Gold Nanoparticles Modulate CYP3A4 Activity and Cellular Toxicity in Human Hepatocellular Carcinoma: Influence of Size, Surface Chemistry, and Protein Corona
Abstract
We investigated how 40‑ and 80‑nm gold nanoparticles (AuNP) functionalized with cationic branched polyethylenimine (BPEI), anionic lipoic acid (LA), or neutral polyethylene glycol (PEG) interact with the human hepatocellular carcinoma (HCC) cell line C3A, both in the presence and absence of a human plasma protein corona (PC). Bare AuNP, except 80‑nm LA‑AuNP, were cytotoxic to C3A cells, but the PC significantly mitigated these effects. Time‑dependent cellular uptake increased for 40‑nm BPEI‑AuNP, whereas PC reduced uptake of 80‑nm PEG‑AuNP. BPEI‑AuNP elicited biphasic oxidative/nitrosative stress responses, while PEG‑AuNP acted as a potent antioxidant. All bare AuNP inhibited CYP3A4 activity regardless of size or surface charge, but PC restored activity except for PEG‑AuNP. The 40‑nm PEG‑AuNP induced gene expression changes primarily affecting mitochondrial fatty‑acid β‑oxidation and, to a lesser extent, hepatic efflux/uptake transporters. These findings deepen our understanding of AuNP‑cell interactions and their molecular underpinnings in HCC, offering insights for improved therapeutic strategies.
Background
Hepatocellular carcinoma (HCC) remains one of the most prevalent cancers worldwide and the fastest‑growing cause of cancer mortality in the United States [1, 2]. Current curative options—liver transplantation or surgical resection—are limited to early‑stage disease, while advanced tumors rely on chemotherapy or radiotherapy, both of which suffer from low selectivity and significant systemic toxicity [3, 4]. Gene‑silencing therapies, particularly RNA interference (RNAi), promise higher specificity, but efficient delivery requires vectors that penetrate target cells safely. While viral vectors offer high transduction efficiency, non‑viral platforms such as nanoparticles (NP) are preferred for their safety profile and tunable physicochemical properties [5–8]. Gold nanoparticles (AuNP) are especially attractive due to their facile surface modification and optical features, enabling targeted delivery of therapeutic cargos [11, 12]. However, upon entering the bloodstream, AuNP acquire a protein corona (PC) that alters their surface chemistry and consequently their biological fate, including cellular uptake and toxicity [13–17]. Understanding how NP size, surface charge, and the PC modulate cellular responses in HCC cells is essential for designing safe and effective nanomedicines.
Methods
Gold Nanoparticle Synthesis
40‑ and 80‑nm AuNP were custom fabricated by nanoComposix (San Diego, CA). Functionalization with BPEI, LA, or PEG was achieved via EDC chemistry, dihydrolipoic acid conjugation, or thiol‑methoxy PEGylation, respectively. Size, polydispersity index (PDI), and zeta potential were confirmed by dynamic light scattering (DLS), transmission electron microscopy (TEM), and UV‑Vis spectroscopy. The AuNP were purified by tangential flow filtration (TFF) and sterile‑filtered.
Protein Corona Preparation
Human plasma (n=5) from the Biological Specialty Corp. (Colmar, PA) was mixed with AuNP at 55 % (v/v) and incubated at 37 °C, 250 rpm for 1 h. Weakly bound proteins were removed by repeated PBS washes (20 000 × g, 20 min). The resulting PC‑coated AuNP were resuspended in PBS and then diluted into cell culture medium.
Physicochemical Characterization of AuNP
Hydrodynamic diameters (DH), PDI, and zeta potential of bare and PC‑coated AuNP were measured in DI water, PBS, and complete EMEM (10 % FBS) at 25 °C and 37 °C (0 and 24 h). Optical absorption spectra were recorded at room temperature.
Transmission Electron Microscopy
AuNP morphology was assessed by TEM (Tecnai G2 Spirit BioTWIN, 120 kV). Samples were dropped onto 200‑mesh copper grids and air‑dried.
Cell Culture and Viability Measurement
C3A cells (ATCC CRL‑10741) were cultured in EMEM + 10 % FBS, seeded at 8 × 10^4 cells/well (96‑well plates), and allowed to attach for 48 h before dosing. Viability was measured using the alamarBlue® assay after 24 h of exposure to AuNP concentrations ranging from 0 to 250 µg/cm^2.
