Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
Abstract
The use of cells as carriers to encapsulate chemotherapy agents has emerged as a promising strategy in oncology. By reducing systemic toxicity, improving tumor targeting, and enhancing drug penetration, cell‑based delivery systems offer significant clinical potential. Recent advances have demonstrated that drugs encapsulated within erythrocytes, leukocytes, platelets, immune cells, and even tumor cells can outperform free chemotherapeutics. Moreover, multifunctional platforms now enable simultaneous delivery of chemotherapeutics, photosensitizers, and photothermal agents, providing synergistic treatment against drug‑resistant and metastatic cancers. Nonetheless, most studies remain preclinical; key challenges—such as long‑term preservation of drug‑laden cells, safe infusion protocols, and immune compatibility—must be addressed before clinical translation.
Introdution
Cancer remains one of the most formidable diseases worldwide. Conventional chemotherapy, while effective, is hampered by systemic toxicity and the emergence of drug resistance [1]. In recent years, researchers have turned to cellular and nanoparticle (NP) encapsulation strategies to mitigate these drawbacks. Direct intravenous injection of chemotherapeutics often produces severe side effects, limited tumor penetration, and poor aqueous solubility. Cell‑based drug loading—particularly via hypotonic swelling—offers a promising solution, though it can compromise membrane integrity. Hybrid systems that combine NPs with cell membranes have emerged as a hot research area, enhancing biocompatibility, tumor targeting, and circulation time [3,4]. Surface modification of carrier cells to improve tumor homing has become a pivotal focus, enabling higher intratumoral drug concentrations and improved therapeutic outcomes [5,6]. This review surveys recent developments in drug and NP encapsulation across erythrocytes, platelets, immune cells, tumor cells, and stem cells (Table 1) [7–35].
Erythrocyte
Erythrocytes are attractive carriers due to their intrinsic biocompatibility, long lifespan, and abundance. Early approaches loaded drugs directly into red blood cells; subsequent work incorporated functional NPs, enabling a transition from simple chemotherapy to targeted therapy, immunotherapy, and photothermal therapy (PTT) [36]. Erythrocyte membranes can fuse with NPs via several methods—lipid insertion, biotin‑avidin bridging, EDC/NHS coupling, antibody/ligand–receptor conjugation, and passive adsorption (Fig. 1) [36]. Hybrid membranes combining erythrocyte and cancer cell membranes have demonstrated immune camouflage and tumor‑targeting capabilities [4]. By regulating blood circulation time and tumor microenvironment interactions, a magnetofluorescent nanocarrier cloaked in erythrocyte membrane achieved a 92‑hour circulation half‑life and high tumor uptake in murine models [8]. Targeting splenic antigen‑presenting cells with aged erythrocytes, combined with PD‑L1 blockade, inhibited tumor growth and metastasis in vivo [9]. For PTT, Prussian blue (PB) NPs offer strong photothermal conversion and magnetic properties but suffer from rapid clearance; erythrocyte coating extends circulation and tumor accumulation [10]. Mimetic vesicles derived from red blood cells, engineered through fracture and self‑assembly, allow precise size control and have been used to co‑deliver P‑glycoprotein siRNA and doxorubicin to multidrug‑resistant tumors, demonstrating synergistic anticancer effects [11]. Size and surface phosphatidylserine exposure critically influence erythrocyte‑derived carrier pharmacokinetics; microsized carriers exhibit longer half‑lives than nanosized ones, and repeated dosing further prolongs circulation [39]. These insights underscore the necessity of detailed mechanistic studies to optimize erythrocyte‑based platforms for clinical use.
