Zebrafish: A Real‑Time Model for Nanotechnology‑Enabled Brain Drug Delivery
Targeting drugs to the brain remains a formidable challenge, largely due to the blood–brain barrier (BBB). This barrier is a sophisticated network of capillaries that tightly regulates entry of substances into the central nervous system. Consequently, a deep understanding of BBB physiology is essential for therapeutic success. Recent evidence points to the zebrafish (Danio rerio) as a highly conserved and versatile model for studying BBB permeability, thanks to its genetic, anatomical, and functional similarities with mammals. The zebrafish offers advantages such as low maintenance cost, high fecundity, and transparent embryos that enable live imaging of drug distribution. Nanoparticle‑based platforms are emerging as powerful tools to ferry neurospecific agents across the BBB, yet their biocompatibility and toxicity must be rigorously evaluated. The zebrafish model is uniquely positioned to screen these nanocarriers in a real‑time, high‑throughput setting, accelerating the discovery of novel neurospecific therapies for brain disorders. Drug delivery involves the precise transport of therapeutic compounds to specific tissues or cell populations. Successful delivery hinges on controlled release, absorption, and distribution, with common routes including enteral, parenteral, inhalation, transdermal, topical, and oral. When the target organ is the brain, the BBB becomes the principal obstacle, allowing only small, lipid‑soluble molecules (≤400 Da) to passively diffuse into the CNS. Most neurologic drugs fail to cross this barrier, leaving central nervous system (CNS) diseases—such as Alzheimer’s, Parkinson’s, and ALS—under‑treated. Addressing this unmet need requires novel delivery technologies that can breach the BBB while minimizing systemic toxicity.
Nanotechnology has rapidly evolved as a promising strategy to surmount the BBB. Nanoparticles (NPs) can encapsulate hydrophobic drugs, facilitate receptor‑mediated transcytosis, and enable real‑time tracking of therapeutic distribution. However, the clinical translation of NP‑based CNS therapies demands comprehensive safety profiling. The zebrafish has emerged as a gold‑standard model for nanotoxicology, combining cost‑effectiveness, high fecundity, and optical transparency. Although its nervous system is less complex than mammals', zebrafish share ~70 % genomic similarity and 84 % homology in disease‑causing genes with humans, making it an attractive surrogate for human BBB studies.
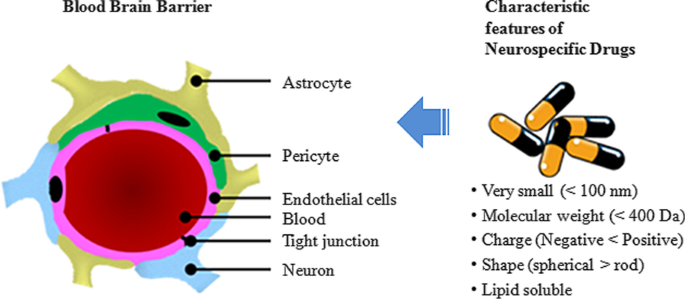
This review surveys recent advances in nanotechnology‑mediated brain drug delivery using zebrafish, summarizing BBB challenges, NP optimization strategies, toxicity assessment, and therapeutic potential for neurodegenerative diseases. We also discuss the strengths and limitations of the zebrafish model and its prospective role in translational research. The BBB is a highly selective interface composed of endothelial cells, pericytes, astrocytes, tight junctions, and a basal lamina. Its primary function is to preserve brain homeostasis by restricting paracellular diffusion of ions and hydrophilic molecules. Endothelial cells lack fenestrations and are sealed by tight junctions and adherens junctions, while gap junctions composed of connexins enable intercellular communication. Molecules traverse the BBB via paracellular diffusion or transcellular routes, including receptor‑mediated transport and transcytosis. Factors influencing permeability include molecular weight, charge, lipophilicity, and size. Current strategies exploit nanocarriers to bypass the BBB, leveraging their tunable size, surface charge, and functionalization to achieve selective uptake. These approaches aim to improve drug solubility, enable combinatorial therapy, target specific CNS sites, and facilitate real‑time monitoring of therapeutic distribution. Nanoparticles, defined as materials with at least one dimension between 1 and 100 nm, offer a suite of advantages for CNS delivery: improved solubility of hydrophobic drugs, co‑delivery of multiple agents, site‑specific targeting, traversal of tight barriers, enhanced imaging capabilities, and precise pharmacokinetic tracking. These attributes position nanotechnology as a transformative tool for neurospecific therapeutics. Characteristics of neurospecific drugs. The BBB is typically composed of tight junctions in the endothelial cells surrounded by astrocytes, pericytes, and neurons. Preferred properties for BBB penetration are: Abstract