Cellular Uptake Measurement by ICP‑MS
Cells were treated with 1.56 µg/cm^2 of AuNP for 0.5–24 h. After an etching step to remove surface‑bound particles, cells were digested in aqua regia, and intracellular Au was quantified by ICP‑MS. Uptake was expressed as AuNP per cell.
Oxidative/Nitrosative Stress Measurements
ROS/RNS production was quantified using a total ROS/SO assay kit (Enzo Life Sciences) at 1, 3, and 24 h following exposure to 40‑nm BPEI‑ and PEG‑AuNP up to 125 µg/cm^2.
Cytochrome P450 3A4 Activity
CYP3A4 activity was assessed with the P450‑Glo™ assay (Promega) after 24 h exposure to LC_50 concentrations of each AuNP type. Luminescence was normalized to vehicle controls.
Gene Expression Profiling
Given the pronounced effects of 40‑nm PEG‑AuNP, C3A cells were exposed to its LC_50 (129.5 µg/cm^2) for 24 h. Total RNA (RIN ≥ 7.8) was isolated, reverse‑transcribed, and analyzed by the Human Molecular Toxicology Pathway Finder and Human Drug Transporters RT^2 Profiler™ PCR arrays (Qiagen). Differentially expressed genes (|fold change| > 2, p < 0.05) were validated by qRT‑PCR for nine representative genes.
Statistical Analysis
LC_50 values were fitted using a variable‑slope Hill equation in GraphPad Prism. One‑way ANOVA (SAS 9.4) followed by Tukey’s HSD test assessed significance (p < 0.05).
Results and Discussion
Physicochemical Characterization of Bare and PC‑Coated AuNP
As shown in Figure 1, TEM confirmed monodisperse AuNP with sizes matching the nominal 40 and 80 nm diameters. PC formation induced a modest increase in DH and a red‑shift in the UV‑Vis plasmon peak, indicating protein adsorption. In complete EMEM, DH and PDI remained stable for 24 h, except for PC‑BPEI‑AuNP, which displayed a transient decrease in PDI, suggesting enhanced colloidal stability. Zeta potentials shifted toward neutral values upon PC coating, reflecting protein masking of surface charges.
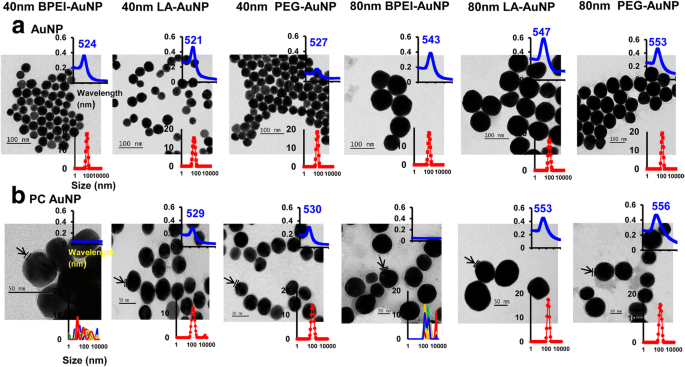
Transmission electron micrographs of (a) bare AuNP in DI water and (b) PC‑AuNP in PBS at 0 h, 25 °C. UV‑Vis spectra and DLS distributions are inset. Arrows indicate PC formation. PC: human plasma protein corona; BPEI: branched polyethylenimine; LA: lipoic acid; PEG: polyethylene glycol.
AuNP Cytotoxicity
LC_50 values for bare AuNP ranged from 127.3 to 205.5 µg/cm^2 (Figure 2a). All 40‑nm BPEI‑, LA‑, and PEG‑AuNP, as well as 80‑nm BPEI‑AuNP, were cytotoxic, whereas 80‑nm LA‑AuNP showed no significant toxicity. PC markedly reduced cytotoxicity across most AuNP types, with the exception of 80‑nm BPEI‑AuNP, which retained 51 % viability at 250 µg/cm^2 (Figure 2b). These findings align with prior work showing PC‑mediated attenuation of AuNP toxicity in primary hepatocytes and other cell lines [7–9].