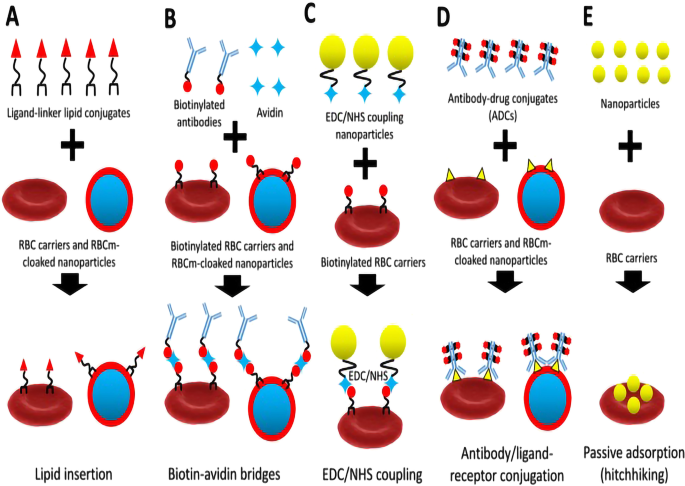
Reprinted with permission from Ref. [30]. Copyright © 2019, Theranostics
Schematic diagram of binding method between erythrocyte membrane and NPs (A), biotin‑avidin bridges (B), EDC/NHS coupling (C), antibody/ligand‑receptor conjugation (D), and passive adsorption (hitchhiking) (E) methods for refunctionalization of erythrocyte-based nanomedicine.
Platelet
Platelets, anucleate blood cells with an 8–10‑day lifespan, present an attractive balance between prolonged circulation and reduced nonspecific accumulation compared to erythrocytes [41]. The CD47 “don’t eat me” signal on platelets mitigates macrophage phagocytosis, enhancing tumor retention. Platelets also contribute to tumor angiogenesis and metastasis via VEGF, MMPs, EGF, and PDGF, stimulating MAPK and AKT pathways [42]. These properties make platelet‑derived carriers suitable for targeting tumor vasculature and circulating tumor cells (CTCs). Platelet membranes are typically prepared by ultrasound or freeze–thaw cycles [41]. Hu et al. demonstrated that platelets can encapsulate NPs, achieving superior tumor targeting and therapeutic efficacy relative to free drugs [43]. Liu et al. developed a pH‑responsive platelet‑lipid hybrid carrier that extended plasma half‑life and enhanced doxorubicin accumulation in tumors, outperforming conventional pH‑sensitive liposomes (Fig. 2) [18]. Recent work combines platelet membranes with photothermal agents and immunotherapeutics; after thermal ablation, the inflamed tumor microenvironment recruits drug‑loaded platelets, amplifying immune activation and reducing residual tumor burden [19]. Collectively, platelet‑based DDSs hold considerable promise for multimodal cancer therapy.
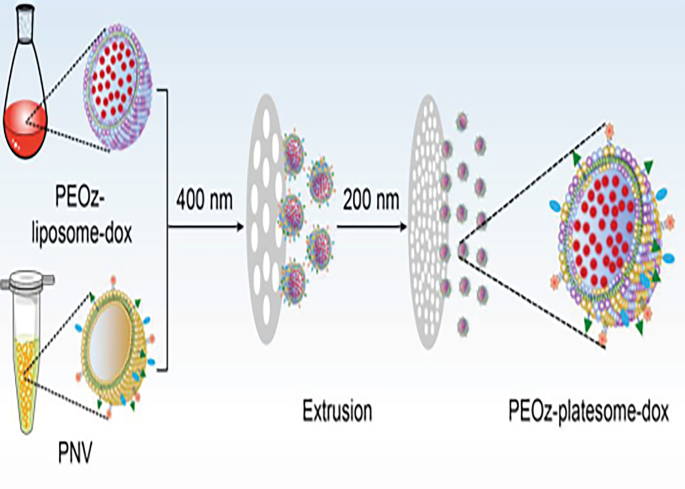
Reprinted with permission from Ref. [14]. Copyright © 2019, Wiley
Schematic illustration of the preparation of PEOz-platesome-dox. PEOz-platesome-dox was generated by coextrusion of PEOz-liposome-dox and PNV.