Introduction
Blood–Brain Barrier: The Main Obstacle in Neurospecific Drug Delivery
Modern Technologies for BBB‑Targeted Drug Delivery
Why Nanoparticles Are a Current Choice for Neurospecific Drug Delivery
Zebrafish as a Model for Neurospecific Drug Delivery
The Danio rerio model has become a cornerstone for developmental biology and disease modeling due to its cost‑effective upkeep, high fecundity, and genetic conservation with mammals. Embryonic zebrafish develop externally and remain transparent, allowing real‑time visualization of organogenesis and drug distribution. This transparency, coupled with rapid development, facilitates high‑throughput screening of teratogenic, genotoxic, and cardiovascular effects of nanoparticles. For example, gold nanoparticles disrupt eye development and pigmentation, while silica NPs induce cardiovascular toxicity and mortality. Conversely, chitosan NPs exhibit superior biocompatibility relative to their non‑modified counterparts. Nanoparticle safety assessment in zebrafish is indispensable before clinical translation. Toxicity endpoints include mortality, teratogenicity, immune activation, and genomic damage. Zebrafish studies have revealed dose‑ and time‑dependent effects of metal oxide NPs, such as TiO₂, ZnO, and Cu, on embryonic development and adult physiology. Carbon‑based nanomaterials (fullerenes, CNTs, graphene QDs) generally display low toxicity, though surface charge and morphology modulate their biological interactions. The zebrafish’s unique advantages—transparent embryos, rapid organ development, and high genetic homology—make it an unparalleled system for elucidating NP behavior in vivo and guiding safe, effective neurospecific delivery strategies.
Insights from Nanoparticle Studies on Embryos
Nanoparticle exposure in embryos has uncovered critical developmental effects: TiO₂ NPs can induce early hatching or embryonic death depending on dose, alter swimming behavior, and disrupt thyroid signaling. Lead accumulation has been implicated in endocrine disruption, while BDNF, C‑fos, and C‑jun expression is up‑regulated, suggesting neurodevelopmental perturbations. Other studies report teratogenic malformations from silver, copper, and gold NPs, and delayed hatching from ZnO and SiO₂ NPs. Notably, chitosan NPs demonstrate high tolerance, highlighting the importance of surface chemistry in toxicity mitigation. Advanced imaging in zebrafish embryos—using fluorescent tags and confocal microscopy—enables real‑time tracking of NP distribution, providing insights into uptake mechanisms and organ-specific accumulation.
Insights from Nanoparticle Studies on Adults
Adult zebrafish studies complement embryonic data, revealing behavioral changes, cardiotoxicity, and immune activation following NP exposure. For instance, gold NP exposure alters locomotion, while silver BSA NPs accumulate in skin and elicit inflammatory responses. Cardiovascular endpoints such as heart rate, rhythm, and QT interval have been extensively modeled in zebrafish, offering translational relevance to human drug safety profiling. Hepatic metabolism studies in zebrafish also mirror mammalian CYP450 activity, underscoring the model’s utility in pharmacokinetic assessment.
Zebrafish Offers a Complete Pathological Study Model for Neurospecific Drug Delivery
Using transparent embryos, researchers can readily observe teratogenic effects (eye pigmentation, mortality, cardiovascular alterations), immunotoxicity (neutrophil and macrophage recruitment), and genotoxicity (DNA damage assays). Carbon‑based NPs, such as fullerenes and CNTs, show minimal developmental toxicity at low doses, while larger or positively charged variants exhibit higher cytotoxicity. These observations underscore the necessity of precise NP design to balance therapeutic efficacy and safety.
