PEG‑PCCL Nanoparticles: Low‑Toxicity, Sustained Paclitaxel Delivery and Enhanced Anti‑Tumor Efficacy in Hepatocarcinoma Models
Abstract
Conventional chemotherapy often fails to achieve therapeutic concentrations at tumor sites because of physiological barriers, leading to suboptimal efficacy and systemic toxicity. Nanocarriers that encapsulate hydrophobic drugs can overcome these hurdles by improving drug solubility, prolonging circulation time, and enhancing tumor accumulation via the enhanced permeability and retention (EPR) effect. In this study we evaluate the systemic safety of a novel amphiphilic diblock copolymer, poly(ethylene glycol) carboxyl‑poly(ε‑caprolactone) (PEG‑PCCL), and its ability to deliver paclitaxel (PTX). PEG‑PCCL nanospheres were characterized by dynamic light scattering and transmission electron microscopy, revealing an average diameter of 97 ± 2.6 nm and a negative zeta potential of –18.4 mV. In vitro cytotoxicity assays (MTT and LDH leakage) demonstrated negligible toxicity to HEK293T and Hep‑G2 cells even at 1 mg/mL, while hemolysis and phlebitis studies in mice and rabbits confirmed excellent hemocompatibility and minimal vascular irritation. The encapsulation efficiency of PTX in PEG‑PCCL was 55.98 %. Pharmacokinetic analysis in H22‑bearing mice showed that PEG‑PCCL/PTX maintained a higher plasma concentration (Cmax = 283 ± 2.79 µg/mL) and a significantly prolonged mean residence time (Tmax = 4 ± 1.22 h) compared with free PTX (Cmax = 312 ± 2.59 µg/mL, Tmax = 0.54 ± 0.20 h). In vivo, PEG‑PCCL/PTX reduced ascites volume and extended survival of tumor‑bearing mice more effectively than free PTX. Collectively, PEG‑PCCL nanospheres exhibit excellent biocompatibility and represent a promising vehicle for hydrophobic anticancer agents.
Introduction
With the global rise in cancer incidence and an aging population, the therapeutic window of conventional chemotherapies remains narrow due to limited tumor uptake and severe off‑target toxicity, such as neutropenia and cardiomyopathy. Nanoparticle‑mediated drug delivery offers a rational solution: by engineering size, surface chemistry, and hydrophilicity, these carriers can selectively accumulate in tumors while evading the reticuloendothelial system. Among FDA‑approved polymers, PEG and poly(ε‑caprolactone) (PCL) form core‑shell structures that combine a hydrophobic drug‑laden core with a hydrophilic, stealthy shell. However, many such polymers still face challenges—high clearance rates, premature drug leakage, and residual toxicity. PEG‑PCCL is a recently developed diblock copolymer that incorporates carboxyl groups into the PCL backbone, enhancing hydrophilicity and stability through intramolecular hydrogen bonding. While its physicochemical profile has been documented, systematic toxicity evaluation and therapeutic performance remain underexplored. This study addresses that gap by assessing PEG‑PCCL’s acute in vitro and in vivo toxicity and by evaluating its capacity to improve paclitaxel pharmacokinetics and anti‑tumor efficacy in an H22 hepatocarcinoma model.
Methods
Materials, Cells, and Animals
All reagents were of analytical grade. Male Balb/C mice (7–8 weeks, 20–25 g) and New Zealand rabbits (2.5–3.0 kg) were maintained under SPF conditions and used in accordance with institutional ethical guidelines. Hep‑G2, HEK293T, and H22 hepatocarcinoma cells were cultured in DMEM supplemented with 10 % FCS and antibiotics at 37 °C, 5 % CO₂.
