Reimagining Nanoparticle Therapies Beyond Cancer
Abstract
Nanoparticle‑based drug delivery has dominated oncology research for over a decade, largely driven by the enhanced permeability and retention (EPR) effect. Yet a comprehensive systematic review by Wilhelm et al. revealed that only a median of 0.7 % of injected nanoparticles reach tumors, underscoring the mononuclear phagocytic system (MPS) as a formidable barrier. This article argues that nanotechnologists should broaden their focus beyond cancer, targeting diseases that either do not rely on EPR or benefit from MPS‑mediated organ accumulation. Systemic applications—such as sepsis, organ failure, metabolic and autoimmune disorders—can leverage intravenous delivery, while localized strategies for lungs, rectum, and bladder can achieve high tissue concentrations with reduced clearance. Ex vivo perfusion offers a novel platform for organ repair and isolated‑organ chemotherapy, presenting a promising translational pathway for nanoparticle therapeutics.
Background
The past decade has seen a surge in nanoparticle drug‑delivery studies that outpace gene therapy and stem‑cell research. Like these fields, the field’s primary ambition has been a cancer cure. The EPR effect was long regarded as the cornerstone of nanoparticle therapy. However, Wilhelm et al.’s meta‑analysis demonstrates that a mere 0.7 % of administered dose actually reaches tumors—a figure too low to translate into meaningful therapeutic benefit in humans. Consequently, only a handful of nanoparticle formulations—such as Abraxane and Doxil—have received FDA approval, mainly offering a safer toxicity profile rather than superior efficacy or diagnostics. The authors propose a 30‑year research roadmap that calls for deeper mechanistic insight into nanoparticle–cell interactions. This article builds on that call, suggesting that by redefining therapeutic targets and exploiting MPS‑mediated delivery, we can unlock the full potential of nanomedicine.
Beyond fundamental studies, we advocate for innovative, clinically relevant applications: systemic treatments for sepsis, organ failure, metabolic and autoimmune disorders, and targeted local delivery to organs such as the lung, rectum, or bladder—both in vivo and ex vivo. Figure 1 illustrates these alternative therapeutic avenues.
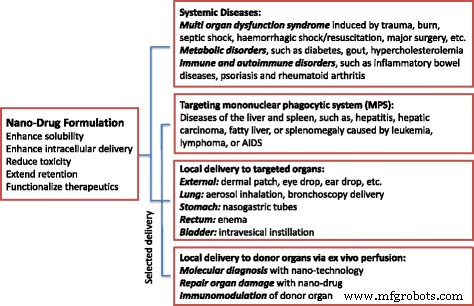
New avenues for nanoparticle‑related therapies. Potential application of nanodrug formulations for clinical situations that rely less on passive enhanced permeability and retention (EPR) effects.
Converting the Limitations of Nanoparticle Delivery to Therapeutic Advantages
The core challenge in translating nanotherapies is the sequestration of nanoparticles by the mononuclear phagocytic system (MPS), which comprises liver, spleen, and lung macrophages. These cells efficiently capture particles based on size, shape, and surface chemistry, leading to rapid clearance. Various strategies—steric stabilization, “self” peptide conjugation, and cell‑membrane cloaking—have been explored, yet the precise biophysical interactions remain poorly understood.
Reframing the therapeutic goal toward diseases affecting MPS organs—such as hepatic inflammation, fibrosis, fatty liver, and splenic disorders—can turn MPS‑mediated accumulation into an advantage. Targeted nanomedicine for liver inflammation, hepatitis, and even rare lysosomal storage disorders could harness this organ‑specific uptake, offering new avenues for orphan drug development.
Changing the Targets: Application of Nanoparticle Therapy for Systemic Diseases
A systematic review of nanoparticle clinical trials confirms that most research remains cancer‑centric, despite the MPS’s strong clearance of systemically administered particles. For systemic diseases—sepsis, multi‑organ failure, metabolic disorders—nanoparticles can enhance drug solubility, reduce systemic toxicity, and provide targeted delivery. For instance, gold nanoparticles have been engineered to carry peptide inhibitors of Toll‑like receptors, mitigating hyperinflammatory responses in sepsis models.
