Rapid Electrochemical Sensor for Polychlorinated Biphenyls Using β‑Cyclodextrin/Tin Disulfide‑Modified Screen‑Printed Carbon Electrodes
Abstract
Polychlorinated biphenyls (PCBs) are persistent, endocrine‑disrupting pollutants that pose serious health risks, including carcinogenesis and reproductive toxicity. This study presents the synthesis of a composite material comprising 3A‑amino‑3A‑deoxy‑(2AS,3AS)-β‑cyclodextrin hydrate (β‑CD) and tin disulfide (SnS2) and its application as a modified screen‑printed carbon electrode (SPCE) for PCB detection. SnS2 nanoflakes were produced hydrothermally, then sequentially deposited onto the SPCE by micropipette, followed by β‑CD coating. The resulting β‑CD/SnS2/SPCE exhibits high conductivity and selective PCB binding through host–guest inclusion, enabling electrochemical monitoring via cyclic voltammetry (CV) and differential pulse voltammetry (DPV). The sensor displays a linear detection range of 0.625–80 µM with a limit of detection (LOD) of 5 µM, and retains 88 % of its initial response after 7 days of storage.
Introduction
PCBs remain ubiquitous environmental contaminants due to their historical use in electrical insulation and their resistance to degradation. Despite regulatory bans, PCBs persist in soil, water, and biota, necessitating sensitive, rapid detection methods. Conventional analytical techniques—LC/MS and GC/MS—are accurate but costly, time‑consuming, and require skilled operators. Electrochemical sensing offers a low‑cost, portable alternative, yet few reports address PCB detection via modified electrodes. Enhancing electrode conductivity and selectivity through nanomaterials is essential; here, SnS2 provides an n‑type semiconductor with high surface area, while β‑CD introduces a hydrophobic cavity that selectively encapsulates PCBs.
β‑Cyclodextrin (CD) is a cyclic oligosaccharide with a hydrophilic exterior and a hydrophobic interior cavity, enabling host–guest interactions with hydrophobic analytes. The 3A‑amino‑3A‑deoxy‑β‑CD variant offers improved solubility and functionalization potential. Tin disulfide (SnS2) is a layered metal dichalcogenide (MDC) with a 2.2 eV indirect band gap, widely studied for electrochemical sensors due to its favorable electron‑transfer characteristics.
In this work, we synthesize SnS2 nanoflakes via a hydrothermal route, combine them with β‑CD on an SPCE, and evaluate the composite’s electrochemical response to PCBs, demonstrating a rapid, selective, and stable sensor platform.
Materials and Methods
Materials
SnCl4·5H2O, thioacetamide, methanol, phosphate buffers, potassium ferrocyanide/ferricyanide, and 3A‑amino‑3A‑deoxy‑β‑CD were sourced from Alfa, Sigma‑Aldrich, and BaseChem. PCBs (Aroclor 1016) were purchased from Merck.
Instruments
Morphology was examined by FE‑SEM (ZEISS), phase analysis by XRD (PANalytical), and electrochemical measurements by CHI‑6114E in a conventional three‑electrode setup (SPCE as working, Ag/AgCl reference, Pt counter). Electrolytes were 3 mM ferro/ferricyanide + 0.1 M KCl or 10 mM PBS (pH 7.4). Potential window: −0.6 to +1.0 V; scan rate: 0.05 V s−1.
Synthesis of Tin Disulfide
In 70 mL water, 0.351 g SnCl4·5H2O and 0.3 g thioacetamide were stirred for 1 h. 1 M NaOH was added dropwise to reach pH ≈ 10.5. The solution was transferred to a stainless‑steel autoclave, heated from 25 °C to 200 °C (1 h) and then held at 200 °C for 11 h. After cooling, the product was washed with water and ethanol, centrifuged (6000 rpm, 30 min), and dried.
