studies [176–178]. Coating strategies—silica, polymers, gold, or silver—mitigate toxicity and enable functionalization [180–184]. Gold shells, in particular, are biocompatible and facilitate conjugation of therapeutic agents [185, 187]. Thus, core‑shell architectures combine magnetic steering with safe, functional surfaces, surpassing non‑magnetic counterparts in both delivery and imaging efficacy [190–191].
Current Medicinal Uses of Gold‑Coated Iron Oxide Nanoparticles
Gold‑shell superparamagnetic nanoparticles have rapidly entered translational research. Kayal and Ramanujan’s in‑vitro circulatory system demonstrated magnetic capture of doxorubicin‑loaded Au‑Fe3O4 particles, underscoring clinical potential [194]. In photothermal therapy, Bhana et al. combined photothermal and photodynamic modalities against KB‑3‑1 and SK‑BR‑3 cells, achieving a 64% viability reduction at lower laser powers [197]. Kirui et al. reported enhanced apoptosis in SW1222 colorectal cancer following gold‑shell treatment, attributed to higher uptake [199]. Imaging studies by Cho et al. validated Au‑Fe3O4 nanoparticles for T1 and T2 MRI and as biosensor platforms [202]. Cheng et al. showcased dual magnetic targeting and photothermal ablation in a mouse tumour model, with minimal toxicity at 1.6 mg/kg and 1 W/cm2 laser power [203]. These findings illustrate the versatility of core‑shell constructs across diagnostics and therapeutics.
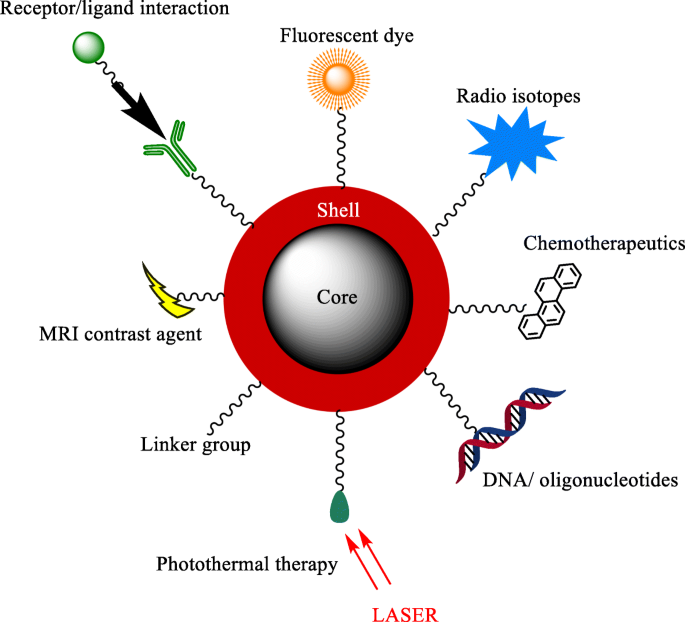
Illustrative applications of core‑shell nanoparticles
Conclusions
Single‑metal nanoparticles already exhibit diverse biomedical functionalities, yet precise targeting remains the critical barrier to clinical adoption. Embedding a superparamagnetic core within a biocompatible shell—most often gold—offers a robust solution, enabling magnetic steering, reduced toxicity, and functional conjugation. Current synthesis methods (reverse micelle, thermal, colloidal, and seeding) each present trade‑offs in reproducibility, size control, and scalability. Addressing these limitations—particularly achieving uniform, reproducible particle populations at scale—will be essential for routine medical use. Ongoing research into long‑term biocompatibility will further solidify the role of core‑shell nanoparticles in the next era of precision medicine.
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Spherical Gold–Cockle Shell Calcium Carbonate Nanoparticles: Fabrication, Characterization, and Cytotoxicity for Biomedical Use
- Magnetite Nanocluster-Based Theranostic Agents for T2‑Weighted MRI and pH‑Responsive Doxorubicin Delivery
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Biotin‑Functionalized Redox‑Sensitive Chitosan Nanoparticles Coated with Phycocyanin for Targeted Curcumin Delivery
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



