Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
Abstract
Nanoparticle carriers can modulate drug hydrophilicity, influencing cellular uptake and reducing off‑target effects. Metal–organic frameworks (MOFs), crystalline porous nanomaterials assembled from metal ions and organic linkers, offer biodegradability, tunable pore size, and versatile surface chemistry. Their rigid structure preserves encapsulated therapeutics under physiological stress, while ligand modification allows selective binding to overexpressed tumor biomarkers. MOFs engineered to respond to tumor‑specific cues—such as elevated H₂O₂, hypoxia, and glutathione—ensure minimal leakage in circulation and controlled release at malignant sites. This review surveys stimulus‑responsive MOFs for tumor therapy, highlighting design strategies and therapeutic outcomes.
Introduction
Tumors remain a leading cause of morbidity and mortality worldwide, with conventional chemotherapy and surgery often limited by severe side effects and recurrence [1,2,3]. In recent decades, nanocarriers have emerged as powerful platforms for tumor imaging, theranostics, and targeted therapy [4]. Among these, MOFs have attracted particular attention due to their high crystallinity, large surface area, and the ability to respond to diverse stimuli [5,6]. MOFs consist of metal ions or clusters linked by organic bridging ligands, enabling precise control over pore architecture, chemistry, and morphology [7,8,9]. Their organic active sites and open frameworks provide routes for functionalization via encapsulation, grafting, or infiltration, enhancing biocompatibility, solubility, and target interaction [10,11].
Encapsulation via coprecipitation or biomimetic mineralization offers a rapid, one‑step strategy to embed drugs directly into the MOF lattice, yielding high payloads and circumventing crystal‑growth challenges associated with pre‑functionalized ligands [12–16]. Nevertheless, challenges such as complex synthesis, rapid clearance by the immune system, and short blood half‑life limit clinical translation [17–19]. Here we summarize recent advances in stimulus‑responsive MOFs that address these limitations and enhance tumor theranostics.
pH/ATP Responsive
Zeolitic imidazolate frameworks (ZIFs) are a subclass of MOFs with tunable pore size, ultra‑large surface area, and facile synthesis, making them ideal for tumor theranostics [20]. ZIF‑8 nanoparticles can escape endosomes via a proton‑sponge effect driven by imidazole protonation in acidic endosomes, facilitating cytosolic delivery [21].
Gene therapy faces challenges such as nuclease degradation and limited cellular uptake of naked nucleic acids [22–24]. ZIF‑8, synthesized from low‑toxicity Zn²⁺ and 2‑methylimidazole under mild conditions, offers excellent encapsulation and protection of plasmid DNA (pDNA) [25]. Li et al. demonstrated a one‑step biomimetic mineralization method to load large pDNA into ZIF‑8 and ZIF‑8‑polymer hybrids, achieving efficient intracellular delivery and gene expression [26]. Functionalization with cationic polymers like PEI further enhances electrostatic interaction, cellular uptake, and endo‑lysosomal escape [27].
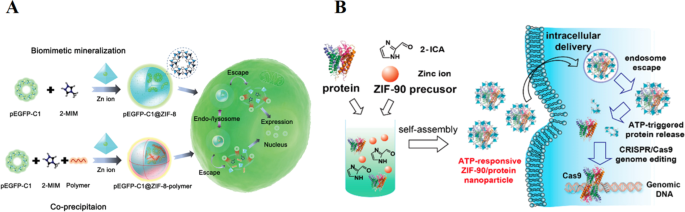
Copyright 2019 American Chemical Society. B Schematic illustration of the self‑assembly of ZIF‑90/protein nanoparticle and ATP‑triggered protein release from ZIF‑90 nanoparticle inside cells [29]. Copyright 2019 American Chemical Society
A Schematic representation for synthesis of pEGFP‑C1@ZIF‑8 nanostructures and pEGFP‑C1@ZIF‑8‑polymer nanostructures via biomimetic mineralization and coprecipitation, and their cellular delivery and expression process [26].
Extracellular ATP concentrations (<0.4 mM) contrast sharply with intracellular levels (1–10 mM) [28], providing a tumor‑specific trigger. Yang et al. reported an ATP‑responsive ZIF‑90 nanocarrier that disassembles in the presence of ATP, releasing encapsulated proteins such as superoxide dismutase or bovine serum albumin while preserving functionality [29]. This platform demonstrates that MOFs can deliver a wide range of biomacromolecules, opening avenues for protein delivery and genome editing in oncology.
Light Responsive
Photothermal therapy (PTT) exploits localized heating to induce irreversible tumor cell damage at temperatures above 44 °C, while sparing normal tissues that dissipate heat via blood flow [30–33]. Photodynamic therapy (PDT) employs near‑infrared (NIR) light, oxygen, and photosensitizers (PSs) to generate reactive oxygen species (ROS) that selectively kill cancer cells [34–38].
Park et al. engineered a Zr(IV)-based porphyrinic MOF (PCN‑224) capable of ROS generation under NIR irradiation, accumulating in tumors through the enhanced permeability and retention (EPR) effect [39]. However, targeting efficacy was suboptimal, prompting folic acid modification to improve tumor homing and PDT outcomes [40].
