Visible‑Light‑Assisted Au Nanoparticle‑Modified Glassy Carbon Electrodes for Sensitive Uric Acid Detection
Abstract
We report a highly sensitive, non‑enzymatic approach for uric acid (UA) determination that leverages visible‑light illumination on a glassy carbon electrode (GCE) modified by electrodeposited gold nanoparticles (AuNPs). Scanning electron microscopy (SEM) confirmed that the AuNPs are well‑dispersed, spherical particles with an average diameter of 26.1 nm. Cyclic voltammetry (CV) and differential pulse voltammetry (DPV) studies revealed that visible light dramatically enhances both the magnitude and stability of the response current through the surface plasmon resonance (SPR) effect of AuNPs. DPV displayed a linear correlation between peak current and UA concentration from 2.8 to 57.5 µM, described by Ipa (µA)=0.0121 cUA (µM)+0.3122 (R²=0.9987). These results demonstrate the potential of the visible‑light‑enhanced Au/GCE as a reliable electrochemical sensor for UA.
Background
Uric acid is the terminal product of purine metabolism and its concentration in blood and urine serves as a biomarker for conditions such as gout, kidney disease, hypertension, and cardiovascular disorders. Conversely, low UA levels have been linked to multiple sclerosis, Parkinson’s disease, Alzheimer’s disease, and optic neuritis. Consequently, accurate monitoring of UA in biological fluids is clinically indispensable.
Traditional UA assays—including enzymatic tests, HPLC, mass spectrometry, capillary electrophoresis, chemiluminescence, and colorimetry—often require complex sample preparation and lengthy analysis times. Electrochemical methods, exploiting the irreversible oxidation of UA to allantoin, offer a faster and simpler alternative. Recent advances have introduced a variety of non‑enzymatic sensors: palladium‑loaded carbon nanofibers, carbon ionic liquid electrodes, PtAu hybrid films, and chitosan–graphene composites, among others.
Nanometals—Ag, Au, Pt, Cu, Pd—have been extensively employed to fabricate electrochemical sensors due to their excellent conductivity, large surface area, and biocompatibility. Gold nanoparticles, in particular, benefit from a pronounced SPR that can harvest visible light and enhance catalytic activity. This property motivates the present study, which combines AuNPs with visible‑light illumination to boost UA detection.
Methods
Apparatus
The morphology of the modified electrode was examined with a Hitachi SU8010 SEM. Electrochemical measurements were conducted on a CHI760D workstation using a three‑electrode configuration: Au/GCE (0.07 cm²) as the working electrode, a platinum wire counter electrode, and a saturated calomel reference electrode. pH values were measured with a pHS-2F meter.
Reagents
Uric acid was sourced from Acros Organics. Chloroauric acid, disodium hydrogen phosphate, sodium dihydrogen phosphate, potassium ferrocyanide, potassium ferricyanide, and potassium chloride were purchased from Sinopharm. All reagents were used as received; experiments were performed at room temperature.
Electrode Preparation
An L‑type GCE was polished with 0.05 µm alumina, ultrasonically cleaned, and rinsed until the CV in 2.5 mM Fe(CN)₆³⁄⁴⁻/0.1 M KCl displayed symmetrical peaks with ΔEp <100 mV. AuNPs were electrodeposited from 1 mM H₃PO₄/0.24 mM HAuCl₄ at –0.2 V, with mass loading tuned by the deposited charge.
Results and Discussion
Morphology and Characterization of Electrode
SEM images (Figure 1) show a densely packed, monodisperse distribution of spherical AuNPs on the GCE surface. The size histogram indicates an average diameter of 26.1 nm, confirming a narrow size distribution that enhances the active surface area for UA oxidation.