Fabrication of β‑CD/SnS2/SPCE
0.02 g SnS2 was dispersed in 5 mL water; 2 µL of this suspension was pipetted onto the SPCE surface, dried in a vacuum dryer for 10 min, and repeated five times. Next, 2 µL of 1 mM β‑CD in water was applied, dried similarly, yielding the final composite electrode (see Fig. 2).
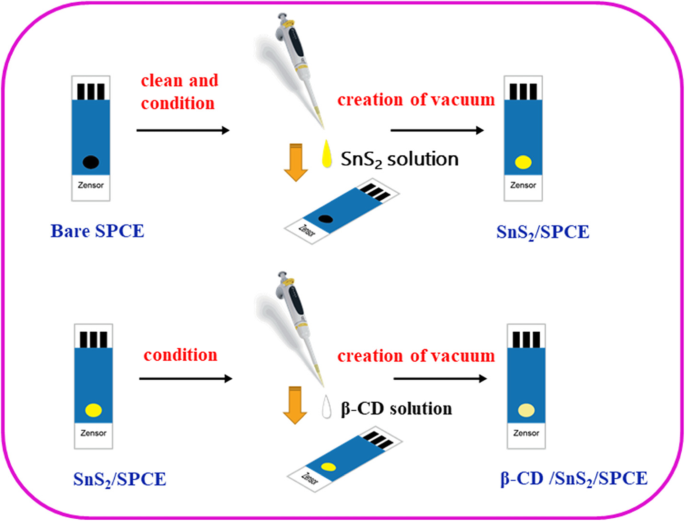
The preparation and fabrication of β‑CD/SnS2/SPCE
Results and Discussion
Crystal Structure of SnS2
XRD analysis confirmed the hexagonal SnS2 phase (JCPDS 89‑2358), with characteristic peaks at 15°, 29°, 30°, 31°, 41°, 46°, 50°, 51°, 53°, and 70° (Fig. 3).
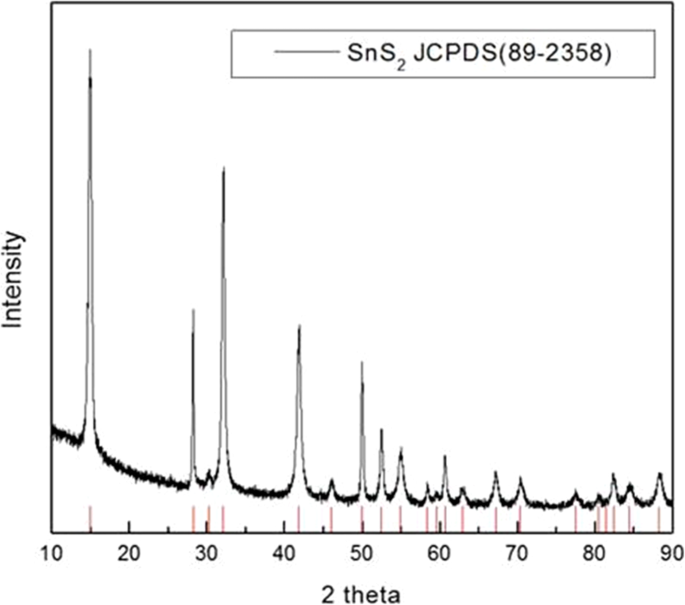
The XRD pattern of SnS2
Morphology of SnS2
FE‑SEM images revealed hexagonal nanoflakes with widths ranging from 220 nm to 322 nm (Fig. 4).

a FE‑SEM images of SnS2 at different magnifications.b Nanoflake widths: 322, 298, 220 nm.
Electrochemical Impedance and Electrolyte Effect
Electrochemical impedance spectroscopy (EIS) showed that β‑CD/SnS2/SPCE has the lowest charge‑transfer resistance, indicating superior conductivity (Fig. 5a). When tested in two electrolytes, the mixed ferro/ferricyanide + KCl solution produced a well‑defined redox peak, whereas PBS did not (Fig. 5b). Thus, the mixed electrolyte is optimal for PCB detection.
