Dual‑Mode MRI/Fluorescence Nanoprobe cRGD‑Gd‑Cy5.5 for Targeted Imaging of Integrin αvβ3‑Positive Tumors
Abstract
Malignant tumours remain a leading cause of mortality worldwide, and the ability to detect early disease and predict metastatic spread is critical for selecting the most effective treatment strategy. Integrin αvβ3 is a well‑validated biomarker of tumour angiogenesis, making it an attractive target for molecular imaging. Here we report the synthesis and in‑vitro evaluation of a liposome‑based dual‑mode probe, cRGD‑Gd‑Cy5.5, that couples T1-weighted magnetic resonance (MR) contrast with near‑infrared fluorescence. The nanoparticles are ~60 nm in diameter, exhibit excellent aqueous dispersion, a positive zeta potential (+39.5 mV), and an r1 relaxivity of 10.5 mM‑1 s‑1—over twice that of the clinical agent Magnevist. Cytotoxicity assays show >70 % cell viability at Gd concentrations up to 400 µM. Fluorescence and MR studies in A549 and SUNE‑1‑5‑8F cells demonstrate selective uptake in integrin‑αvβ3-rich cells, whereas normal MCF‑10A cells exhibit negligible binding. In vivo imaging in a nude‑mouse nasopharyngeal carcinoma model reveals tumour‑specific signal enhancement that persists for at least 6 h post‑injection. These findings establish cRGD‑Gd‑Cy5.5 as a safe, highly responsive dual‑modality probe for real‑time monitoring of tumour metastasis.
Background
Metastatic spread is the principal cause of treatment failure in solid tumours. A hallmark of this process is the remodeling of the extracellular matrix (ECM) and the formation of new vasculature, both of which are mediated by integrin αvβ3. This integrin is expressed at high levels on endothelial cells of tumour neovasculature and on many tumour cell types, yet is low or absent in normal tissues, making it an ideal imaging target.
Recent advances in molecular imaging have made it possible to non‑invasively interrogate tumour biology. Effective probes require a targeting moiety and a detectable signal. The RGD peptide sequence, which binds integrin αvβ3 with high affinity, has become the standard targeting ligand. Combining this with magnetic resonance, which offers high soft‑tissue contrast and deep penetration, and near‑infrared fluorescence, which provides high sensitivity for intra‑operative guidance, yields a powerful dual‑modality approach.
While Gd‑based T1 agents are widely used clinically, they lack inherent tumour specificity. By encapsulating Gd ions within liposomes and decorating the surface with RGD and a Cy5.5 fluorophore, we created a probe that not only improves relaxivity but also delivers a highly specific targeting capability.
Results
Characterization of the cRGD‑Gd‑Cy5.5 Nanoprobe
The probe appears as a pale‑pink, clear liquid with no precipitation after standing, indicating good colloidal stability. Transmission electron microscopy (TEM) shows uniformly sized, spherical liposomes (~60 nm) with no aggregation (Fig. 1a). Dynamic light scattering (DLS) gives a hydrodynamic diameter of 124.2 ± 0.215 nm and a positive zeta potential of 39.5 ± 1.65 mV, confirming surface amino‑group functionalisation (Fig. 1b,c).
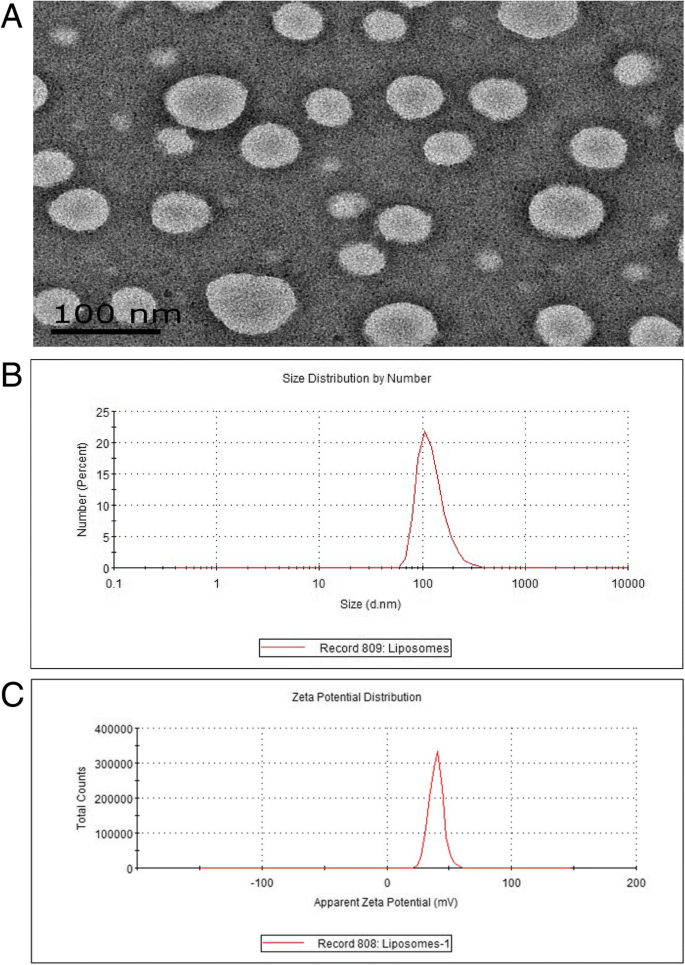
Characterization data of cRGD‑Gd‑Cy5.5. a Low‑magnification TEM images. b Size distribution by DLS (average 124.2 nm). c Zeta potential (≈39 mV).