Preparation of PEG‑PCCL and PTX‑Loaded Nanoparticles
PEG‑PCCL was synthesized by ring‑opening polymerization of ε‑caprolactone (ε‑CL) in the presence of PEG (Mn = 1000) and a palladium catalyst, as illustrated in Fig. 1. After purification, the polymer was lyophilized and stored in airtight bags. PTX loading was achieved via a solvent‑free solid dispersion method: PTX was co‑evaporated with PEG‑PCCL, then re‑dispersed in water at 65 °C to form a homogeneous nanoparticle suspension, which was filtered (0.22 µm) and freeze‑dried. Encapsulation efficiency (EE) was calculated by HPLC using the minicolumn centrifugation method.
Characterization
Dynamic light scattering (Malvern Nano‑ZS 90) measured size and zeta potential. Transmission electron microscopy (TEM, Hitachi H‑6009IV) visualized morphology after phosphotungstic acid staining.
Cytotoxicity Assays
MTT and LDH leakage assays were performed on Hep‑G2 and HEK293T cells exposed to 0–1 mg/mL PEG‑PCCL for 24 h. Survival was expressed as a percentage relative to untreated controls. Annexin V/PI staining and fluorescence microscopy assessed apoptosis.
Hemocompatibility Assays
RBC hemolysis was measured in vitro after incubating 2 % RBC suspension with PEG‑PCCL (0.5 mg/mL) for 1–3 h at 37 °C. In vivo hemolysis was quantified by counting circulating RBCs after 3 h of intravenous injection (20 mg/kg).
Rabbit Phlebitis
Intravenous infusion of PEG‑PCCL (0.5 mg/mL) into auricular veins of rabbits was followed by histological evaluation of venous walls 24 h post‑injection.
Hepatorenal Function Tests
Serum AST, ALT, ALP, bilirubin, creatinine, uric acid, and albumin were measured 7 days post‑injection (20 mg/kg) to assess liver and kidney function.
Histopathology
H&E staining of lung, liver, and kidney tissues collected 3 days post‑IV injection examined for inflammatory changes.
Blood Concentration
PTX plasma levels were quantified by UV spectrophotometry at 760 nm from serial blood samples (0.08–24 h) after IV administration of free PTX (10 mg/kg) or PEG‑PCCL/PTX (20 mg/kg).
Tumor Model and Treatment
H22 cells (4 × 10⁶) were intraperitoneally injected into Balb/C mice. Once ascites formed (days 3–5), mice received either saline, free PTX (10 mg/kg), or PEG‑PCCL/PTX (20 mg/kg) on days 3 and 10. Abdominal perimeter (AP), ascites volume, and survival were monitored up to day 20.
Statistical Analysis
Data were expressed as mean ± SD. Statistical significance was determined by ANOVA with Dunnett’s post‑hoc test; p < 0.05 was considered significant.
Results
Morphology, Diameter, and Zeta Potential of PEG‑PCCL
Dynamic light scattering revealed an average diameter of 97 ± 2.6 nm. TEM images (Fig. 2) confirmed a uniform, spherical morphology. The measured zeta potential was –18.4 mV, indicative of a stable, negatively charged surface. Encapsulation efficiency of PTX was 55.98 %.

The characteristic of PEG‑PCCL: TEM image. Scale bars were 100 nm
Cytotoxicity
Both HEK293T and Hep‑G2 cells displayed concentration‑dependent viability reductions when exposed to PEG‑PCCL, yet the decline was significantly milder than that observed with PEI, particularly at 0.75 and 1 mg/mL (p = 0.023). LDH leakage mirrored these findings, with 19 % and 42 % cell death at 24 and 48 h, respectively, at 0.5 mg/mL PEG‑PCCL—substantially lower than PEI (p = 0.037). Annexin V/PI staining confirmed early apoptosis in both cell lines, but PEI induced a higher proportion of apoptotic cells compared to PEG‑PCCL (Fig. 5).