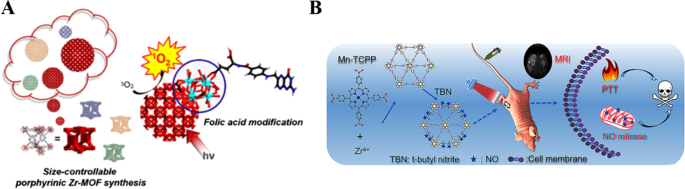
Copyright 2018 American Chemical Society. B Scheme for the synthesis of the NMOF–SNO nanocomposite and the NIR light‑triggered NO release and PTT [42]. Copyright 2018 American Chemical Society
A Illustration of PCN‑224 structure and synthesis of spherical nanoparticles via cubic unit assembly [39].
Building on this, Zhang et al. introduced Mn‑porphyrin MOFs that combine MRI contrast with photothermal conversion, further conjugated with an NO donor (S‑nitrosothiol) to deliver synergistic photothermal and NO‑based therapy [42,43]. The MOF platform achieves efficient tumor accumulation, high photothermal conversion, and controlled NO release, providing a dual‑modality theranostic system that guides treatment timing and improves therapeutic index.
Incorporating fluorescent porphyrins into MOFs yields materials that support both imaging and PDT, paving the way for next‑generation tumor theranostics.
H₂O₂ Responsive
Tumor microenvironments (TMEs) are characterized by elevated H₂O₂, hypoxia, acidic pH, and high glutathione (GSH) levels [44–46]. MnO₂ nanosheets can decompose H₂O₂ into Mn²⁺ and O₂, enhancing oxygen availability and ROS generation under laser irradiation, thereby boosting PDT efficacy [49–52].
Sun et al. constructed a BSA‑MnO₂/Ce6@ZIF‑8 nanoplatform that releases O₂ in response to H₂O₂ and acidic conditions, enabling MRI‑guided, H₂O₂‑responsive PDT with minimal side effects [53]. Loading chlorin e6 (Ce6) into ZIF‑8 improves aqueous solubility and ROS production under 650 nm laser irradiation, while BSA‑decorated MnO₂ enhances dispersibility, biocompatibility, and oxygen generation.
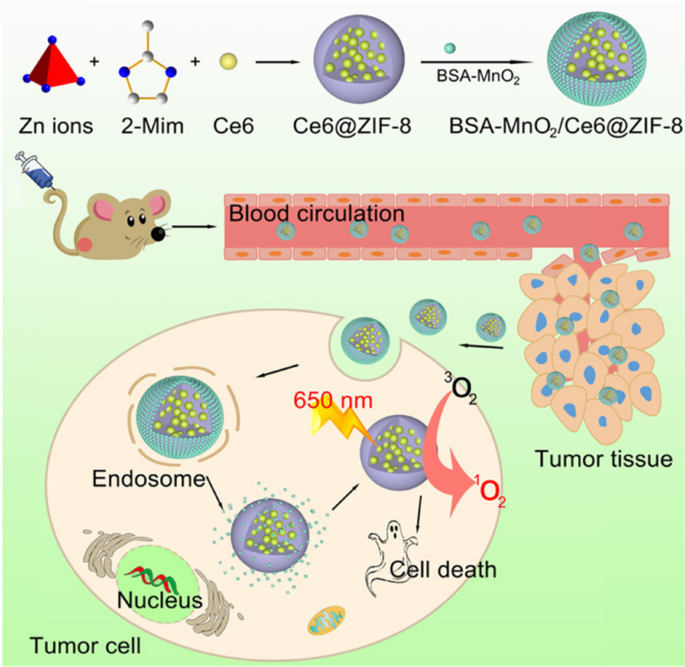
Copyright 2019 American Chemical Society
Schematic illustration for the formation of a BSA‑MnO₂/Ce6@ZIF‑8 nanoplatform and its TME‑responsiveness for ROS generation upon 650 nm NIR laser irradiation, enabling MRI‑guided photodynamic therapy [53].
GSH Responsive
High intracellular GSH levels (2–10 mM) in cancer cells neutralize ROS, undermining PDT efficacy [54,55]. Wan et al. developed a GSH‑unlocked Mn(III)‑sealed MOF that disintegrates in the presence of excess GSH, releasing Mn²⁺ and triggering ROS generation for comprehensive tumor inhibition [58].
To overcome rapid clearance and enhance tumor accumulation, biomimetic cloaking with cancer cell membranes has emerged. Min et al. engineered 4T1‑cell‑membrane‑camouflaged MnO₂‑coated Zr‑MOF loaded with VEGFR‑2 inhibitors (aMMTm), achieving GSH‑responsive ROS production, photothermal conversion, and anti‑angiogenesis while evading macrophage uptake [55,61,62]. The MnO₂ shell neutralizes tumor GSH and H₂O₂, modulating the TME and improving PDT outcomes; released Mn²⁺ serves as an MRI contrast agent, and apatinib prevents revascularization.