Fluorescence spectroscopy confirms successful Cy5.5 conjugation, with a distinct emission peak at 685 nm upon 600‑nm excitation (Fig. 2a). In vivo small‑animal imaging demonstrates bright red fluorescence in liposome‑Cy5.5, whereas blank liposomes show no signal (Fig. 2b).
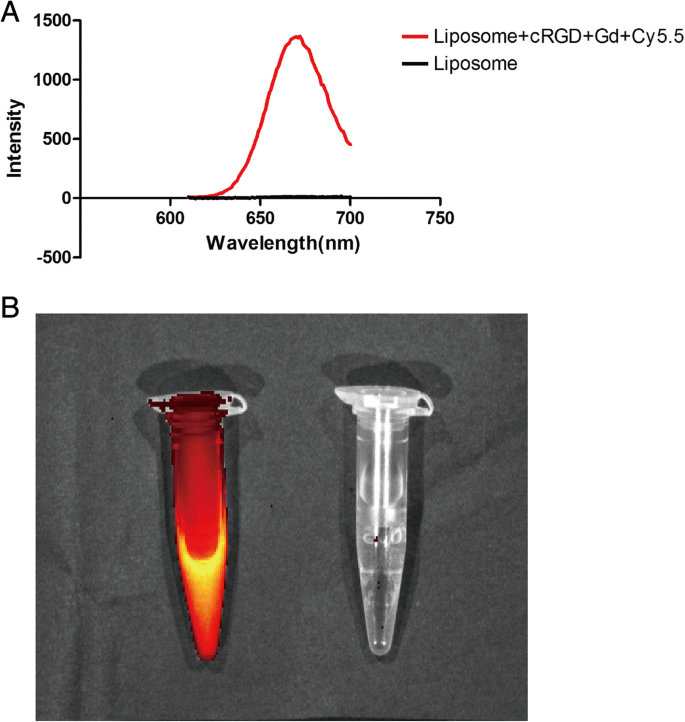
Fluorescence properties of cRGD‑Gd‑Cy5.5. a Emission at 670 nm under 600‑nm excitation. b Red fluorescence in Cy5.5‑labelled liposomes versus none in blanks.
MR Relaxometry of the cRGD‑Gd‑Cy5.5 Nanoprobe
Linear fitting of T1 relaxation times versus Gd concentration yields an r1 relaxivity of 10.515 mM‑1 s‑1, more than twice that of Magnevist (4.56 mM‑1 s‑1). The probe shows a dose‑dependent increase in signal intensity, confirming its suitability as a positive contrast agent (Fig. 3a,b).