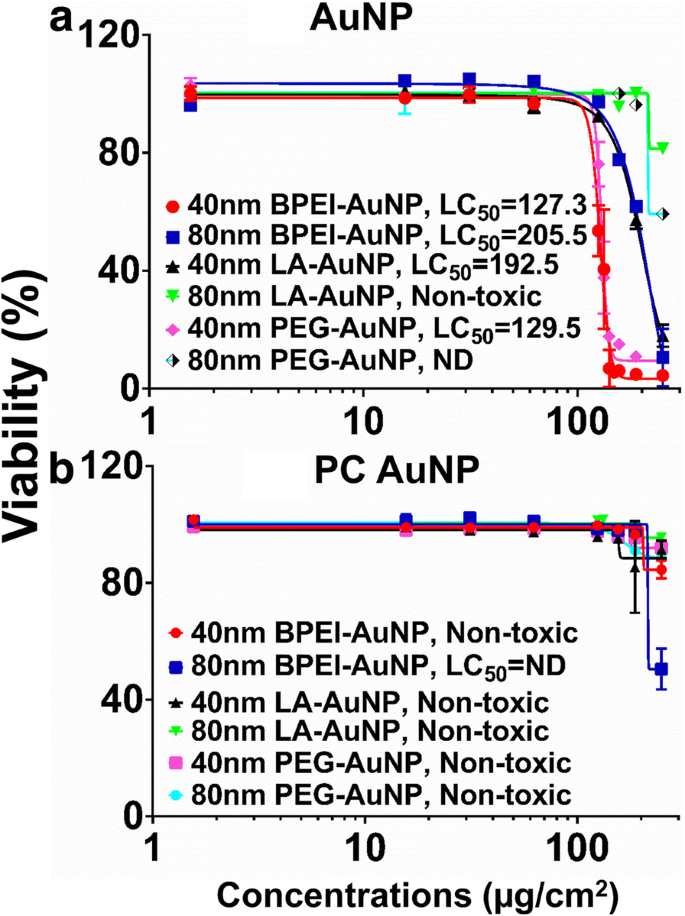
C3A viability and LC_50 values for (a) bare and (b) PC‑coated AuNP. Data represent mean ± S.D. (n = 3). PC: human plasma protein corona; ND: not determined; BPEI: branched polyethylenimine; LA: lipoic acid; PEG: polyethylene glycol; LC_50: median lethal concentration.
Intracellular Uptake of AuNP
Time‑dependent uptake analyses (Figure 3) revealed that 40‑nm BPEI‑AuNP accumulated rapidly, peaking at 6 h before declining. In contrast, PC attenuated uptake of 40‑ and 80‑nm BPEI‑ and LA‑AuNP but enhanced 80‑nm PEG‑AuNP internalization. These trends are consistent with literature indicating that PC can either hinder or promote NP entry depending on NP size, surface chemistry, and cell type [8, 9, 33].
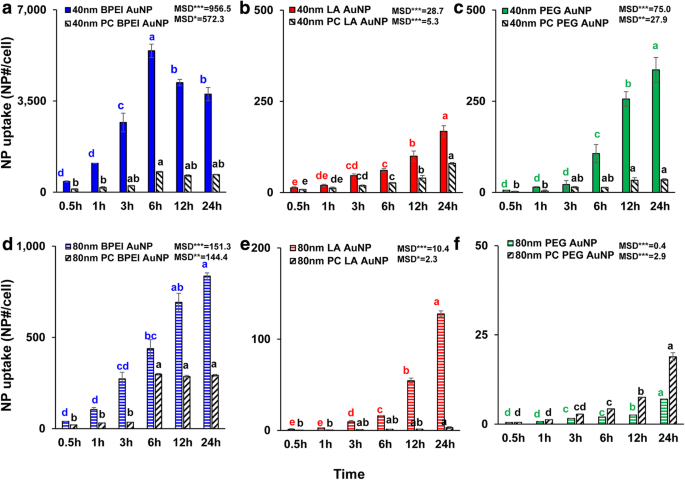
Time‑dependent cellular uptake of 40‑nm (a) BPEI‑, (b) LA‑, and (c) PEG‑AuNP, and 80‑nm (d) BPEI‑, (e) LA‑, and (f) PEG‑AuNP in the absence and presence of PC. Data represent mean ± S.D. (n = 3). Letters indicate significant differences (Tukey’s HSD, p < 0.05). PC: human plasma protein corona.
Oxidative and Nitrosative Stress Measurements
Both AuNP types induced ROS/RNS in a time‑ and concentration‑dependent manner (p < 0.0001). High concentrations of 40‑nm BPEI‑AuNP produced a biphasic response: an initial antioxidant effect at 1 h, followed by a pronounced pro‑oxidant surge at 3 h and beyond. In contrast, 40‑nm PEG‑AuNP consistently suppressed ROS/RNS production (fold change < 0.5) across all time points, suggesting a protective antioxidant role (Figure 4).
