Tin Disulfide Nanoflake-Based Electrochemical Biosensor for Rapid, Sensitive Detection of Salivary Cortisol
Abstract
Cortisol, the quintessential stress hormone, is secreted by the hypothalamic‑pituitary‑adrenal (HPA) axis and serves as a critical biomarker for psychological and physiological stress. Prolonged cortisol over‑expression can lead to serious disorders such as Cushing’s and Addison’s disease, underscoring the need for rapid, point‑of‑care (POC) detection. Here, we report a non‑invasive electrochemical assay for salivary cortisol based on a multilayer film of tin disulfide (SnS₂) nanoflakes, a cortisol‑specific antibody (C‑M_ab), and bovine serum albumin (BSA) immobilized on a glassy carbon electrode (GCE). The BSA/C‑M_ab/SnS₂/GCE sensor was characterized by electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV). Differential pulse voltammetry (DPV) revealed a linear response to cortisol concentrations ranging from 100 pM to 100 µM, with a limit of detection (LOD) of 100 pM and a sensitivity of 0.0103 mA M⁻¹ cm⁻² (R² = 0.9979). Saliva samples measured with the sensor matched ELISA results within a 5 % margin, demonstrating its clinical relevance and specificity. The device offers a rapid, cost‑effective alternative to conventional immunoassays for monitoring stress‑related biomarkers.
Introduction
Cortisol is a steroid hormone released by the HPA axis and is the most widely used biomarker for assessing psychological stress. Its circadian rhythm peaks in the early morning and declines by night, and dysregulation can trigger conditions ranging from Cushing’s syndrome to Addison’s disease. Accurate, real‑time monitoring of cortisol is therefore essential for diagnosing and managing a variety of stress‑related disorders, including PTSD, depression, and autism spectrum disorder.
Current laboratory methods—chromatography, radioimmunoassay, ELISA, SPR, and QCM—are laboratory‑bound, time‑consuming, and expensive, limiting their use in POC settings. Electrochemical immunosensors, which rely on specific antigen–antibody interactions, offer a promising solution with simple instrumentation, rapid readouts, low cost, and high sensitivity. Salivary cortisol measurement is especially attractive because it is non‑invasive and requires minimal sample handling.
While numerous metal sulfide nanomaterials have been explored for biosensing, tin disulfide (SnS₂) has rarely been used as a protein‑immobilization matrix. SnS₂ is an n‑type semiconductor with a bandgap of 2.18–2.44 eV, high carrier mobility, and excellent chemical stability, making it an ideal platform for electrochemical transduction. In this work, we harness 2‑D SnS₂ nanoflakes to construct a highly sensitive cortisol sensor that operates across the physiological concentration range.
Materials and Methods
Materials
Hydrocortisone, anti‑rabbit cortisol antibody (C‑M_ab), potassium hexacyanoferrate (II/III), β‑estradiol, testosterone, progesterone, corticosterone, and bovine serum albumin (BSA) were sourced from Sigma‑Aldrich (St. Louis, MO). Tin(IV) chloride pentahydrate and thioacetamide were supplied by Showa (Japan) and Alfa Aesar (UK), respectively. Phosphate‑buffered saline (PBS) and micro‑polished alumina were obtained from Sigma‑Aldrich and Buehler (UK). All reagents were used without further purification. A commercial cortisol ELISA kit (Cat # SA E‑6000) was purchased from LDN (Germany).
Synthesis of Tin Disulfide
SnCl₄·5H₂O and C₂H₅NS were dissolved in 70 mL deionized water and adjusted to pH 7.4. The solution was transferred to a hydrothermal autoclave and heated to 200 °C within 1 h, then maintained for 11 h. The resulting SnS₂ powder was washed with water and ethanol at 6000 rpm for 15 min and dried at 80 °C in air, yielding uniform nanoflakes.
Materials Characterization
X‑ray diffraction (XRD) confirmed the hexagonal SnS₂ phase (JCPDS 89‑2358). Field‑emission scanning electron microscopy (FE‑SEM) revealed flake‑like morphology with lateral dimensions ~300 nm. Transmission electron microscopy (TEM) and selected‑area electron diffraction (SAED) confirmed single‑crystalline structure with lattice spacings of 0.167 nm and 0.316 nm; the nanoflakes were less than 10 nm thick.
Fabrication of BSA/C‑M_ab/SnS₂/GCE Biosensors
Glass‑carbon electrodes (GCE) were polished with alumina slurry and drop‑cast with a 5 M SnS₂ dispersion. The electrodes were then sequentially incubated with 1 mg mL⁻¹ C‑M_ab and 1 % BSA solutions in PBS. The finished sensors were stored at 4 °C when not in use. The detection concept and setup are illustrated in Fig. 1.
