PEG‑Modified Cadmium Telluride Quantum Dots Deliver Doxorubicin for Targeted Treatment of Extramedullary Multiple Myeloma
Abstract
Extramedullary multiple myeloma (EMM) remains a therapeutic challenge, with high‑dose chemotherapy offering modest survival benefits that are frequently offset by severe systemic toxicity. In this study, doxorubicin (DOX) was encapsulated in polyethylene glycol‑modified cadmium telluride quantum dots (PEG‑CdTe QDs), creating a nanocarrier that enhances intracellular drug accumulation and releases DOX in a pH‑controlled manner. In vitro, PEG‑CdTe‑DOX markedly increased apoptosis in PRMI8226 myeloma cells compared to free DOX, and modulated the expression of key apoptosis‑associated proteins. These results demonstrate that PEG‑CdTe‑DOX offers a highly selective and potent therapeutic strategy for patients with EMM.
Introduction
Multiple myeloma (MM) is the second most common hematologic malignancy, characterized by clonal proliferation of plasma cells in the bone marrow. Although advances in chemotherapy and targeted agents have extended overall survival, the subset of patients with extramedullary disease (EMM) continues to have a dismal prognosis, with median survival of fewer than six months after relapse. Current therapeutic options, including thalidomide and bortezomib, provide limited benefit for EMM, whereas high‑dose chemotherapy can improve outcomes but is frequently intolerable due to cardiotoxicity and marrow suppression.
Doxorubicin remains a cornerstone in MM therapy; however, its clinical use is constrained by dose‑dependent side effects, particularly in the elderly population that constitutes the majority of MM patients. Nanoparticle‑based drug delivery systems offer a promising solution by extending systemic circulation, enabling controlled release, and concentrating therapeutic agents at tumor sites while sparing healthy tissues.
Cadmium telluride quantum dots (CdTe QDs) exhibit unique optical properties and can be functionalized with polyethylene glycol (PEG) to achieve “immune‑evasion,” reducing opsonization and prolonging blood residence time. PEGylated CdTe QDs have been shown to internalize efficiently via fluid‑phase endocytosis and release payloads in response to acidic microenvironments typical of tumors. In this work, we fabricated a PEG‑CdTe‑DOX nanoconjugate and evaluated its physicochemical characteristics, pH‑responsive drug release, cellular uptake, cytotoxicity, and induction of apoptosis in a human EMM cell line.
Materials and Methods
Materials
PEG‑CdTe QDs, the Pierce™ bicinchoninic acid (BCA) protein assay kit, and doxorubicin (DOX) were purchased from Sigma‑Aldrich, Dalian Meilun Biology Technology Co., and other suppliers as listed in the original manuscript. PRMI‑1640, FBS, Annexin V‑FITC, CCK‑8, and related reagents were obtained from standard commercial sources. Antibodies against Bax, Caspase‑3, Bcl‑2, and β‑actin were sourced from Cell Signaling Technology. Analytical instrumentation included a TEM (Japan Electron Optics Laboratory), a Zetasizer NanoZs (Malvern), HPLC (Jasco), a Nikon C‑2 confocal microscope, a BD Accuri C6 flow cytometer, and a Tanon 5200 ECL detection system.
Preparation of PEG‑CdTe‑DOX
DOX was adsorbed onto PEG‑CdTe QDs via electrostatic interactions by mixing equal volumes (100 µl) of 1 mg/ml PEG‑CdTe and 1 mg/ml DOX in 800 µl deionized water, stirring at 100 rpm for 24 h at 37 °C in the dark. The mixture was centrifuged (30 min, 4 °C, 30,000 rpm) to collect PEG‑CdTe‑DOX, which was washed twice to remove unbound DOX. Encapsulation efficiency (EE) and drug loading (DL) were calculated as follows:
$$\text{DL}=\frac{\text{Weight of drug in NPs}}{\text{Weight of NPs}}\times 100\%$$
$$\text{EE}=\frac{\text{Weight of drug in NPs}}{\text{Initial weight of drug}}\times 100\%$$
Characterization
The morphology of PEG‑CdTe and PEG‑CdTe‑DOX was examined by TEM. Hydrodynamic diameters and size distributions were measured by dynamic light scattering (DLS) in PBS. To assess pH‑responsive release, PEG‑CdTe‑DOX (1 mg/ml DOX) was dialyzed (MWCO 3.5 kDa) against PBS at pH 5.0, 6.0, 7.4, and 8.0, with samples collected every 2 h for 24 h. DOX concentration was quantified by spectrophotometry at 450 nm.