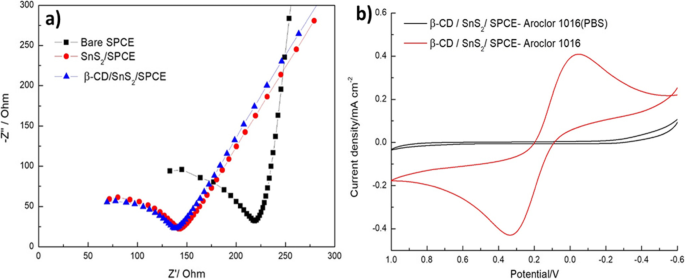
a EIS spectra of bare SPCE, SnS2/SPCE, and β‑CD/SnS2/SPCE.b β‑CD/SnS2/SPCE in PBS (black) and mixed electrolyte (red) with 80 µM PCBs.
Electrochemical Response of Modified Electrodes
CV measurements in the mixed electrolyte demonstrated that SnS2/SPCE outperforms bare SPCE, while β‑CD/SnS2/SPCE shows the highest peak currents due to improved conductivity and lack of electron‑transfer barriers (Fig. 6a). Upon PCB addition, the peak current decreased sharply because β‑CD encapsulates PCBs, blocking the ferro/ferricyanide redox process.
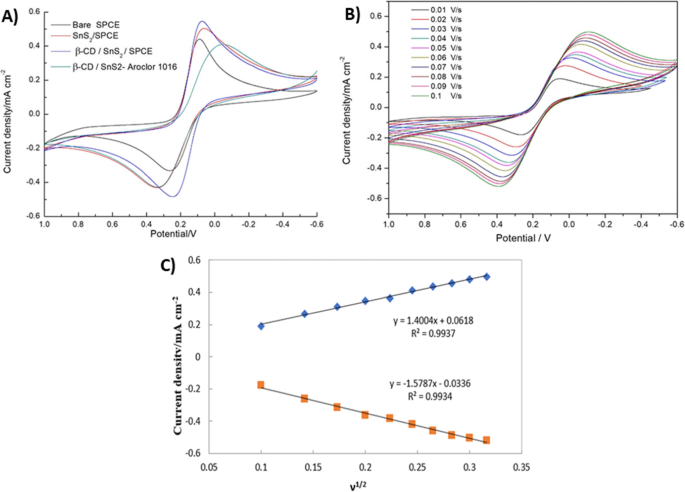
a CVs of bare SPCE, SnS2/SPCE, and β‑CD/SnS2/SPCE in mixed electrolyte.b CVs of β‑CD/SnS2/SPCE with 80 µM PCBs at scan rates 0.01–0.10 V s−1.c Calibration plots of anodic and cathodic peak currents vs. √scan rate, confirming diffusion‑controlled kinetics (R2 = 0.9937/0.9934).
Effect of Scan Rate
Peak currents increased linearly with √scan rate, yielding an electron‑transfer rate constant ks = 0.039 s−1 and surface coverage τ = 8.14 × 10−9 mol cm−2.
Concentration Dependence
CV analysis showed negligible response changes below 2.5 µM PCBs; significant current reduction began at 5 µM. A linear relationship between log concentration (5–80 µM) and peak current (R2 ≈ 0.98) confirms reliable quantification (Fig. 7).
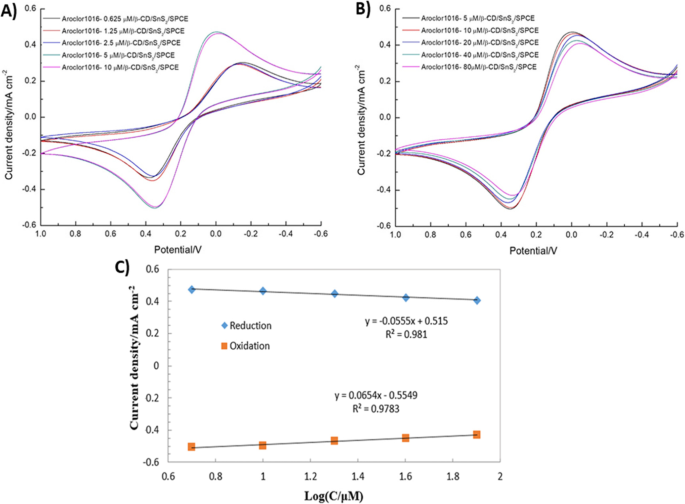
a CVs for 0.625–2.5 µM PCBs.b CVs for 5–80 µM PCBs.c Log‑concentration vs. anodic/cathodic peak current density.