Other systemic applications include:
- nanoparticle‑based delivery of antidiabetic agents
- functionalized particles for gout treatment
- lipid‑nanoparticle encapsulation of statins for hypercholesterolemia
- nanocarriers that inhibit drug efflux in leukemia, restoring chemosensitivity
- pro‑coagulant or anticoagulant nanoparticles for clotting disorders
- nanoparticle‑mediated immunotherapy for autoimmune and allergic diseases, including encapsulated allergens for asthma and DNA vaccines for vaccine development
Because many systemic illnesses involve multiple organs and cell types, broad‑spectrum therapeutics delivered via nanoparticles can simultaneously modulate several pathological pathways, improving efficacy while minimizing off‑target effects.
Local Delivery of Nanoparticle Therapies to Targeted Organs In Vivo
Targeted drug delivery—whether via eye drops, inhalers, or transdermal patches—is a well‑established strategy. Nanotechnology can further refine these approaches by enhancing penetration, retention, and controlled release. For example, a hydrophobic Src inhibitor (PP2) was successfully encapsulated in self‑assembling peptide nanoparticles, improving intratracheal delivery and mitigating acute lung injury.
Other organs amenable to local delivery include the stomach (via nasogastric or gastrostomy tubes), rectum (via enemas), bladder (via intravesical instillation), and joints (via intra‑articular injection). These routes bypass first‑pass metabolism and enable high local drug concentrations with minimal systemic exposure. Nanoparticles can also ferry biomaterials across the round‑window membrane for inner‑ear therapies and enhance delivery to inflamed joints for rheumatoid arthritis.
Local Delivery of Nanoparticle Therapies to Targeted Organs Ex Vivo
Ex vivo lung perfusion (EVLP) has revolutionized donor lung assessment and repair. By gradually warming, ventilating, and perfusing lungs outside the body, EVLP enables the safe delivery of therapeutic agents—drugs, anti‑inflammatory genes, antibiotics, and stem cells—directly to the lung tissue. Nanoparticles, free from systemic clearance by liver and spleen, can be administered at lower doses, reducing toxicity and allowing real‑time evaluation of efficacy. Similar ex vivo perfusion platforms are emerging for kidneys, hearts, and livers, offering a controlled environment to study nanomaterial pharmacodynamics and to validate therapies before clinical use.
For example, siRNA nanoparticles delivered during ex vivo perfusion successfully knocked down MHC‑II expression in human arterial allografts, demonstrating the translational potential of this approach. Interdisciplinary collaboration between nanoscientists and transplant surgeons is essential to advance these technologies.
Local Delivery of Nanoparticle Therapies to Targeted Organs In Vivo—Back to Cancers
Building on EVLP, an in‑vivo lung perfusion system has been developed to administer high‑dose chemotherapy exclusively to the lung after resection of primary tumors. This strategy targets micrometastases while sparing systemic organs. Nanoparticle‑based chemotherapeutics can further reduce pulmonary toxicity and avoid MPS sequestration.
Isolated limb infusion for melanoma and isolated hepatic perfusion for liver metastases are established clinical procedures that isolate drug delivery, effectively bypassing the MPS. These platforms provide immediate translational opportunities for nanoparticle formulations, offering lower systemic exposure and higher local concentrations. While active targeting remains critical for disseminated micrometastases, these local perfusion techniques open a new frontier for nanoparticle‑mediated cancer therapy.
In conclusion, the foundational insights of Wilhelm et al. underscore the need for deeper mechanistic studies. Simultaneously, emerging delivery technologies—systemic, local, ex vivo, and perfusion‑based—offer pragmatic routes to translate nanoparticle therapeutics into clinical practice.
Abbreviations
- EPR:
Enhanced permeability and retention
- EVLP:
Ex vivo lung perfusion
- MPS:
Mononuclear phagocytic system
Nanomaterials
- MIT Study Introduces Comprehensive Life‑Cycle Assessment for Paving and Building Materials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Introducing Ultem 1010 & ASA: Advanced FDM Materials for Stratasys Fortus Printers
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Innovative Nucleoside–Lipid Nanocarriers Enhance Sorafenib Delivery and Reduce Toxicity
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Redox‑Responsive Gelatin/Silica Nanogels Functionalized with AS1411 Aptamer for Targeted siRNA Delivery
- Revolutionizing Last‑Mile Delivery in 2024: Robotics, Collaboration, and Superior Customer Experience
- New Study Offers Design Guidelines for Thermoplastic Composite Overmolding
- Warwick Manufacturing Group Breaks 5G Speed Record for Autonomous Vehicles