Immunological Cell
Immune cells naturally home to sites of inflammation and tumor tissue, offering an intrinsic targeting advantage. Macrophages, monocytes, cytotoxic T lymphocytes (CTLs), leukocytes, neutrophils, natural killer (NK) cells, and other leukocytes have been explored as drug carriers. Macrophage‑derived nanocarriers enhance tumor permeability and can be combined with photothermal agents to potentiate antitumor activity [23,24]. CTLs, with their prolonged circulation and high adhesion molecule expression, can be engineered to deliver therapeutic NPs directly to tumor sites [26]. Leukocytes, comprising 4–10 × 10^9 cells/L in healthy adults [45], exhibit robust migration toward inflamed vasculature. DOX‑loaded leukocytes demonstrated superior tumor suppression and survival extension in breast cancer and melanoma models [27,47]. Neutrophils, the most abundant leukocyte type, rapidly accumulate at inflammatory foci and can serve as delivery vehicles for cRGD liposomes across the blood–brain barrier in cerebral ischemia models [49]. NK cell membranes, rich in receptors for tumor antigens, enable targeted delivery of photodynamic agents, producing strong antitumor immunity and inhibiting distant metastases [28]. These studies highlight the versatility of immune‑cell‑based DDSs in delivering chemotherapeutics, immunomodulators, and phototherapies.
Cancer Cell
Cancer cells express homotypic adhesion molecules—such as E‑cadherin, N‑cadherin, and galectin‑3—that facilitate cellular aggregation and tumor progression. Harnessing these molecules, researchers have coated nanoparticles with cancer‑cell membranes to achieve homologous targeting. Chen et al. created indocyanine green (ICG)‑loaded polymeric cores cloaked in MCF‑7 cell membranes, achieving deep tumor penetration and dual‑modal imaging guidance for photothermal therapy in vivo (Fig. 4) [29]. Targeting mitochondria—a critical regulator of apoptosis and multidrug resistance—has also emerged as a strategy. Metal‑oxide NPs (e.g., Zn‑doped CuO) co‑loaded with docetaxel and lonidamine exhibit strong mitochondrial targeting and potent antitumor efficacy [52,54]. These approaches demonstrate the potential of tumor‑cell membrane‑based carriers for precise, multimodal cancer therapy.
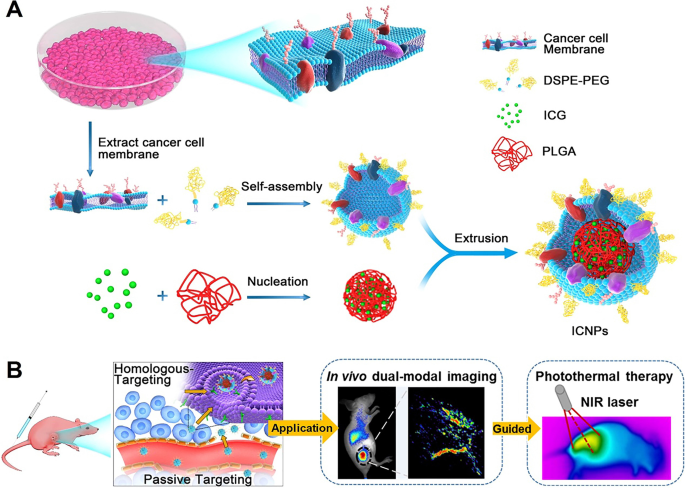
Reprinted with permission from Ref. [24]. Copyright © 2016, ACS Publication
Illustration of the cancer cell membrane–biomimetic NPs for targeting recognition of source cancer cell, dual‑modal imaging, and photothermal therapy. (A) Preparation procedure of ICNPs. Extracting cancer MCF‑7 cell membrane hybridized with PEGylated phospholipids (DSPE‑PEG) and then coated onto ICG‑loaded polymeric cores by extrusion. (B) Schematic of homologous targeting ICNPs for dual‑modal imaging guided photothermal therapy. Through specific homologous targeting and the EPR effect (passive targeting), ICNPs realized perfect tumor accumulation, dual‑modal FL/PA imaging, and effective photothermal therapy after intravenous injection.