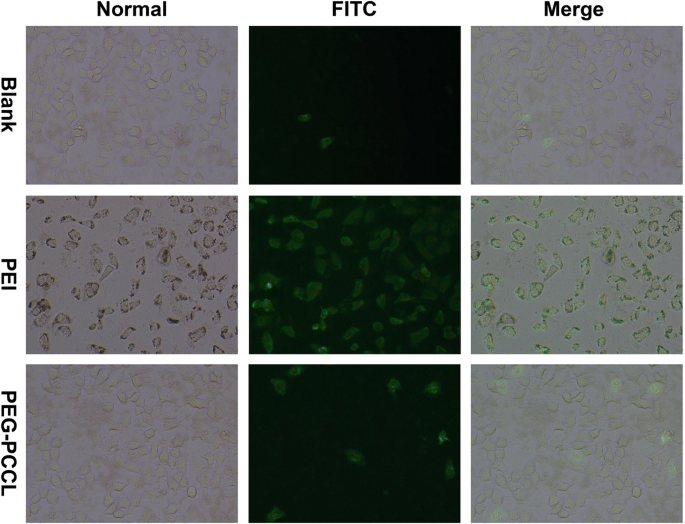
FITC‑Annexin V stain represented cell apoptosis. HepG2 cells incubated with nanoparticles at a concentration of 0.5 mg/mL for 48 h and then co‑stained with propidium iodide (red) and annexin V (green) were imaged at × 40
Biocompatibility
Hemolysis
In vitro hemolysis assays revealed a time‑dependent increase in hemolysis for all samples, yet PEG‑PCCL consistently displayed lower hemolysis than saline and dramatically less than PEI (Fig. 6a). In vivo, RBC counts after 3 h of intravenous injection mirrored these results, confirming excellent hemocompatibility (Fig. 6b).
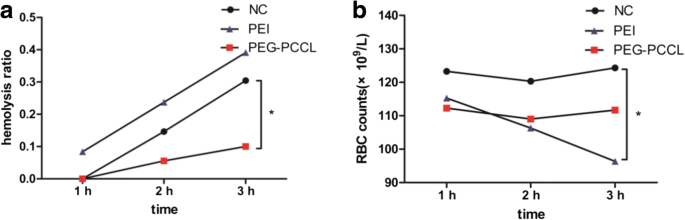
Hemolysis ratio and cell count (× 10⁹ L⁻¹) of blood sample in 3 h. In vitro (a) in vivo (b) *p < 0.05 versus negative control group
Rabbit Phlebitis
Histological examination of auricular veins after 24 h infusion showed minimal endothelial damage and inflammatory infiltration in the PEG‑PCCL group compared with saline (Fig. 7). No statistically significant difference in phlebitic scores was observed (p > 0.05).
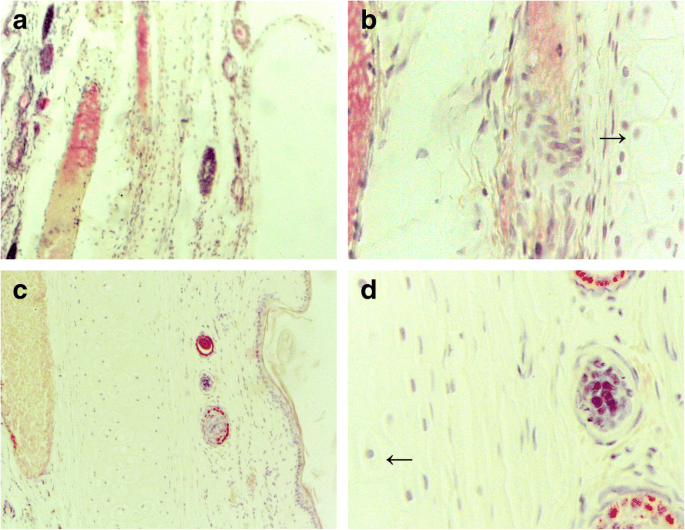
Photomicrographs of the ear vein after infusion of 24 h stained by H&E. a, b Group of infusion with normal saline. c, d Group of infusion with PEG‑PCCL. The needles represent the auricular cartilage. (Left image × 10, right image × 40)
Organ Toxicity
H&E staining of lung, liver, and kidney tissues collected 3 days post‑IV injection revealed no overt histopathological lesions in the PEG‑PCCL group, in contrast to mild inflammation observed with PEI (Fig. 8). Serum biochemistry confirmed no significant differences in AST, ALT, ALP, bilirubin, creatinine, uric acid, or albumin between PEG‑PCCL and saline groups (Table 2).