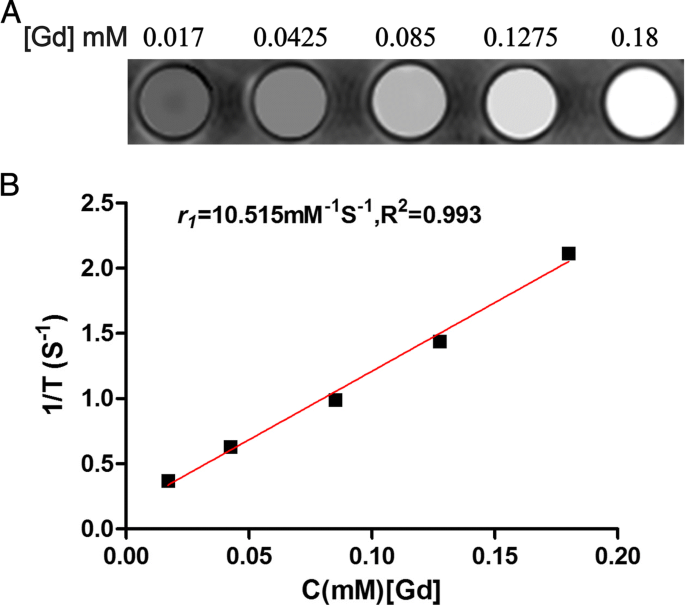
Relaxation properties. a T1-weighted images at varying Gd concentrations. b Linear fit yields r1 = 10.515 mM‑1s‑1.
Cytotoxicity Study of the cRGD‑Gd‑Cy5.5 Nanoprobe
CCK‑8 assays across A549, SUNE‑1‑5‑8F, HUVEC, and MCF‑10A cells show >70 % viability at Gd concentrations up to 400 µM after 24 h, indicating excellent biocompatibility (Fig. 4).
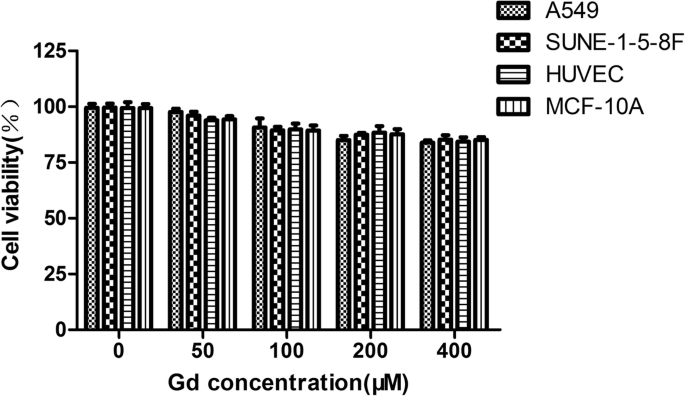
Cytotoxicity assay. Cell viability >70 % for all cell lines at 400 µM Gd.
Immunofluorescence Staining and Flow Cytometry Assay of Integrin αvβ3
Confocal imaging shows strong integrin αvβ3 staining on A549 and SUNE‑1‑5‑8F cell membranes, with negligible signal in MCF‑10A cells (Fig. 5a). Flow cytometry quantifies surface expression: A549 (89.07 %), SUNE‑1‑5‑8F (63.84 %), MCF‑10A (1.56 %).
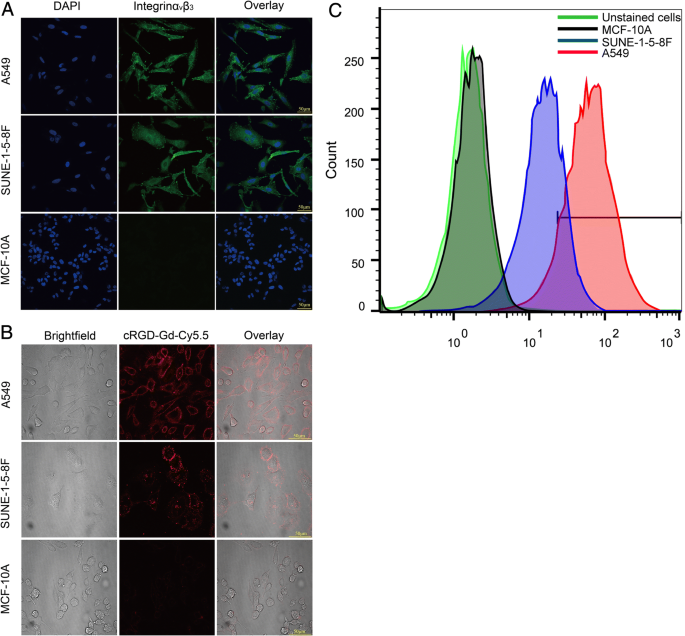
Integrin αvβ3 expression. a Confocal images. b Probe binding at 200 µg mL‑1. c Flow cytometry results.
Cellular Uptake of cRGD‑Gd‑Cy5.5
Red fluorescence is evident on the membranes of A549 and SUNE‑1‑5‑8F cells after 1 h incubation, with intensity proportional to integrin expression. No signal is observed in MCF‑10A cells, confirming target specificity.