Stem Cell
Mesenchymal stem cells (MSCs) are recruited by tumor‑derived chemokines to support tumor growth and metastasis [55]. MSCs possess self‑renewal, multilineage differentiation, and immunomodulatory capabilities. Their natural tropism toward injured or inflamed tissues makes them attractive drug carriers. MSCs derived from umbilical cord have been shown to inhibit pancreatic cancer growth in murine models [57,58]. Genetically engineered MSCs can be expanded in vitro and serve as vectors for viral gene delivery [59]. Recent work has leveraged MSCs as “Trojan horses” for photodynamic therapy: Chlorin e6‑coupled polydopamine NPs were loaded into MSCs, which trafficked to tumors and released the photosensitizer via an endocytosis–exocytosis–endocytosis cycle, achieving a 60% release within 72 h and significant tumor suppression in osteosarcoma and melanoma models [33,34]. These findings underscore MSCs’ potential to overcome drug resistance and deliver therapeutics to otherwise inaccessible tumor sites.
Development
Cell‑derived drug delivery systems retain surface proteins that preserve native biological functions, enhancing biocompatibility, immune evasion, and targeted accumulation while mitigating protein corona formation. Nanoparticles, typically ~100 nm in diameter, are readily cleared by the reticuloendothelial system; coating them with cell membranes dampens macrophage activation and prolongs circulation [60]. The incorporation of fluorescent dyes such as ICG facilitates real‑time imaging of biodistribution and therapeutic efficacy, bridging diagnosis and therapy [62]. Despite these advantages, most cell‑based DDSs remain confined to preclinical studies. Critical gaps include potential immunogenicity of modified membranes, timing and control of drug release, and the fate of biodegradable NPs within the human body. Addressing these issues will be essential for translating cell‑based platforms into clinical practice [63].
Conclusions
Compared with conventional chemotherapy, cell‑based drug delivery systems improve biocompatibility, immune evasion, circulation time, and targeting precision. Encapsulating chemotherapeutics, photosensitizers, and other agents within erythrocytes, platelets, leukocytes, or tumor cells enables synergistic multimodal treatment strategies. Current research predominantly focuses on the combination of blood‑cell membranes with nanoparticles, leveraging the enhanced permeability and retention effect, high drug loading capacity, and inherent targeting properties of both platforms to advance cancer therapy.
Availability of Data and Materials
Not applicable.
Abbreviations
- NPs:
Nanoparticles
- PTT:
Photothermal therapy
- MMFn:
Magnetofluorescent nanocarrier
- PD-L1:
Programmed death ligand‑1
- APCs:
Antigen‑presenting cell
- PB:
Prussian blue
- DDS:
Drug delivery system
- EV:
Extracellular vesicle
- MVs:
Mimic vesicle
- P‑gp:
P‑glycoprotein
- DOX:
Doxorubicin
- P‑MPs:
Platelet‑derived microparticles
- VEGF:
Vascular endothelial growth factor
- MMP:
Matrix metalloproteinase
- EGF:
Epidermal growth factor
- PDGF:
Platelet‑derived growth factor
- CTCs:
Circulating tumor cells
- CTLs:
Cytotoxic T lymphocytes
- TEM:
Tumor microenvironment
- NK:
Natural killer cell
- MDR:
Multidrug resistance
- TPP‑ZC‑IR‑PNPs:
Zn‑doped CuO NPs
- cl‑M/DL:
Co‑loaded with docetaxel and lonidamine
- MSCs:
Mesenchymal stem cells
- Ce6:
Chlorin e6
- PDA:
Polydopamine
- ICG:
Indocyanine green
- EPR:
Enhanced permeability and retention effect
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- Efficient Colon Cancer Gene Therapy Using Cationic DOTAP–PEG‑PCL Micelles for siRNA Delivery
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Chitosan‑Capped, Enzyme‑Responsive Hollow Mesoporous Silica Nanoplatforms for Targeted Colon Drug Delivery
- Microwave‑Reduced, Chitosan‑Coated Graphene Oxide for Targeted Adriamycin Delivery and Enhanced Breast Cancer Therapy
- Quantitative Study of PLGA Nanoparticle Uptake in Laryngeal Cancer and Immune Cells to Improve Drug Delivery
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



