Metallic Nanoparticles: Potent Efflux Pump and Biofilm Inhibitors to Restore Antibiotic Efficacy
Antibiotic resistance remains a critical global health challenge, driven by bacterial adaptations such as drug modification, target alteration, reduced permeability, and active extrusion via efflux pumps. These pumps not only expel antibiotics but also regulate quorum‑sensing molecules that drive biofilm formation, a key factor in chronic infections. Metallic nanoparticles have emerged as promising efflux‑pump inhibitors; by blocking these transporters, they can restore the potency of conventional antibiotics and diminish biofilm formation. This review examines recent evidence on the synergy between metallic nanoparticles and antibiotics, focusing on efflux‑pump inhibition and biofilm disruption. Chronic infections associated with biofilms are notoriously difficult to eradicate because biofilms shield bacteria from both host immunity and antibiotics [1]. The biofilm matrix is a primary barrier that converts acute infections into persistent, hard‑to‑treat conditions [2]. According to the National Institutes of Health and Centers for Disease Control, 65–80 % of infectious diseases are caused by biofilm‑forming bacteria, especially the Gram‑negative species Pseudomonas aeruginosa and Escherichia coli, and the Gram‑positive pathogen Staphylococcus aureus [3]. Conventional antibiotics often fail against biofilm‑embedded bacteria because they cannot penetrate the extracellular matrix and are rapidly expelled by bacterial efflux systems [4]. Efflux pumps—membrane‑bound transporters with broad substrate specificity—serve as a major drug resistance mechanism, expelling toxic compounds and antibiotics alike [5–6]. These intertwined challenges of biofilm formation and efflux activity drive the emergence of multidrug‑resistant (MDR) and extensively drug‑resistant (XDR) strains. Combining metallic nanoparticles with standard antibiotics offers a promising strategy to counteract both biofilm integrity and efflux‑mediated drug extrusion, thereby revitalizing the antibacterial potency of existing drugs [7–8]. Metal nanoparticles bring several advantages: low cytotoxicity at appropriate doses, high surface‑area‑to‑volume ratio, and broad‑spectrum antibacterial action [12–14]. Importantly, synergistic use of nanoparticles and antibiotics can lower the required antibiotic concentration, reducing toxicity to human cells [15]. Research into the mechanistic basis of nanoparticle antibacterial action continues to evolve. A leading hypothesis posits that metallic nanoparticles directly inhibit efflux pumps. In 2010, Banoee et al. demonstrated that zinc oxide nanoparticles (ZnO‑NPs) increased the ciprofloxacin inhibition zone by 27 % against S. aureus and 22 % against E. coli by targeting the NorA pump [16]. In 2014, Padwal and colleagues showed that polyacrylic acid‑coated iron oxide (magnetite) nanoparticles (PAA‑MNP) synergized with rifampicin to achieve fourfold greater growth inhibition in Mycobacterium smegmatis, correlating with a threefold rise in intracellular rifampicin accumulation as measured by ethidium bromide transport assays [17]. Two primary mechanisms are proposed: Supporting the first mechanism, Christena et al. reported that copper nanoparticles (Cu‑NPs) inhibited the NorA pump, likely via direct interaction, while the release of Cu²⁺ ions also contributed to membrane potential disruption [9]. Chatterjee et al. observed a gradual loss of membrane potential in E. coli exposed to 3.0 and 7.5 µg ml⁻¹ Cu‑NPs over one hour, dropping from –185 mV to –105 mV and –75 mV, respectively [22]. Despite these insights, the exact inhibitory pathway remains to be fully elucidated. Biofilm resilience is amplified when the constituent bacteria are drug‑resistant. Multiple studies have shown that metallic nanoparticles can penetrate and dismantle biofilm matrices through diverse mechanisms [24–27]. Their superior penetration capability renders them especially effective against biofilm‑associated infections [28–30]. Combining nanoparticles with antibiotics presents a novel dual‑action