In Vitro MR and Fluorescence Imaging
T1-weighted MR images become progressively brighter with increasing Gd concentrations in A549 cells, with a statistically significant difference (p < 0.05). Fluorescence imaging shows a corresponding increase in Cy5.5 signal, outperforming the non‑targeted Gd‑Cy5.5 control (Fig. 6).
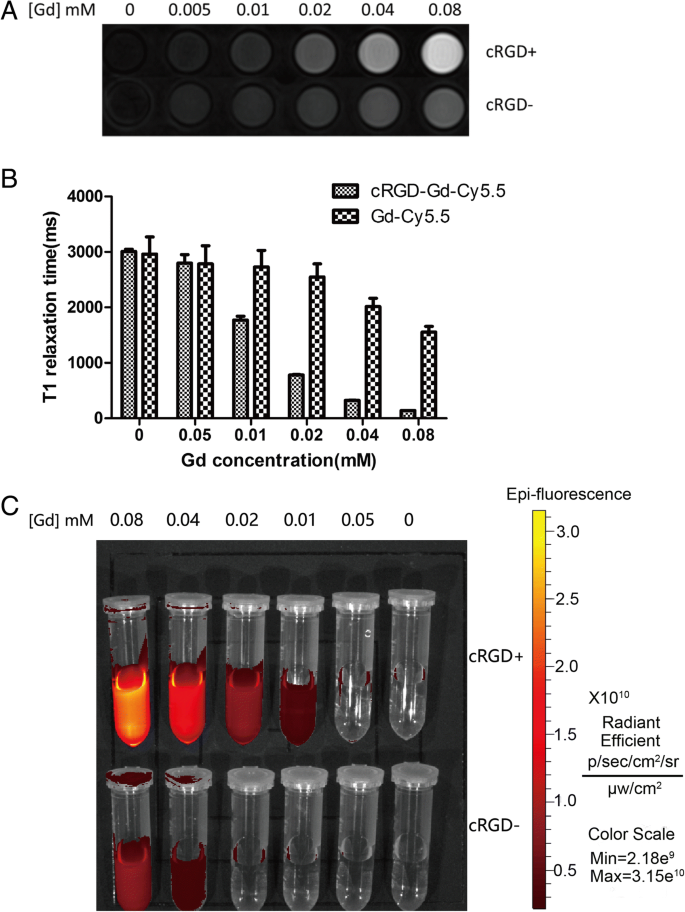
In vitro imaging. a Cells incubated with varying Gd (0–0.08 mM). b Corresponding T1 relaxation times. c Small‑animal fluorescence images.
In Vivo MR and Fluorescence Imaging
In nude mice bearing nasopharyngeal carcinoma xenografts, tumour contrast improves markedly 5 min post‑injection and remains stable up to 6 h. The receptor‑blocked group shows only marginal enhancement, confirming integrin‑mediated uptake. Fluorescence imaging mirrors the MR findings, with peak tumour signal at 6 h and renal excretion evident at 6 h (Fig. 7).
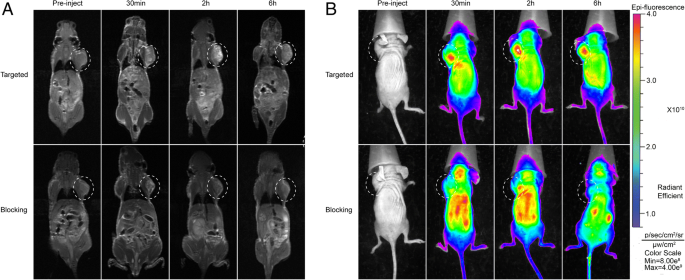
In vivo imaging. a 3.0‑T MR scans pre‑injection, 30 min, 2 h, 6 h. b Fluorescence images at corresponding times.
Discussion
Integrin αvβ3 is highly expressed on tumour vasculature and many malignant cell types, making it a compelling target for non‑invasive imaging. Traditional Gd chelates lack tumour specificity and offer limited relaxivity. By encapsulating Gd within liposomes and decorating the surface with a cyclic RGD peptide and Cy5.5, we achieved a dual‑mode probe that combines the high spatial resolution of T1 MR with the sensitivity of NIR fluorescence.
The probe’s r1 of 10.5 mM‑1 s‑1 surpasses clinical agents, while the positive surface charge and ~60 nm size favor efficient tumour accumulation and retention. Cytotoxicity studies confirm excellent safety, with viability above 70 % even at supra‑clinical Gd doses. Importantly, the probe’s uptake correlates strongly with integrin expression levels, as shown by immunofluorescence, flow cytometry, and in vitro imaging.