Differential Pulse Voltammetry
DPV provided higher sensitivity, with a clear linear decline in peak current from 5 to 80 µM PCBs. The sensor’s LOD of 5 µM matches the CV results, and methanol addition lowered the LOD to 1.25 µM without compromising selectivity (Fig. 8–10).
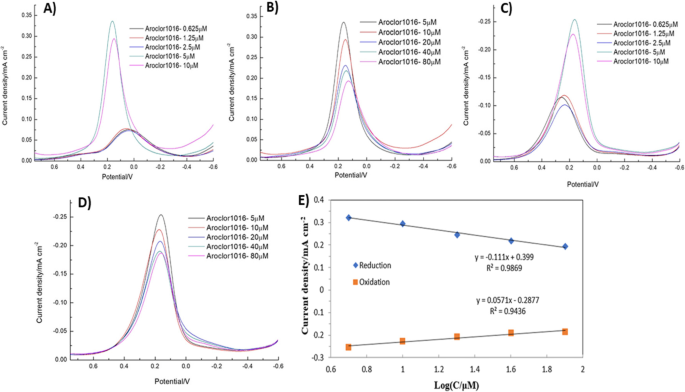
a–b Reduction peak currents vs. PCB concentration.c–d Oxidation peak currents vs. PCB concentration.e Plot of peak current density vs. log concentration.
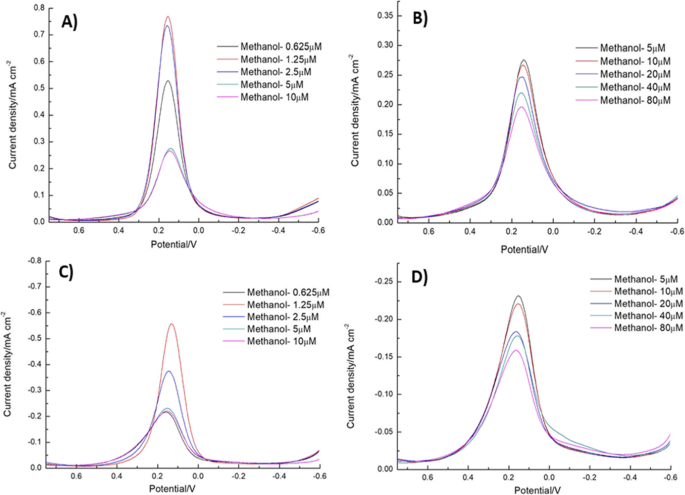
a–c Reduction and oxidation currents for 1.25–10 µM PCBs in methanol.b–d Currents for 5–80 µM PCBs in methanol.
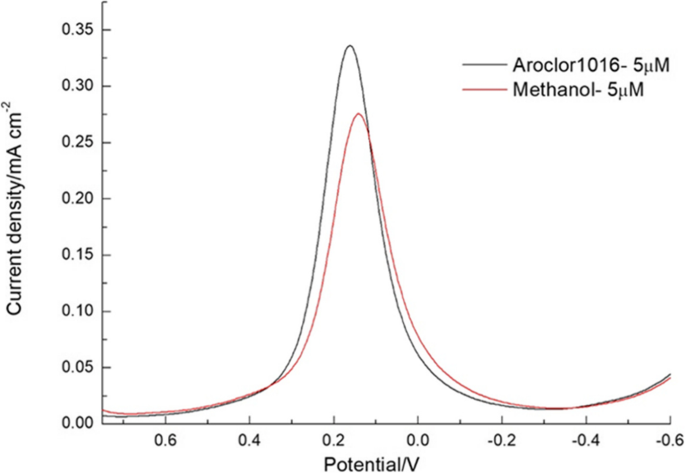
DPV comparison of 5 µM PCBs in methanol vs. methanol alone.