SEM images of Au/GCE at lower (a) and higher magnification (b). Inset: corresponding Au nanoparticle size distribution histogram
Cyclic Voltammetric Behaviors
CVs in 0.1 M PBS (pH 7.0) containing 1.0 mM UA (Figure 2) demonstrate that visible‑light illumination (20 cm from a Xe lamp) increases the anodic peak current from 12 µA (dark) to 20 µA (lit) and stabilizes the response over successive scans. The peak potential shifts ~6 mV negative under illumination, indicating enhanced electron transfer.
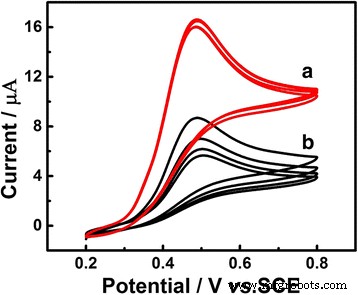
CVs of Au/GCE in 0.1 M PBS (pH 7.0) containing 1.0 mM UA with (a) and without (b) visible‑light illumination. Scan rate 50 mV s−1
Effect of Visible Light Illumination
During successive CV cycles, illumination triggered a rapid rise in current (Figure 3a). Photocurrent-time curves (Figure 3b) show immediate decreases when the lamp is switched off and quick recoveries upon re‑illumination, confirming a steady and reversible SPR‑driven enhancement. UV–Vis spectra (Additional file 1: Figure S2) exhibit an absorption peak at 597 nm, consistent with SPR of AuNPs. Hot‑electron transfer from Au to the GCE surface accelerates UA oxidation and improves sensitivity.
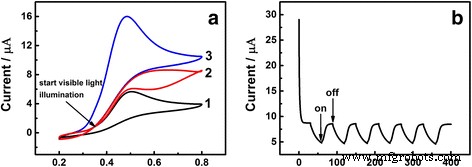
a Potential vs. SCE. CV of Au/GCE in 0.1 M PBS (pH 7.0) containing 1.0 mM UA without (1) and with (3) visible‑light illumination; illumination starts at the arrow (2). Scan rate 50 mV s−1. b Time/s. Photocurrent response at 0.48 V with 30‑s lamp on/off cycles.
Effect of Gold Quantity
Varying the Au loading (0.96–57.8 µg cm−2) revealed an optimal density of 28.9 µg cm−2, where peak current was maximized and potential most negative. Excess Au (>57.8 µg cm−2) slightly reduced current due to hindered electron transfer. The chosen loading provides the best balance of surface area and conductivity.
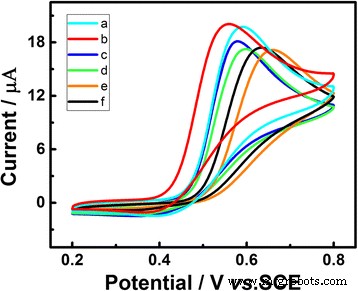
CVs of Au/GCE with different Au loadings (57.8 (a), 28.9 (b), 9.6 (c), 4.8 (d), 0.96 (e), bare GCE (f)) in 0.1 M PBS (pH 7.0) with 1.0 mM UA under visible‑light illumination. Scan rate 50 mV s−1; deposition potential –0.2 V.
Effect of pH Value
The optimal pH for UA detection was found to be 7.0, as indicated by the maximum anodic peak current (Figure 5). Current decreased gradually from pH 7.0 to 9.0, confirming that neutral conditions favor the electrochemical oxidation of UA.
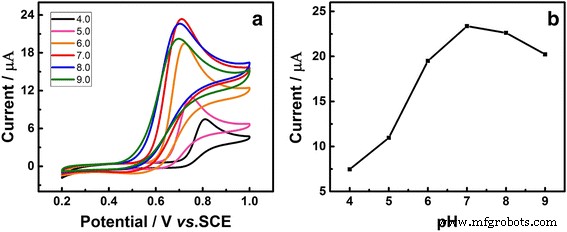
a CVs of Au/GCE in 1 mM UA at various pH (4.0–9.0). b Peak current vs. pH. Scan rate 50 mV s−1
Effect of Scan Rate
Increasing the scan rate from 20 to 200 mV s−1 produced a positive shift in peak potential, indicating a quasi‑reversible reaction. A linear relationship between anodic peak current and √ν (R²=0.9702) confirms diffusion‑controlled UA oxidation (Figure 6).