Confocal Fluorescence Microscopy
PRMI8226 cells were incubated with PEG‑CdTe, free DOX, or PEG‑CdTe‑DOX for 4 h, fixed with DAPI, and imaged with a Nikon C‑2 confocal microscope. Emission wavelengths: DAPI 450 nm; DOX 585 nm.
Cell Culture
PRMI8226 and 293T cell lines were maintained in RPMI‑1640 and DMEM respectively, supplemented with 10 % FBS, 100 U/ml penicillin, and 100 µg/ml streptomycin at 37 °C, 5 % CO₂.
Cell Viability Assay
CCK‑8 was used to evaluate viability of PRMI8226 and 293T cells after treatment with PBS, PEG‑CdTe, free DOX, or PEG‑CdTe‑DOX at various concentrations and time points. Cell viability was expressed as (1 – ODₜ/OD_c) × 100 %.
Cellular Uptake
Flow cytometry quantified intracellular DOX fluorescence in PRMI8226 cells after 24 h incubation with the different formulations. Relative fluorescence intensity (FI) was calculated as FI_treated / FI_PEG‑CdTe.
Apoptosis Assay
PRMI8226 cells were stained with Annexin V‑FITC and PI, then analyzed by flow cytometry to determine early and late apoptosis.
Western Blotting
Protein extracts from treated PRMI8226 cells were separated by 10 % SDS‑PAGE, transferred to PVDF membranes, and probed for Bax, Bcl‑2, Caspase‑3, and β‑actin. Bands were visualized with ECL and quantified using Image J.
Statistical Analysis
Data are presented as mean ± SD. Statistical significance was assessed by Student’s t‑test, with *p* < 0.05 considered significant. Experiments were performed in triplicate.
Results
Characterization of PEG‑CdTe‑DOX
TEM images confirm a crystalline core and uniform dispersion of PEG‑CdTe and PEG‑CdTe‑DOX (Fig. 2a,b). DLS analysis shows hydrodynamic diameters of 8.20 nm for PEG‑CdTe and 78.31 nm for PEG‑CdTe‑DOX (Fig. 2c). EE and DL were 77.20 % ± 1.12 % and 42.12 % ± 0.98 %, respectively (Fig. 2e). Zeta potentials were –20.12 ± 2.45 mV (PEG‑CdTe) and –10.50 ± 1.26 mV (PEG‑CdTe‑DOX).
Drug release studies demonstrate a pH‑triggered profile: ~92.5 % of DOX is released at pH 5.0 and 88 % at pH 6.0 within 24 h, whereas release is markedly slower at physiological pH 7.4 and 8.0 (Fig. 2d). This behavior ensures preferential DOX release in the acidic tumor microenvironment.
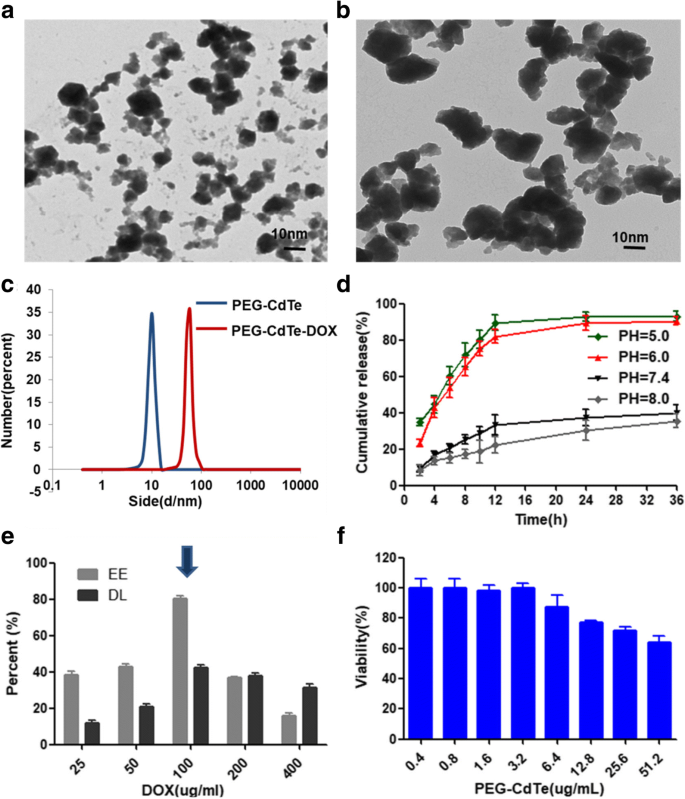
Characteristics of PEG‑CdTe and PEG‑CdTe‑DOX. Notes: a, b TEM images; c DLS; e EE and DL; d pH‑dependent release; f viability of 293T cells after PEG‑CdTe exposure.