Stability
After 7 days of storage at room temperature, the sensor retained 88 % of its initial current response, indicating robust operational stability (Fig. 11).
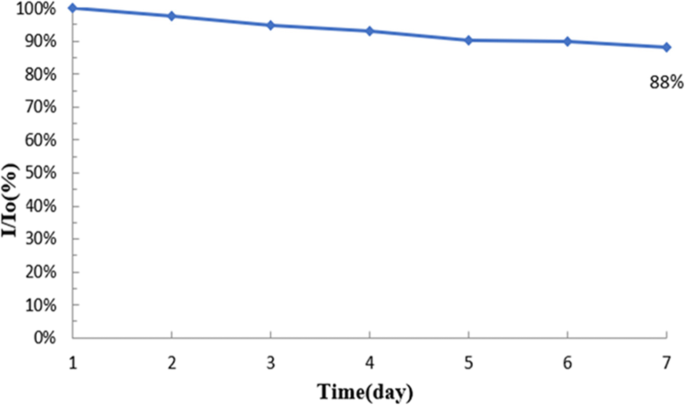
Stability test over 7 days at room temperature.
Conclusion
The hydrothermally synthesized SnS2 nanoflakes, when combined with β‑CD on a screen‑printed carbon electrode, yield a sensitive and selective electrochemical sensor for PCBs. With a linear range of 0.62–80 µM, an LOD of 5 µM, and 88 % stability over 7 days, the β‑CD/SnS2/SPCE platform offers a rapid, cost‑effective alternative to conventional chromatographic methods for on‑site PCB monitoring.
Availability of Data and Materials
All data generated or analyzed during this study are included in the published article.
Abbreviations
- 2D
- Two‑dimensional
- CV
- Cyclic voltammetry
- DPV
- Differential pulse voltammetry
- EIS
- Electrochemical impedance spectroscopy
- FE‑SEM
- Field emission scanning electron microscope
- GC/MS
- Gas chromatography‑mass spectrometry
- LC/MS
- Liquid chromatography‑mass spectrometry
- MDCs
- Metal dichalcogenides
- PBS
- Phosphate‑buffered saline
- PCBs
- Polychlorinated biphenyls
- POPs
- Persistent organic pollutants
- SnS2
- Tin disulfide
- SPCE
- Screen‑printed carbon electrode
- XRD
- X‑ray diffraction
- β‑CD
- 3A‑amino‑3A‑deoxy‑(2AS,3AS)-β‑cyclodextrin hydrate
Nanomaterials
- Visible‑Light‑Assisted Au Nanoparticle‑Modified Glassy Carbon Electrodes for Sensitive Uric Acid Detection
- Dual-Mode Carbon Nanodot Sensors for Rapid, Selective Hydrogen Peroxide Detection
- Monodisperse Carbon Nanospheres with Hierarchical Porosity Deliver Superior Supercapacitor Performance
- High-Sensitivity Graphene Oxide Fluorescent Aptasensor for Rapid Detection of CCRF-CEM Leukemia Cells
- Electrochemical Energy Storage: Enhancing Capacitance with Reduced Graphene Oxide/Carbon Nanotube Composites
- Rapid, Low‑Cost Microfluidic Electrochemical Sensor Fabricated Directly on Screen‑Printed Electrodes for Ultra‑Sensitive PSA Detection
- Tin Disulfide Nanoflake-Based Electrochemical Biosensor for Rapid, Sensitive Detection of Salivary Cortisol
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Advanced Detection of miRNA‑335‑5p Using Interdigitated Electrodes to Assess Abdominal Aortic Aneurysm Severity
- Optimizing LaCoO3 with Sr Doping and rGO for Superior Energy Storage Performance