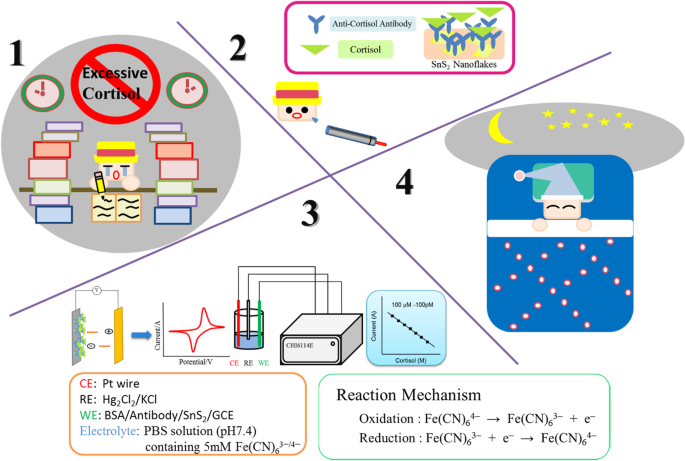
Research concept and setup of the detection system
Electrochemical Analysis
Electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) were employed to assess the sensor’s electroactive behavior. All measurements used a three‑electrode cell: the BSA/C‑M_ab/SnS₂/GCE as working electrode, a Pt wire as counter, and a saturated calomel electrode as reference, in 10 mM PBS (pH 7.4) containing 5 mM Fe(CN)₆³⁻/⁴⁻. CV and differential pulse voltammetry (DPV) scans were performed from –0.4 V to 1.0 V at 10 mV s⁻¹.
Saliva Sample Collection and Electrochemical Sensing
Two healthy volunteers provided 2 mL saliva samples at noon. Samples were frozen at –20 °C, thawed, centrifuged (3500 rpm, 15 min), and the supernatant was used for analysis. The sensor’s cortisol measurements were compared with results from the ELISA kit.
Interference Study
Potential interferents—β‑estradiol, testosterone, progesterone, corticosterone (each 100 nM)—were tested by incubating the sensor for 10 min and measuring CV response. The influence on cortisol detection was below 5 %.
Detection of Salivary Cortisol by ELISA
ELISA followed the manufacturer’s protocol. A standard curve was generated with six cortisol concentrations (0, 0.1, 0.4, 1.7, 7.0, 30 ng mL⁻¹). Absorbance was read at 450 nm and the resulting curve was used to quantify unknown samples.
Results and Discussion
Material Analysis of SnS₂
The XRD pattern (Fig. 2a) matches the hexagonal SnS₂ phase (JCPDS 89‑2358). FE‑SEM images (Fig. 2b,c) show uniform, 300‑nm flake‑like particles. TEM (Fig. 2d–f) confirms single‑crystalline flakes with interlayer spacings of 0.167 nm and 0.316 nm, and a total thickness below 10 nm.
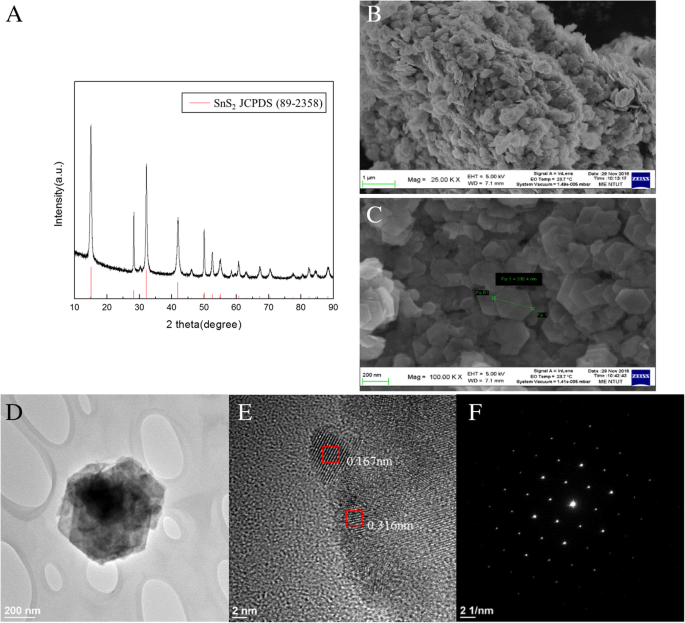
a XRD pattern of SnS₂. FE‑SEM images of SnS₂ nanoflakes at (b) ×250,000 and (c) ×100,000. d TEM images of SnS₂ nanoflakes. e Cross‑sectional TEM and zoomed view. f SAED pattern.
Electrochemical Responses of the Electrode
Adding SnS₂ to GCE significantly enhanced oxidation current (Fig. 3a,b). Subsequent antibody and BSA modification increased charge‑transfer resistance, as reflected in EIS data (Fig. 3b). CV of the BSA/C‑M_ab/SnS₂/GCE at scan rates 10–100 mV s⁻¹ showed a linear increase in peak current (Fig. 3c,d), confirming a surface‑controlled, reversible redox process.