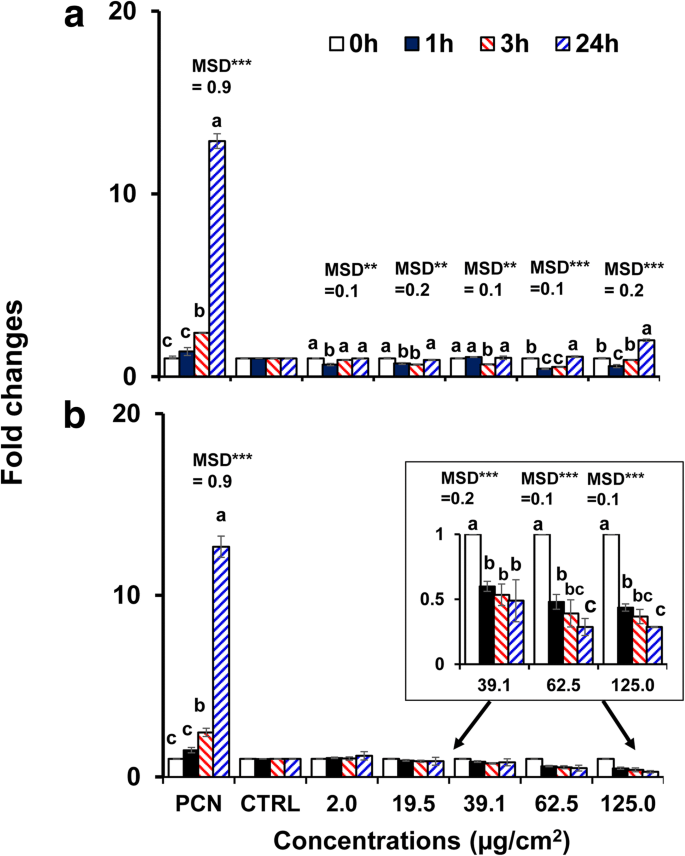
Time‑ and concentration‑dependent ROS/RNS production in C3A cells exposed to (a) 40‑nm BPEI‑AuNP and (b) 40‑nm PEG‑AuNP up to 24 h. Data represent mean ± S.D. (n = 3). Significant differences are marked by asterisks (Tukey’s HSD, p < 0.05).
CYP3A4 Activity Measurement
All bare AuNP (40‑ and 80‑nm BPEI, LA, and PEG) inhibited CYP3A4 activity to 20–31 % of control levels at LC_50 concentrations (Figure 5). Non‑toxic concentrations of 80‑nm LA‑ and PEG‑AuNP also suppressed activity (~30 %). PC largely restored CYP3A4 activity for all AuNP except for PEG‑AuNP, which retained 63 % and 46 % activity for 40‑ and 80‑nm particles, respectively. These results corroborate previous reports of AuNP‑mediated CYP3A4 inhibition via direct enzyme interaction or substrate pocket blocking [7, 25].
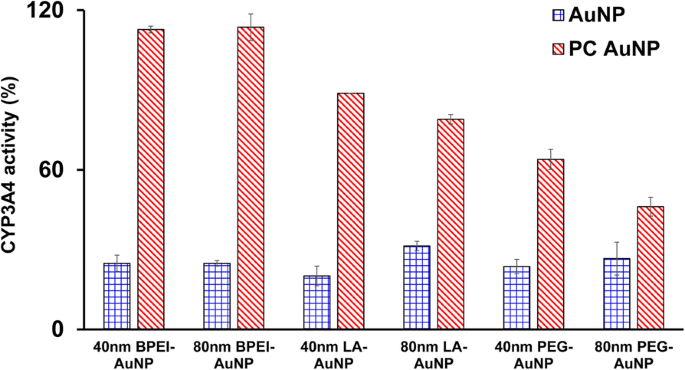
Inhibitory effect of AuNP on CYP3A4 activity in C3A cells after 24 h exposure to 40‑ and 80‑nm BPEI‑, LA‑, and PEG‑AuNP, with and without PC. Values represent mean ± S.D. (n = 3). PC: human plasma protein corona.
