High‑Loading 5‑Aminolevulinic Acid–Squalene Nanoparticles: Enhanced Tumor Photodetection and Therapy in Cell Models
Abstract
Protoporphyrin IX (PpIX), the endogenous photosensitizer produced after 5‑aminolevulinic acid (5‑ALA) administration, is a cornerstone of photodiagnosis (PD) and photodynamic therapy (PDT) for several malignancies. Yet, 5‑ALA’s zwitterionic nature limits its penetration across biological barriers, curbing its systemic clinical utility. Here we describe a novel squalenoylated 5‑ALA (5‑ALA‑SQ) that self‑assembles into nanoassemblies (NAs) with a mean diameter of 70 nm, a low polydispersity index (0.12), a +36 mV zeta potential, and a remarkable drug loading of 26 %. In human prostate (PC3) and glioblastoma (U87MG) cell lines, 5‑ALA‑SQ NAs generated PpIX fluorescence that matched or exceeded that of the commercial hexyl ester (5‑ALA‑Hex) at comparable concentrations, while exhibiting superior performance at early time points (≤4 h). These results highlight 5‑ALA‑SQ NAs as a promising platform for systemic delivery of 5‑ALA in PD and PDT applications.
Background
Nanotechnology offers biocompatible, biodegradable carriers that can enhance the chemical stability, pharmacokinetics, and site‑specific delivery of therapeutic agents. However, many nanoparticle (NP) systems suffer from low drug loading (<5 %) and a burst‑release effect that compromises efficacy and safety. Squalene, a natural triterpene and cholesterol precursor, has emerged as a versatile scaffold for “squalenoylation” – covalent conjugation of drugs to squalene that drives spontaneous self‑assembly into high‑loading, stable nanoassemblies (NAs). Squalenoylated NAs have been successfully employed for chemotherapeutics such as gemcitabine, paclitaxel, cisplatin, and doxorubicin, often yielding superior pharmacological activity and, in some cases, enabling theranostic functionalities.
5‑ALA, the precursor of PpIX, is already used clinically for PD, PDT, and fluorescence‑guided resection of gliomas. Its therapeutic window is constrained by its charged structure, which limits systemic absorption and tumor accumulation. While esterified derivatives like 5‑ALA‑Hex have improved bioavailability, their use remains largely topical or limited to specific cancers such as bladder carcinoma. Recent studies have explored encapsulation of 5‑ALA in polymeric NAs, liposomes, dendrimers, and gold nanoparticles, yet none have translated into clinically viable systemic formulations.
Our goal was to engineer a 5‑ALA‑squalene conjugate that self‑assembles into NAs with high drug loading, robust stability, and efficient tumor uptake, thereby expanding the clinical reach of 5‑ALA for PD and PDT in solid tumors beyond the current indications.
Results and Discussion
Synthesis of the 5‑ALA‑SQ Building Block
The conjugate was assembled in four convergent steps. Boc‑protected 5‑ALA was coupled to a hydroxylated squalene derivative via EDC/DMAP‑mediated esterification, followed by mild acid‑mediated deprotection and final purification by reverse‑phase HPLC. The overall yield was high and the product was fully characterized by NMR and mass spectrometry.
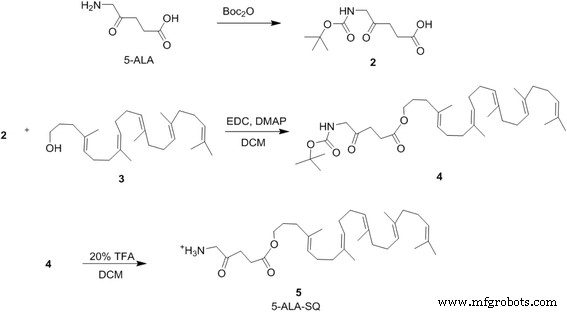
Figure 2. Synthesis of the 5‑ALA‑SQ building block in four steps.