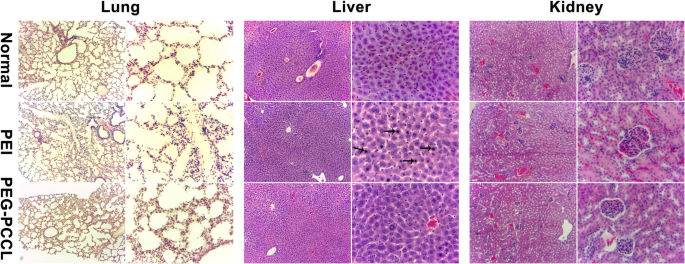
The H&E dyeing light microscopic images of the lung, liver, and kidney. The images are collected from mice intravenously administrated with NS, PEI, and PEG‑PCCL. The needles represent the karyopyknosis of hepatocyte. (Images of left columns, × 10; images of right columns × 40)
Pharmacokinetic Study
After IV administration of free PTX (10 mg/kg) or PEG‑PCCL/PTX (20 mg/kg), the plasma concentration‑time profile of PEG‑PCCL/PTX displayed a markedly prolonged Tmax (4 ± 1.22 h) versus free PTX (0.54 ± 0.20 h). The area under the curve (AUC₀–∞) for PEG‑PCCL/PTX was 282.21 ± 21.08 µg h/mL, roughly 5.4 times higher than that of free PTX (52.00 ± 4.30 µg h/mL), indicating enhanced systemic exposure (Fig. 9).
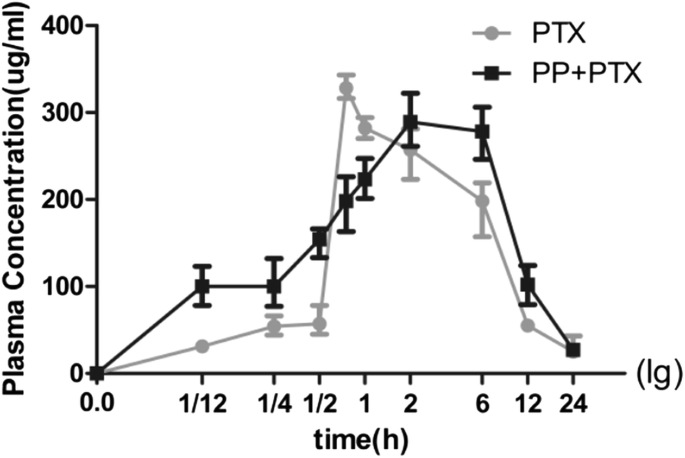
The blood concentration–time curve. In mice after intravenously administered with PTX or PP+PTX. (n = 6)
Effect of PEG‑PCCL/PTX on Tumor‑Bearing Mice
Daily measurement of abdominal perimeter (AP) showed that PEG‑PCCL/PTX and free PTX both slowed ascites accumulation relative to saline (Fig. 10a). At day 10, ascites volume was significantly lower in the PEG‑PCCL/PTX group (p = 0.0052) than in the free PTX group (p = 0.0005), with the PEG‑PCCL/PTX group exhibiting the smallest volume (p = 0.0138). Survival analysis revealed that mice receiving PEG‑PCCL/PTX lived longer and displayed a higher survival rate compared with both free PTX and saline controls (Fig. 10c).