In vivo data demonstrate specific tumour targeting, sustained contrast for up to 6 h, and renal clearance, indicating a favourable pharmacokinetic profile. These attributes position cRGD‑Gd‑Cy5.5 as a promising platform for real‑time, multimodal monitoring of tumour progression and metastasis.
Conclusions
The liposomal dual‑mode probe cRGD‑Gd‑Cy5.5 is a robust, safe, and highly responsive tool for targeting integrin αvβ3. Its superior relaxivity, specific tumour binding, and low cytotoxicity provide a solid foundation for future clinical translation in the non‑invasive, real‑time imaging of tumour metastasis.
Methods
Synthesis of cRGD‑Gd‑Cy5.5
Liposomes were prepared by dissolving lecithin, cholesterol, and DSPE‑PEG2000‑NH2 in chloroform, followed by rotary evaporation to form a thin film. The film was hydrated with a GdCl3 solution (10 mg) in carbonate buffer (pH 8.5) at 50 °C, then sonicated and filtered (0.22 µm) before ultrafiltration (10 kDa) to remove free Gd. RGD peptide was activated with EDC/NHS and conjugated to Cy5.5, then coupled to the liposome surface at pH 8.4. The final formulation was stored at 4 °C.
Characterization of Nanoparticles
Particle size and morphology were assessed by TEM and DLS. Optical properties were measured with a microplate reader (400–800 nm absorption, 600‑nm excitation). FT‑IR and UV‑Vis spectra confirmed surface functionalisation.
Measurement of the Relaxation Rate
Serial dilutions of the probe were scanned on a 3.0‑T MR scanner using a MOLLI sequence (TR = 5.8 ms, TE = 3.66 ms). T1 values were extracted from 0.3 cm2 ROIs and plotted against Gd concentration to calculate r1.
Cell Culture and Cytotoxicity Assay
Human tumour and normal cell lines (A549, SUNE‑1‑5‑8F, HUVEC, MCF‑10A) were cultured in DMEM with 10 % FBS. CCK‑8 assays were performed at 50–400 µM Gd for 24 h to determine viability.
Immunofluorescence Staining and Flow Cytometry Assay of Integrin αvβ3
Cells were fixed, blocked, and incubated with anti‑αvβ3 antibody, followed by a fluorescent secondary antibody and DAPI staining. Confocal microscopy and flow cytometry (488 nm excitation) quantified surface expression.
Binding Assay of the Molecular Probe to Integrin αvβ3
Cells were incubated with the probe for 1 h, washed, and imaged by confocal microscopy to assess binding.
In Vitro MR Imaging and Fluorescence Imaging of Tumor Cells
A549 cells were incubated with varying Gd concentrations (0–0.08 mM) for 2 h, then washed, embedded in agarose, and imaged on a 3.0‑T MR scanner. Fluorescence imaging was performed on a small‑animal system.
In Vivo MR and Fluorescence Imaging
Balb/c nude mice bearing nasopharyngeal carcinoma xenografts received tail‑vein injections of 0.05 mmol Gd/kg cRGD‑Gd‑Cy5.5. MR and fluorescence imaging were performed pre‑injection, 30 min, 2 h, and 6 h post‑injection. A receptor‑blocked group received free RGD (10 mg) 1 h prior to probe injection.
Nanomaterials
- IBM Breaks New Ground: First Liquid‑Phase Atomic Imaging of 2D Materials at Room Temperature
- Scientists Successfully Synthesize and Visualize Cyclo[18]Carbon
- Effects of 15‑nm Gold Nanoparticles on Proliferation, Apoptosis, and Spheroid Formation in HT29 Colon Carcinoma and SPEV Embryonic Kidney Cells
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Ti0.91O2/CdS Hybrid Spheres: Synthesis, Structural Characterization, and Exceptional Indirect Optical Transition
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- Neoglycoprotein‑Functionalized Fluorescent Gold Nanoclusters for Plant Lectin Detection and Dendritic Cell Imaging
- Dual-Targeted Paramagnetic Liposomes for αvβ3 Integrin and NRP‑1: A Powerful MRI Tool for Early Tumor Detection
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy
- Eco-Friendly Microplasma Synthesis of Yellow-Emitting Carbon Quantum Dots for Cancer Cell Imaging and Photocatalytic Inactivation