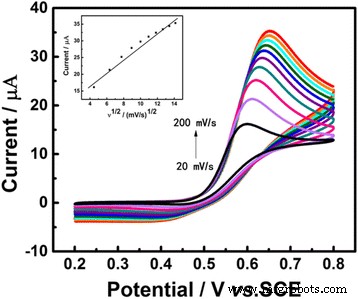
CVs of Au/GCE in 0.1 M PBS (pH 7.0) with 1 mM UA at scan rates 20–200 mV s−1. Inset: calibration curve of peak current vs. √ν.
Determination of UA
DPV analysis (Figure 7) exhibited a linear response from 2.8 to 57.5 µM UA, described by Ipa (µA)=0.0121 cUA (µM)+0.3122 (R²=0.9987). Without illumination, the linear range narrowed to 3–21 µM (R²=0.9943). The sensor demonstrated 7.88 % RSD over eight electrodes and a 9.8 % signal loss after one week storage, indicating good reproducibility and stability. Interference studies and real‑sample analysis (human serum) yielded 96.3 % recovery, confirming practical applicability.
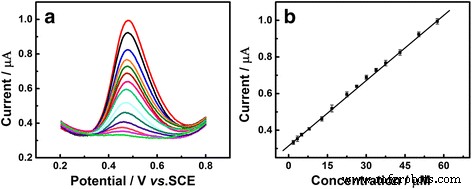
a DPV curves of Au/GCE in 0.1 M PBS (pH 7.0) at various UA concentrations. b Anodic peak current vs. UA concentration.
Conclusions
Homogeneous, spherical AuNPs were electrodeposited onto GCE, yielding a well‑dispersed nanostructure with an average diameter of 26.1 nm. The visible‑light‑assisted Au/GCE exhibited superior electrochemical performance, outperforming the dark condition through SPR‑induced photocurrent enhancement. The resulting sensor offers a simple, rapid, and reliable method for UA quantification, with potential extension to other biomolecules.
Abbreviations
- AuNPs:
Gold nanoparticles
- CE:
Capillary electrophoresis
- CV:
Cyclic voltammetry
- DPV:
Differential pulse voltammetry
- GCE:
Glassy carbon electrode
- HPLC:
High‑performance liquid chromatography
- SEM:
Scanning electron microscope
- SPR:
Surface plasmon resonance
- UA:
Uric acid
Nanomaterials
- Al2O3:SiOC Nanocomposites from Phenyltrimethoxysilane‑Modified Alumina—Synthesis, Structure, and Photoluminescence
- Ultra‑Sensitive Electrochemical DNA Biosensor for Vibrio cholerae Detection Using Polystyrene‑Acrylic Acid Nanoparticles and Gold Nanoparticles
- GoldMag: PSS‑MA Coating Enhances Stability & Optical Precision for Protein Detection
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Poly(γ‑Glutamic Acid) Enhances Fe‑Pd Nanoparticle‑Catalyzed Dechlorination of p‑Chlorophenol Under Alkaline Conditions
- Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
- Rapid Electrochemical Sensor for Polychlorinated Biphenyls Using β‑Cyclodextrin/Tin Disulfide‑Modified Screen‑Printed Carbon Electrodes
- Ultrasensitive Room‑Temperature Formaldehyde Sensor Using Ag‑Doped In₂O₃ Nanograins
- Advanced Detection of miRNA‑335‑5p Using Interdigitated Electrodes to Assess Abdominal Aortic Aneurysm Severity
- Copper Nanoparticles on Porous Carbon Substrates Deliver High-Performance Glucose Sensors