approach: inhibit efflux, restore antibiotic efficacy, and directly disrupt biofilm structure. Gurunathan et al. reported that silver nanoparticles (Ag‑NPs) synergized with ampicillin, enhancing biofilm inhibition by 70 % in Gram‑negative and 55 % in Gram‑positive strains—compared to only 20 % inhibition by Ag‑NPs alone. Similarly, the Ag‑NP/vancomycin pair achieved 55 % and 75 % biofilm suppression in Gram‑negative and Gram‑positive bacteria, respectively [10]. Copper and zinc oxide nanoparticles also demonstrated heightened anti‑biofilm activity when combined with antibiotics, with copper showing superior efficacy—potentially due to Cu²⁺ ion release and increased bonding in the presence of 2 % glucose [9]. Coating nanoparticles with carbohydrates can further modulate cell interaction, uptake, and cytotoxicity [31]. Efflux pumps contribute to quorum‑sensing by exporting signaling molecules essential for biofilm development. Inhibiting these pumps may therefore disrupt quorum‑sensing pathways and reduce biofilm formation [23,32–39]. Although the precise link remains incompletely defined, evidence suggests that efflux inhibition diminishes the concentration of quorum‑sensing autoinducers outside bacterial cells, thereby impairing signal reception and biofilm maturation [40–46]. Nanoparticles (yellow) block the efflux pump (cylinder), preventing extrusion of quorum‑sensing molecules (red) and thereby hindering biofilm formation. Efflux pumps also expel toxic metabolic by‑products that accumulate during rapid biofilm metabolism. Blocking these transporters can lead to intracellular accumulation of harmful compounds, further stalling biofilm development [49–50]. Nanoparticles (yellow) obstruct the efflux pump (cylinder), preventing extrusion of toxic by‑products (red) and thereby impeding biofilm formation. Comprehensive reviews summarizing the synergy of nanoparticles with antibiotics in anti‑biofilm and efflux inhibition contexts are compiled in Table 1. Efflux pumps serve a dual role in bacterial survival: expelling antibiotics and exporting quorum‑sensing molecules that foster biofilm formation. Nanoparticle‑mediated inhibition of these pumps can simultaneously enhance antibiotic retention within bacterial cells and reduce biofilm virulence. This strategy leverages existing antibiotics, reduces required dosages, and mitigates associated toxicity, offering a cost‑effective countermeasure against MDR and XDR pathogens. Targeted nanoparticle design—e.g., conjugation with anti‑efflux antibodies or lectins—may further improve specificity and minimize off‑target effects. As bacterial evolution continues to outpace traditional antibiotic development, exploiting metallic nanoparticles as efflux‑pump inhibitors presents a promising frontier. While the precise mechanisms—particularly PMF disruption versus direct transporter binding—require further investigation, preliminary data support both pathways. Overcoming challenges such as nanoparticle reactivity with non‑target membrane proteins and cytotoxicity at high surface areas will be critical. Tailored delivery systems and optimized dosing regimens will enable clinical translation, potentially restoring the efficacy of existing antibiotics and curbing the spread of MDR infections.Abstract
Review
Nanoparticles as Efflux Pump Inhibitors
Nanoparticles as Anti‑Biofilm Agent
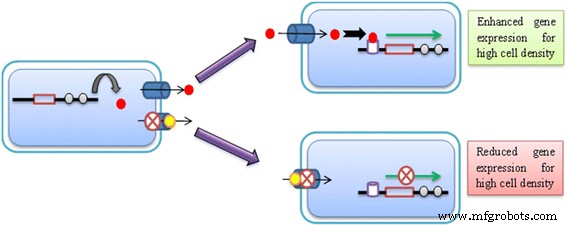
The Alliance Between Efflux Systems and Quorum Sensing
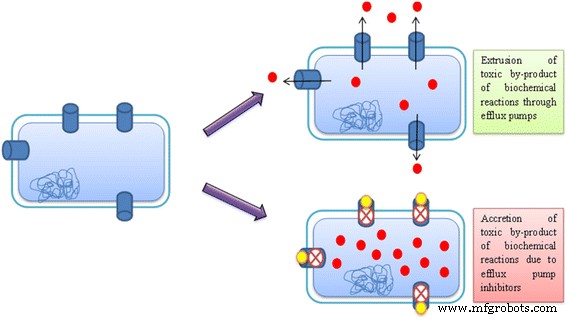
Conclusions
Future Prospects
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Al₂O₃ and SiO₂ Nanoparticles with Ultrasound Significantly Reduce Water Supercooling
- PEG‑PCCL Nanoparticles: Low‑Toxicity, Sustained Paclitaxel Delivery and Enhanced Anti‑Tumor Efficacy in Hepatocarcinoma Models
- Synergistic Antioxidant and Anti‑Amyloid Action of Chlorogenic Acid–Selenium Nanoparticles in Vitro
- Green‑Synthesized Nickel Oxide Nanoparticles from Ginger and Garlic Show Strong Antibacterial & Catalytic Performance
- hGC33-Targeted Sorafenib Nanoparticles Synergistically Inhibit Hepatoma via Wnt Pathway Suppression