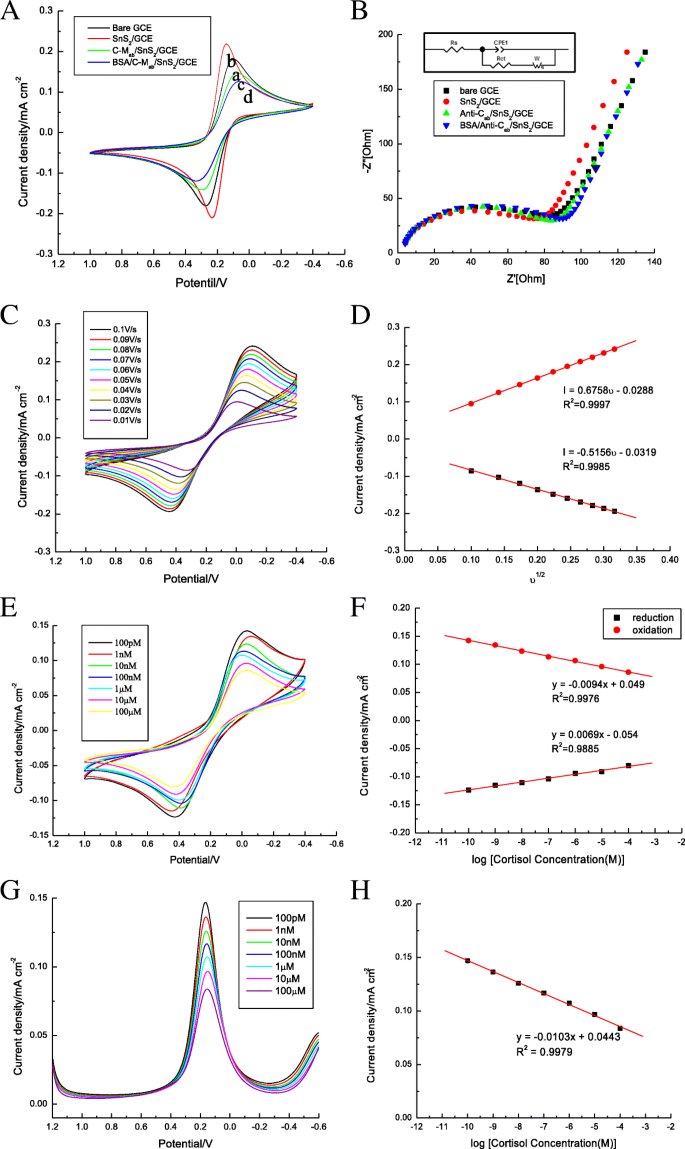
a CV of GCE, SnS₂/GCE, C‑M_ab/SnS₂/GCE, BSA/C‑M_ab/SnS₂/GCE. b EIS of each electrode. Inset: equivalent circuit. c Oxidation current vs. scan rate. d Linear fit of current vs. scan rate. e CV of BSA/C‑M_ab/SnS₂/GCE vs. cortisol (100 pM–100 µM). f Calibration curve (log cortisol). g DPV of BSA/C‑M_ab/SnS₂/GCE vs. cortisol (100 pM–100 µM). h DPV calibration curve.
Both CV and DPV demonstrated a monotonic decrease in peak current with increasing cortisol concentration. DPV yielded a linear relationship: y = –0.0103 x + 0.0443 (R² = 0.9979), with a LOD of 100 pM and a sensitivity of 0.0103 mA M⁻¹ cm⁻². The sensor’s dynamic range (100 pM–100 µM) covers the full physiological spectrum of salivary cortisol.
Storage Stability Study
CV was performed on sensors stored for 1 day to 1 week under two conditions: vacuum‑dry and 4 °C. The 4 °C storage preserved 91 % of the redox peak current after 7 days, compared to 82 % under vacuum, indicating better stability of antibody activity at lower temperature.
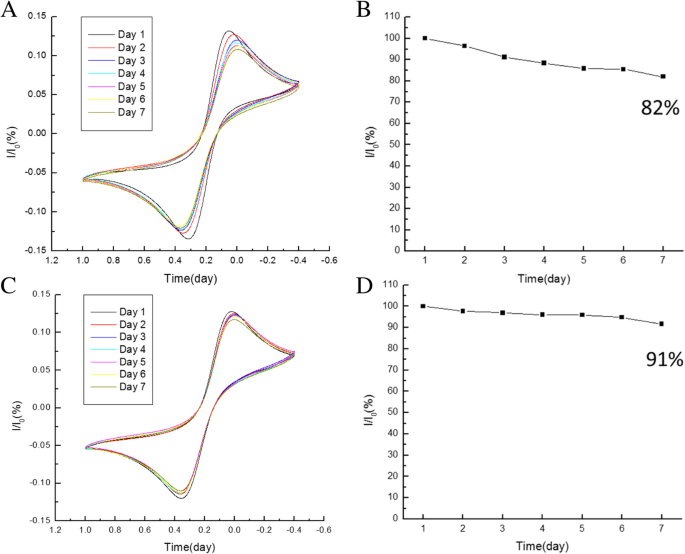
Redox peak stability of BSA/C‑M_ab/SnS₂/GCE with (a,b) vacuum and (c,d) 4 °C storage over 7 days.