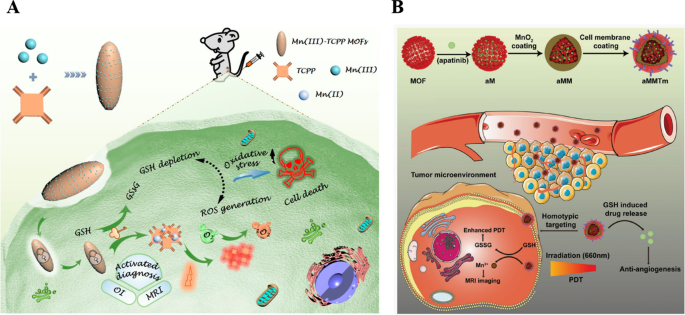
Copyright 2019 American Chemical Society. B Schematic illustration of aMMTm preparation and proposed combination therapy of PDT and anti‑angiogenesis [55]. Copyright 2019 WILEY–VCH Verlag GmbH & Co. KGaA, Weinheim
Schematic illustration of an endocytosis Mn(III)-sealed MOF nanosystem for MRI and optical imaging‑guided PDT via controlled ROS generation and GSH depletion after unlocking by overexpressed GSH in tumor cells [58].
The biomimetic platform offers a streamlined theranostic system that integrates dual imaging with synergistic therapy, setting a new standard for precision oncology.
Hydrogen Sulfide (H₂S) Responsive
Endogenous H₂S, produced by cystathionine β‑synthase, serves as a gasotransmitter and a tumor biomarker. Cu‑based MOFs exhibit strong Cu²⁺–S²⁻ interactions and catalytic activity under acidic conditions, enabling H₂S detection and therapy [65–67].
Li et al. engineered an H₂S‑activated Cu‑MOF that remains “OFF” in circulation but turns “ON” upon encountering elevated H₂S in colon tumors, generating photoactive copper sulfide with superior NIR absorption for photothermal therapy [68]. The Cu‑MOF also mimics peroxidase activity, reacting with H₂O₂ to produce hydroxyl radicals for chemodynamic therapy, achieving potent antitumor effects while sparing healthy tissue.
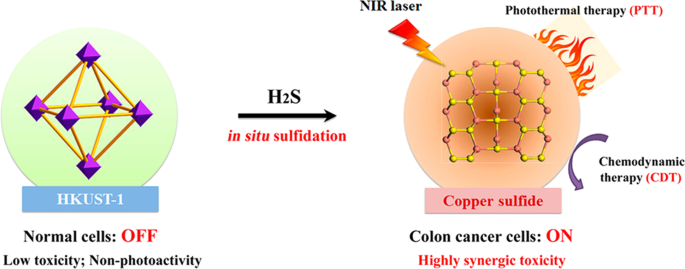
Copyright 2020 American Chemical Society
Schematic Illustration of the H₂S‑Triggered Transformation of Non‑photoactive Cu‑MOFs into a Near‑NIR‑Activatable Photothermal Agent by in‑situ sulfideification and Its Synergic Photothermal and Chemodynamic Therapy for Colon Cancer [68].
Perspectives
MOFs offer unmatched versatility for tumor drug delivery, but hurdles such as complex synthesis, immune clearance, systemic toxicity, suboptimal pharmacokinetics, off‑target accumulation, and untimely drug release must be overcome before clinical adoption.
Biomimetic cloaking with plasma membranes—derived from various cell types—provides a compelling strategy to shield MOFs from the immune system, prolong circulation, and leverage homotypic targeting via membrane proteins. This approach enhances the enhanced permeability and retention (EPR) effect, enabling selective tumor accumulation.
Furthermore, the acidic, hypoxic, and GSH‑rich TME drives the development of intelligent, stimulus‑responsive MOFs that precisely modulate drug release, ROS generation, and imaging. Such designs promise to elevate diagnostic accuracy, reduce collateral damage, and improve therapeutic outcomes in oncology.
Conclusion
We have surveyed diverse MOF architectures engineered for tumor therapy, emphasizing their stimulus‑responsive behavior and therapeutic efficacy. While high costs, intricate synthesis, and limited clinical data pose challenges, targeted delivery, low toxicity, and superior therapeutic performance remain the hallmarks for successful translation of MOF‑based systems into clinical practice.
Availability of data and materials
Not applicable.
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Zebrafish: A Real‑Time Model for Nanotechnology‑Enabled Brain Drug Delivery
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- High‑Loading 5‑Aminolevulinic Acid–Squalene Nanoparticles: Enhanced Tumor Photodetection and Therapy in Cell Models
- Optimized Preparation and Physicochemical Characterization of Dual‑Drug Nanoliposomes Encapsulating Erlotinib and Doxorubicin
- Hydrophilic HBP‑Modified rGO Enables Dual pH/NIR‑Triggered DOX Release for Synergistic Chemo‑Photothermal Tumor Therapy
- PEG‑Modified Cadmium Telluride Quantum Dots Deliver Doxorubicin for Targeted Treatment of Extramedullary Multiple Myeloma
- Mitochondria‑Targeted TiS2 Nanosheets Deliver Resveratrol for Photothermal‑Triggered Tumor Chemotherapy
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