Confocal imaging confirms robust intracellular delivery of DOX by PEG‑CdTe‑DOX, with markedly higher red fluorescence in PRMI8226 cells compared to free DOX (Fig. 3).
Cellular Uptake and Cytotoxicity of PEG‑CdTe‑DOX In Vitro
Flow cytometry reveals a significant increase in intracellular DOX fluorescence in PEG‑CdTe‑DOX–treated cells versus free DOX (*p* < 0.01; Fig. 4a). CCK‑8 assays demonstrate time‑dependent inhibition of PRMI8226 viability: 58 %, 70 %, and 85 % at 24, 48, and 72 h, respectively, surpassing the effect of free DOX (*p* < 0.05). Importantly, PEG‑CdTe alone does not affect cell viability, underscoring its biocompatibility. 293T cells remain unaffected at equivalent PEG‑CdTe concentrations, confirming selective cytotoxicity toward myeloma cells (Fig. 4b).
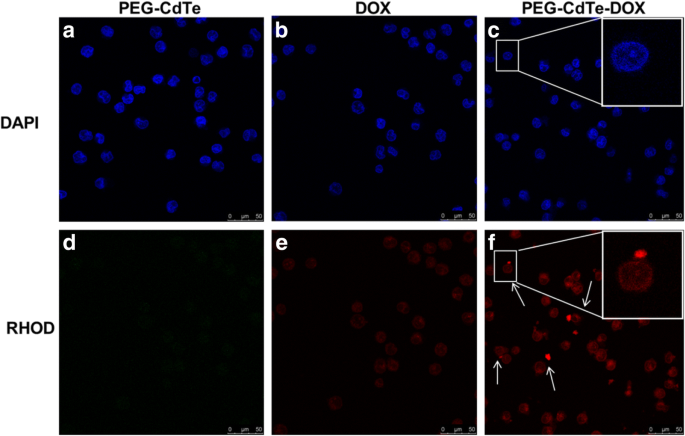
Fluorescence microscopy images of PRMI8226 cells after exposure to PEG‑CdTe, free DOX, or PEG‑CdTe‑DOX. Red fluorescence indicates DOX delivery (scale bar: 50 µm).
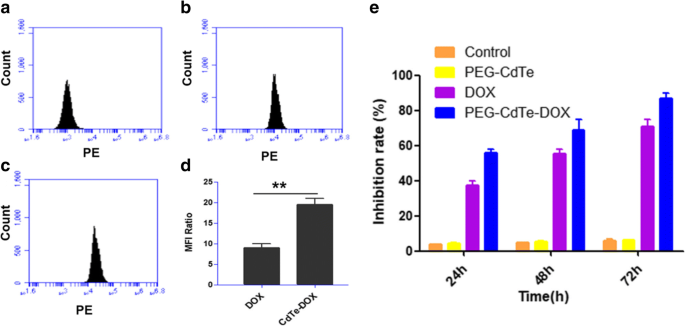
Mean fluorescence intensity (a) and CCK‑8 growth inhibition (b) of PRMI8226 cells treated with PBS, PEG‑CdTe, free DOX, and PEG‑CdTe‑DOX.
Apoptosis of PRMI8226 Cells
Annexin V/PI staining shows a pronounced increase in apoptotic cells in the PEG‑CdTe‑DOX group (66.5 %) versus free DOX (34.1 %) and controls (≤ 7 %) (Fig. 5). This significant difference (*p* < 0.01) indicates that PEG‑CdTe‑DOX enhances apoptosis through a delivery‑dependent mechanism.