Characterization of 5‑ALA‑SQ Nanoassemblies
Self‑assembly was induced by nanoprecipitation: the conjugate was dissolved in a 1:1 acetone/ethanol mix and injected dropwise into Milli‑Q water under stirring, followed by solvent removal. Dynamic light scattering (DLS) and cryo‑TEM revealed monodisperse particles with an average hydrodynamic diameter of 70 nm and a polydispersity index of 0.12. The zeta potential of +36 mV suggests strong colloidal stability and favorable interactions with negatively charged cell membranes. Importantly, the 26 % drug loading represents a substantial improvement over most 5‑ALA nanoparticle formulations, which typically exhibit <5 % loading.
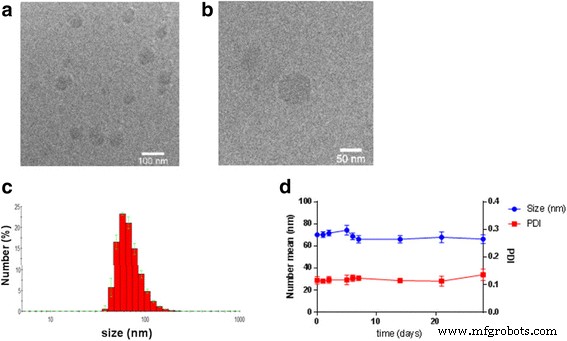
Figure 3. (a) Cryo‑TEM images and (b) DLS analysis of 5‑ALA‑SQ NAs. (c) Stability of particle size at 4 °C over several weeks.
In Vitro PpIX Fluorescence Kinetics in Cancer Cells
PC3 and U87MG cells were incubated with varying concentrations of 5‑ALA‑SQ NAs or the commercial 5‑ALA‑Hex. Fluorescence was monitored over 24 h using a plate reader (405 nm excitation, 630 nm emission). At 1.0–2.0 mM, 5‑ALA‑SQ NAs produced a steady increase in PpIX fluorescence, plateauing after ~8 h. In contrast, 5‑ALA‑Hex peaked at 0.10–0.30 mM but exhibited cytotoxicity above 1 mM, reducing overall fluorescence.
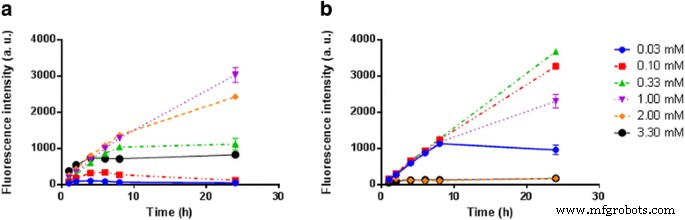
Figure 4. Kinetic PpIX fluorescence in PC3 cells following exposure to 5‑ALA‑SQ NAs (blue) or 5‑ALA‑Hex (red).
Dose–response curves at 4 h and 24 h revealed that 5‑ALA‑SQ NAs matched or surpassed 5‑ALA‑Hex in fluorescence intensity across both cell lines. The maximal fluorescence at 4 h was achieved with 1–2 mM of 5‑ALA‑SQ, while 24 h exposure required higher concentrations (up to 3.3 mM) to approach the same levels. Notably, 5‑ALA‑SQ maintained a more sustained fluorescence profile, with a gradual decline only at the highest concentrations and longest incubation times.
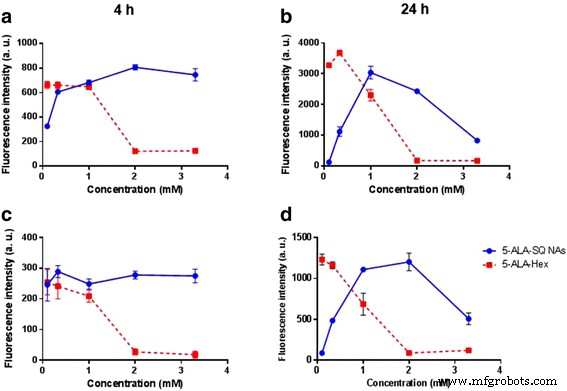
Figure 5. Dose–response curves of PpIX accumulation with 5‑ALA‑SQ NAs (blue) and 5‑ALA‑Hex (red) in PC3 (a,b) and U87MG (c,d) cells after 4 h (left) and 24 h (right).