Toxicity Pathway–Focused Gene Expression Profiling of 40‑nm PEG‑AuNP
Gene array analysis identified 212 differentially expressed genes (186 down‑regulated, 26 up‑regulated) at the LC_50 of 40‑nm PEG‑AuNP (Figure 6). Notably, 12.3 % of these genes involved mitochondrial fatty‑acid β‑oxidation, with key enzymes (ACAD11, ACAD9, ACADM, ACADS, ACAA1, ACAA2, DECR1, ECHS1, EHHADH, HADHA) markedly suppressed (fold change 2–18). The down‑regulation of β‑oxidation enzymes indicates impaired NADH/FADH_2 production, compromising the TCA cycle and ATP synthesis, a hallmark of mitochondrial dysfunction observed in AuNP‑treated hepatocytes [7–9].
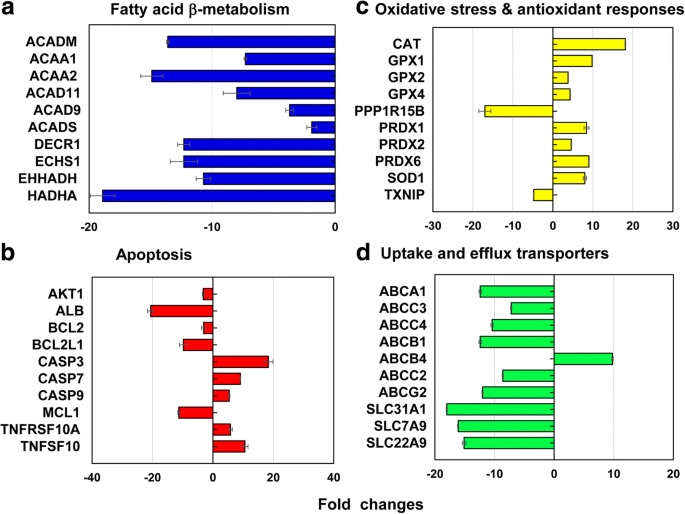
Representative genes involved in (a) mitochondrial fatty‑acid β‑oxidation, (b) apoptosis, (c) oxidative stress/antioxidant responses, and (d) hepatic uptake/efflux transporters at the LC_50 of 40‑nm PEG‑AuNP. Fold change < −2 or > 2, p < 0.05.
Apoptosis genes (CASP3, CASP7, CASP9, TNFRSF10A/B, TNFSF10) were up‑regulated, whereas anti‑apoptotic genes (AKT1, BCL2, MCL1, XIAP) were down‑regulated, consistent with the observed dose‑dependent cytotoxicity. DNA damage and repair checkpoints (CHEK1/2, ERCC1/2/3, LIG4) showed mixed regulation, suggesting impaired genomic integrity. Heat‑shock proteins (HSPA1A/B) were up‑regulated, while HSP40/90/60 subfamilies were down‑regulated, reflecting a complex stress response. Antioxidant genes (GPX1/2/4, PRDX1/2/6, SOD1, CAT) were induced, whereas pro‑oxidant genes (TXNIP, PPP1R15B) were suppressed, aligning with the antioxidant phenotype of PEG‑AuNP.
Drug Uptake and Efflux Transporter Gene Expression Profiling
PC‑induced modulation of drug transporters further underscores potential pharmacokinetic implications. Of the 35 transporter genes examined, 12 ABC transporters were down‑regulated (e.g., ABCB1, ABCC2/3/4, ABCG2/8, ABCA1) and 21 SLC transporters were suppressed (e.g., SLC31A1, SLC22A9). Conversely, MDR4 (ABCB4) and mitochondrial ABC transporter (ABCB6) were up‑regulated, indicating a compensatory response to nanoparticle exposure. These changes may influence the intracellular concentration of chemotherapeutics and warrant careful consideration in combination therapies.
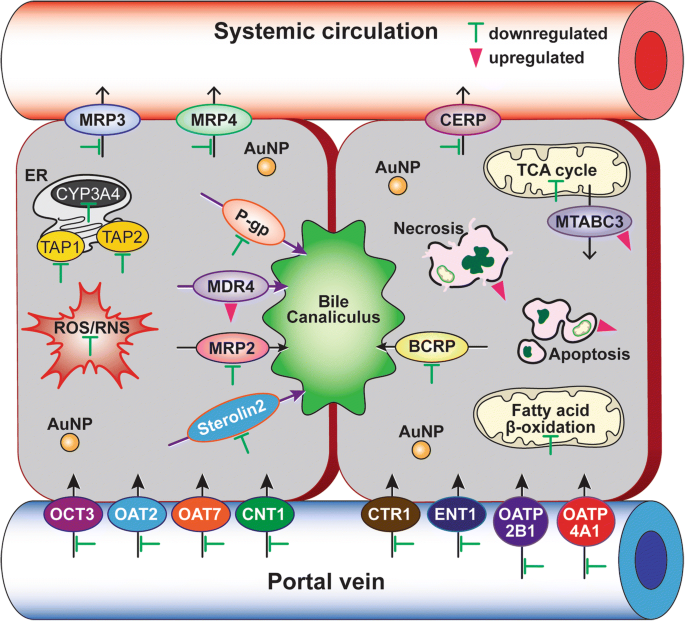
Schematic representation of the key mechanisms by which 40‑nm PEG‑AuNP modulate CYP3A4 activity, mitochondrial β‑oxidation, and drug transporter expression in HCC cells. Green bars (inhibition) and pink triangles (induction) indicate direction of change.