Interference Study
Incubation with 100 nM β‑estradiol, testosterone, progesterone, and corticosterone produced <5 % change in cortisol signal (Fig. 5a), confirming high assay specificity.
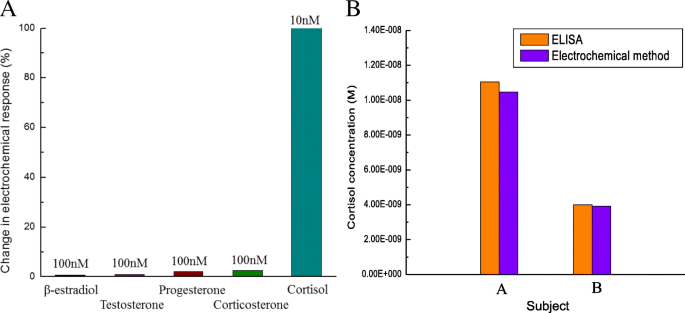
a Interference study with β‑estradiol, testosterone, progesterone, corticosterone (100 nM) vs. cortisol (10 nM). b Comparison of salivary cortisol by ELISA and electrochemical sensor.
Detection of Salivary Cortisol Using ELISA and Electrochemical Methods
Saliva samples from two volunteers yielded cortisol concentrations of 1.105 × 10⁻⁸ M and 3.998 × 10⁻⁹ M by ELISA, and 1.046 × 10⁻⁸ M and 3.911 × 10⁻⁹ M by the electrochemical sensor—only a 2–5 % discrepancy—confirming the sensor’s accuracy.
Comparison with Other Studies
Table 2 and 3 (not shown) compare our sensor to non‑gold‑based cortisol sensors. Advantages of the present work include: (1) significantly lower material cost; (2) a rapid, straightforward fabrication protocol; and (3) an LOD comparable to, or better than, existing devices.
Conclusions
We have demonstrated a tin disulfide nanoflake‑based electrochemical biosensor that delivers a 100 pM LOD and a 100 pM–100 µM detection range for salivary cortisol, matching ELISA accuracy. The sensor exhibits <5 % interference from structurally similar steroids and maintains >90 % activity after 7 days at 4 °C. Its low cost, simple assembly, and rapid readout make it a compelling candidate for POC stress monitoring.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- 2D:
Two‑dimensional
- BSA:
Bovine serum albumin
- C-M_ab:
Cortisol antibody
- CV:
Cyclic voltammetry
- DPV:
Differential pulse voltammetry
- EIS:
Electrochemical impedance spectroscopy
- ELISA:
Enzyme‑linked immunosorbent assay
- FEG-TEM:
Field emission gun transmission electron microscope
- FE-SEM:
Field emission scanning electron microscope
- GCE:
Glassy carbon electrode
- HPA:
Hypothalamic‑pituitary‑adrenal
- PBS:
Phosphate buffered saline
- POC:
Point‑of‑care
- PTSD:
Post‑traumatic stress disorder
- SAED:
Selected area diffraction
- XRD:
X‑ray diffraction
Nanomaterials
- Tin: From Bronze Age to Modern Industry – Uses, Production, and Future
- Tin Nanocrystals: The Next Frontier for High‑Capacity Lithium‑Ion Batteries
- Transparent, Flexible SiOx–Graphene Memory: A Leap Beyond Conventional Flash
- Ultra‑Sensitive Electrochemical DNA Biosensor Using Acrylic‑Gold Nanocomposite for Rapid Arowana Fish Gender Determination
- Dielectric Nanoprism-Based Plasmonic Sensor with Normal‑Incidence SPR for Enhanced Sensitivity
- Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
- Polarization‑Insensitive Plasmonic Electro‑Absorption Modulator Using Epsilon‑Near‑Zero Indium Tin Oxide
- Ultra-Stable Pt–Ni Jagged Nanowire Sensor for Highly Sensitive Caffeic Acid Detection
- Rapid Electrochemical Sensor for Polychlorinated Biphenyls Using β‑Cyclodextrin/Tin Disulfide‑Modified Screen‑Printed Carbon Electrodes
- MoS₂ Nanocomposite-Based Enzyme-Free Glucose Biosensors: A Cutting-Edge Approach