Nanoparticles Focused for Delivering Drug to the Brain
Recent work demonstrates the feasibility of NP‑mediated brain delivery in zebrafish. Polymer NPs conjugated with phenylboronic acid enable fluorescent detection of dopamine in larvae, paving the way for theranostics of dopaminergic disorders. Functionalized poly‑N‑isopropyl acrylamide (PNIPAM) nanogels have successfully delivered donepezil across the BBB with sustained release, illustrating the potential of smart hydrogel carriers. Additionally, gold NPs coated with polysorbate 80 and PEG have been validated to penetrate the BBB and deliver neurospecific therapeutics in vivo. These studies collectively highlight the versatility of diverse NP platforms for targeted CNS therapy.
Translational Approach of Neurospecific Nanoparticles: Zebrafish to Humans
While non‑human primates and rodents remain the gold standard for pre‑clinical research, zebrafish increasingly bridge the gap between in vitro assays and mammalian studies. Their genetic tractability, rapid life cycle, and optical clarity make them ideal for evaluating novel nanodiamonds (NDs) and other emerging nanocarriers. For instance, fluorescent NDs (FNDs) exhibit photostability and negligible toxicity in zebrafish yolk cells, and real‑time confocal imaging confirms their efficient cellular uptake. These findings support the use of zebrafish for screening ND‑based brain delivery systems. Future directions involve combining multiple drugs within a single NP, exploring fullerene‑based and nanodiamond carriers for neurodegenerative disease models, and rigorously assessing brain‑penetrating capabilities of next‑generation NPs. Zebrafish provide a scalable, ethically sound platform to accelerate these investigations and inform human clinical trials.
Future Research Directions
Despite rapid advances, challenges such as NP toxicity, immunogenicity, and efficient delivery persist. Comprehensive nano‑bio interaction studies must precede in vivo deployment, and advanced imaging modalities should map biodistribution and pharmacokinetics. Zebrafish, with its transparent embryos and high throughput capacity, offers an unrivaled system to address these gaps, guiding the transition from bench to bedside for safe, effective neurospecific therapies.
Conclusion
The BBB remains the principal barrier to CNS drug delivery. Nanotechnology presents a promising avenue to overcome this hurdle, enabling targeted, efficient transport of therapeutics across the BBB. Liposomes, dendrimers, micelles, carbon nanotubes, and nanogels represent the leading NP platforms, each with distinct advantages for brain targeting. Zebrafish serve as a powerful model for nanotoxicology and neurospecific delivery studies, and further research using this system will drive breakthroughs in brain‑directed therapeutics.
Availability of data and materials
Not applicable.
Abbreviations
- BBB:
Blood–brain barrier
- NPs:
Nanoparticles
- Au:
Gold
- Ag:
Silver
- Cu:
Copper
- Cd:
Cadmium
- CuO:
Copper oxide
- MgO:
Magnesium oxide
- NiO:
Nickel oxide
- ZnO:
Zinc oxide
- MPs:
Microplastics
- MOFs:
Metal organic frameworks
- CNTs:
Carbon nanotubes
- TiO2:
Titanium dioxide
- QDs:
Quantum dots
- PCR:
Polymerase chain reaction
- GQDs:
Graphene quantum dots
- PNIPAM:
Poly N-isopropyl acrylamide
- NDs:
Nanodiamonds
- FND:
Fluorescent nanodiamonds
- AD:
Alzheimer’s disease
- PD:
Parkinson’s disease
- HD:
Huntington’s disease
- ALS:
Amyotrophic lateral sclerosis
- MND:
Motor neuron diseases
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- PEG‑Modified Cadmium Telluride Quantum Dots Deliver Doxorubicin for Targeted Treatment of Extramedullary Multiple Myeloma
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Intelligent Nanoantenna‑Based Drug Delivery System for Targeted Cancer Therapy
- Chitosan‑Capped, Enzyme‑Responsive Hollow Mesoporous Silica Nanoplatforms for Targeted Colon Drug Delivery
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