When compared to the marketed 5‑ALA formulation, 5‑ALA‑SQ NAs produced significantly higher fluorescence after 4 h in U87MG cells, a critical window for intraoperative imaging. At 24 h, the fluorescence of 5‑ALA‑SQ aligned with that of 5‑ALA at optimal concentrations, indicating comparable overall PpIX production over time.
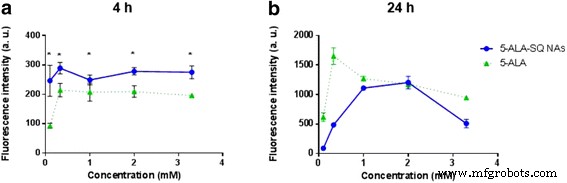
Figure 6. Dose–response curves of PpIX accumulation in U87MG cells after 4 h (a) and 24 h (b) with 5‑ALA‑SQ NAs (blue) and 5‑ALA (green).
The enhanced cellular uptake of 5‑ALA‑SQ is likely driven by albumin‑mediated transport of the monomeric conjugate, as previously demonstrated for squalene‑gemcitabine NAs. The positively charged amino group of 5‑ALA may orient toward the aqueous phase, while the hydrophobic squalene core assembles into the particle interior, accounting for the reduced particle size relative to other squalene NAs.
Conclusions
We have engineered a 5‑ALA‑squalene conjugate that spontaneously forms highly stable, high‑loading (26 %) nanoassemblies with favorable size and surface charge. In vitro studies in prostate and glioblastoma cells demonstrate that these NAs efficiently deliver 5‑ALA, generate PpIX fluorescence that rivals or exceeds commercial formulations, and maintain robust performance over extended incubation. These findings support further in vivo evaluation of 5‑ALA‑SQ NAs as a systemic platform for tumor photodiagnosis and photodynamic therapy.
Methods/Experimental
All reagents were obtained from commercial suppliers and used without further purification. 5‑ALA was Boc‑protected, coupled to a hydroxylated squalene derivative (3) using EDC/DMAP, then deprotected under mild acidic conditions. The final product (5) was purified by reverse‑phase HPLC and characterized by NMR and HRMS.
Nanoassemblies were prepared by nanoprecipitation: 1.2 mg of building block (5) was dissolved in a 50/50 acetone/ethanol mix (500 µL) and added dropwise to 1.25 mL Milli‑Q water at 100 µL/min. After 5 min stirring, solvents were removed by rotary evaporation at 30 °C, yielding a 2.00 mM NA solution.
Particle size, polydispersity, and zeta potential were measured by DLS (Malvern Nano ZS) at 25 °C. Cryo‑TEM was performed on vitrified samples prepared with a Virtobot plunger. Stability was assessed by DLS over 1 month at 4 °C.
PC3 (12,000 cells/well) and U87MG (10,000 cells/well) were seeded in 96‑well plates, incubated with serial dilutions of 5‑ALA‑SQ NAs, 5‑ALA‑Hex, or 5‑ALA (serum‑free media) for up to 24 h. PpIX fluorescence was recorded on a Safire plate reader (405 nm excitation, 630 nm emission). Data were normalized to untreated controls and plotted as mean ± SD. Statistical significance was set at p < 0.05 (GraphPad Prism 6).
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Environment‑Responsive Metal–Organic Frameworks: Precision Drug Delivery for Tumor Therapy
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Antibody‑Functionalized Silica‑Coated Gold Nanorods for Rapid Diagnosis and Photothermal Treatment of Cryptococcus neoformans
- Hydrophilic HBP‑Modified rGO Enables Dual pH/NIR‑Triggered DOX Release for Synergistic Chemo‑Photothermal Tumor Therapy
- Polydopamine Core–Shell Nanoparticles with Redox‑Responsive Polymer Shells for Targeted Drug Delivery and Synergistic Chemo‑Photothermal Therapy
- Mitochondria‑Targeted TiS2 Nanosheets Deliver Resveratrol for Photothermal‑Triggered Tumor Chemotherapy
- Targeted Doxorubicin Delivery Using Anti-EpCAM Aptamer-Functionalized Liposomes: Efficacy in C26 Colon Carcinoma Models
- Easy Fabrication of rGO@Fe3O4 Microspheres for Magnetically Targeted, NIR-Responsive Chemo‑Photothermal Cancer Therapy