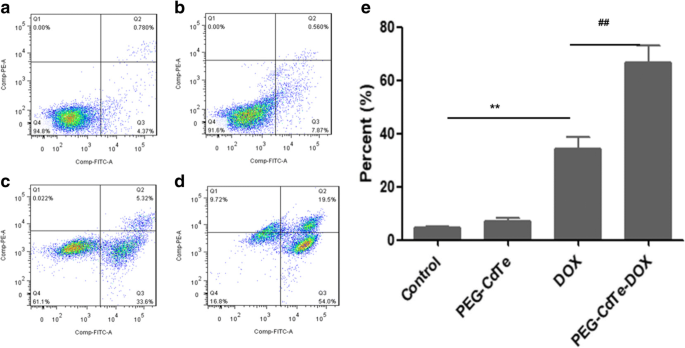
Apoptosis rates of PRMI8226 cells under different treatments.
Western Blotting
Western blot analysis reveals upregulation of pro‑apoptotic Bax and Caspase‑3, and downregulation of anti‑apoptotic Bcl‑2 in PEG‑CdTe‑DOX–treated cells relative to free DOX (Fig. 6). The pronounced shift in the Bax/Bcl‑2 ratio confirms activation of the mitochondrial apoptosis pathway.
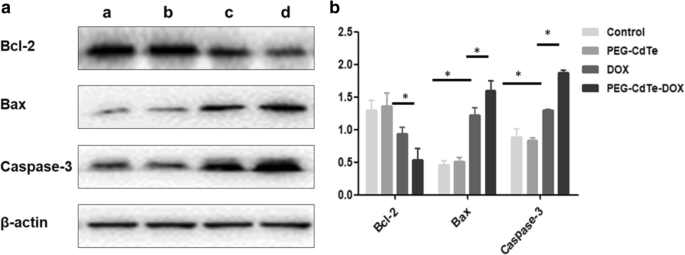
Expression of apoptosis‑related proteins in PRMI8226 cells.
Discussion
The poor tolerance of high‑dose chemotherapy is the principal barrier to effective treatment of EMM. Our PEG‑CdTe‑DOX nanocarrier circumvents this limitation by delivering DOX selectively to myeloma cells, achieving higher intracellular concentrations while sparing normal tissues. The high EE (≈77 %) and DL (≈42 %) enable efficient drug loading. PEGylation confers stealth properties, reducing opsonization and prolonging systemic circulation, while the nanoscale size (~78 nm) allows enhanced permeability and retention (EPR) in tumor tissue.
The acidic tumor microenvironment (pH 5–6) triggers rapid DOX release from PEG‑CdTe‑DOX, as demonstrated by the release kinetics. This localized release maximizes cytotoxicity where needed and limits systemic exposure, thereby mitigating cardiotoxicity and marrow suppression—common limitations of conventional DOX therapy. In vitro, PEG‑CdTe‑DOX produced superior growth inhibition and apoptosis in PRMI8226 cells, accompanied by a shift in the Bax/Bcl‑2 balance and activation of Caspase‑3, confirming mitochondrial pathway engagement.
These findings align with previous reports that PEGylated quantum dots enhance tumor targeting and reduce off‑target effects. Importantly, PEG‑CdTe alone exhibited negligible cytotoxicity, underscoring its suitability as a drug carrier. Future in vivo studies are warranted to confirm pharmacokinetics, biodistribution, and therapeutic benefit in animal models of EMM.
Conclusions
We have successfully engineered a PEG‑modified CdTe quantum dot system that efficiently loads and delivers doxorubicin to extramedullary multiple myeloma cells. The nanoconjugate achieves potent, pH‑triggered drug release, enhances apoptosis via the mitochondrial pathway, and preserves normal cell viability, offering a promising platform for safer, more effective EMM therapy.
Abbreviations
- DAPI
4′,6‑Diamidino‑2‑phenylindole
- DL
Drug loading
- DOX
Doxorubicin
- EE
Encapsulation efficiency
- EMM
Extramedullary multiple myeloma
- FCM
Flow cytometry
- HPLC
High‑performance liquid chromatography
- MM
Multiple myeloma
- PBS
Phosphate‑buffered saline
- PEG‑CdTe‑DOX
Polyethylene glycol‑modified cadmium telluride quantum dots loaded with doxorubicin
- SDS/PAGE
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- TEM
Transmission electron microscope
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Zebrafish: A Real‑Time Model for Nanotechnology‑Enabled Brain Drug Delivery
- 131I‑Labeled PLGA‑Lipid Nanoparticles for Targeted Paclitaxel Delivery and Imaging in Melanoma Therapy
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Chitosan‑Capped, Enzyme‑Responsive Hollow Mesoporous Silica Nanoplatforms for Targeted Colon Drug Delivery
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