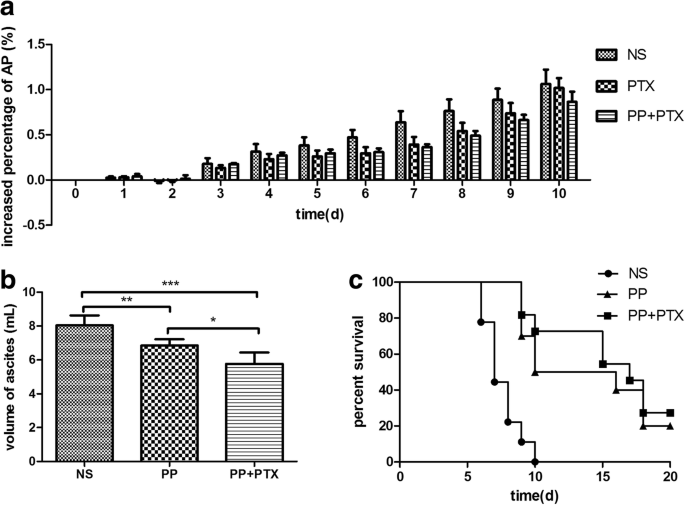
The anti‑tumor effect of PP/PTX in H22 tumor‑bearing mice. a Balb/C mice (n = 5) were intraperitoneally injected with PP/PTX or PTX at day 3. b Ascites of each group (n = 5) were collected at day 10. c The survival of each group (n = 10) was observed daily. *p < 0.05, **p < 0.01, ***p < 0.001
Discussion
The data presented demonstrate that PEG‑PCCL nanospheres are a safe and efficient platform for delivering hydrophobic anticancer drugs. Their negative surface charge and sub‑100 nm size confer stability in circulation and reduced interaction with the reticuloendothelial system, while the carboxylated PCL core ensures robust drug encapsulation without compromising biocompatibility. In vitro assays confirm minimal cytotoxicity and negligible hemolytic activity, positioning PEG‑PCCL ahead of cationic polymers such as PEI, which are notorious for endothelial irritation and systemic toxicity.
In vivo, the extended pharmacokinetic profile of PEG‑PCCL/PTX translated into superior tumor control in the H22 model: ascites formation was markedly suppressed and overall survival was improved compared with free PTX. These findings are consistent with the EPR effect and the prolonged systemic exposure achieved by the nanocarrier. While the encapsulation efficiency of ~56 % is satisfactory, future work could optimize the PEG:PCL ratio or introduce targeting ligands (e.g., folate) to further enhance tumor uptake and reduce off‑target release.
Importantly, the comprehensive toxicity panel—comprising hemolysis, phlebitis, organ histology, and serum biochemistry—indicates no clinically relevant adverse effects, underscoring PEG‑PCCL’s suitability for clinical translation. Future studies should explore biodistribution, long‑term safety, and combinatorial strategies (e.g., magnetic guidance or co‑delivery of immunotherapeutics) to fully harness the therapeutic potential of this platform.
Conclusions
PEG‑PCCL nanospheres exhibit a favorable safety profile, with negligible cytotoxicity, hemolysis, and organ toxicity, while enabling sustained release and enhanced anti‑tumor activity of paclitaxel in a hepatocarcinoma mouse model. These results establish PEG‑PCCL as a promising biocompatible carrier for hydrophobic anticancer agents.
Nanomaterials
- Enhancing On-Chip Optical Isolation with Coin Paradox Spin–Orbit Interaction for Superior Magneto-Optical Performance
- Metallic Nanoparticles: Potent Efflux Pump and Biofilm Inhibitors to Restore Antibiotic Efficacy
- Dose‑Dependent Liver, Brain, and Embryonic Toxicity of Titanium Dioxide Nanoparticles in Mice
- PEG-CoFe₂O₄ Nanoparticles: Assessing Toxicity and Curcumin’s Protective Effect
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Gold Nanoparticles Modulate CYP3A4 Activity and Cellular Toxicity in Human Hepatocellular Carcinoma: Influence of Size, Surface Chemistry, and Protein Corona
- Cellular Uptake and Intracellular Trafficking of Nanoparticles: Mechanisms, Physicochemical Drivers, and Clinical Implications
- Comparative Toxicity of PEG-Coated Cobalt Ferrite Nanoparticles and Nanospheres
- In‑Situ Synthesis of Silver Nanoparticles on Amino‑Grafted Polyacrylonitrile Fibers for Enhanced Antibacterial Performance