Real‑time PCR validation of nine selected genes confirmed the array results, reinforcing the reliability of the transcriptional data.
Conclusions
This study demonstrates that AuNP surface chemistry and the presence of a human plasma protein corona critically dictate their cytotoxicity, cellular uptake, oxidative stress profile, CYP3A4 inhibition, and gene expression changes in C3A HCC cells. Bare BPEI‑ and LA‑AuNP were highly cytotoxic, whereas PEG‑AuNP exhibited antioxidant properties but still suppressed CYP3A4 activity. The PC largely mitigated toxicity and CYP3A4 inhibition, except for PEG‑AuNP, which retained significant effects. Gene expression analysis of 40‑nm PEG‑AuNP revealed predominant down‑regulation of mitochondrial fatty‑acid β‑oxidation enzymes, up‑regulation of pro‑apoptotic genes, and extensive modulation of drug transporters, providing insight into potential mechanisms of hepatotoxicity and drug–nanoparticle interactions. These findings underscore the importance of considering protein corona formation and surface functionalization when designing AuNP‑based therapeutics for HCC.
Abbreviations
- ANOVA:
One-way analysis of variance
- AuNP:
Gold nanoparticles bare: no PC
- BPEI:
Branched polyethylenimine
- CYP:
Cytochrome P450
- D H:
Hydrodynamic diameters
- DLS:
Dynamic light scattering
- EDTA:
Ethylenediaminetetraacetic acid
- EMEM:
Eagle’s minimum essential medium
- HCC:
Human hepatocellular carcinoma
- HPTC:
Human renal proximal tubular cells
- HSD:
Tukey’s honest significant difference test
- HUVEC:
Human umbilical vein cells
- ICP-MS:
Inductively coupled plasma mass spectrometry
- LA:
Anionic lipoic acid
- LC50:
Median lethal concentration
- MDR:
Multidrug resistance
- NP:
Nanoparticles
- PBS:
Phosphate-buffered saline
- PC:
Human plasma protein corona
- PDI:
Polydispersity index
- PEG:
Neutral polyethylene glycol
- RNAi:
RNA interference
- RNS:
Reactive nitrogen species
- ROS:
Reactive oxygen species
- SO:
Superoxide
- TEM:
Transmission electron microscopy
- TFF:
Tangential flow filtration
Nanomaterials
- High‑Performance Au/Ce‑La Nanorod Catalysts for Low‑Temperature CO Oxidation: Synthesis, Characterization, and Mechanistic Insights
- Evaluating C60 Fullerene's Modulation of Diphenyl‑N‑(Trichloroacetyl)amidophosphate-DNA Binding and Cytotoxicity in Human Leukemia Cells
- PEG‑PCCL Nanoparticles: Low‑Toxicity, Sustained Paclitaxel Delivery and Enhanced Anti‑Tumor Efficacy in Hepatocarcinoma Models
- Gold Nanoparticles Modulate CYP3A4 Activity and Cellular Toxicity in Human Hepatocellular Carcinoma: Influence of Size, Surface Chemistry, and Protein Corona
- Neoglycoprotein‑Functionalized Fluorescent Gold Nanoclusters for Plant Lectin Detection and Dendritic Cell Imaging
- Cellular Uptake and Intracellular Trafficking of Nanoparticles: Mechanisms, Physicochemical Drivers, and Clinical Implications
- Shape-Dependent Cytotoxicity & Cellular Uptake of Green Tea‑Reduced Gold Nanoparticles in Cancer Cells
- Gold Nanoparticles Modulate Testosterone Metabolism in Human Liver Microsomes: Size, Surface Chemistry, and Protein Corona Effects
- In‑Situ Synthesis of Silver Nanoparticles on Amino‑Grafted Polyacrylonitrile Fibers for Enhanced Antibacterial Performance
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping



