Dual-Targeted Paramagnetic Liposomes for αvβ3 Integrin and NRP‑1: A Powerful MRI Tool for Early Tumor Detection
Abstract
Magnetic resonance imaging (MRI) is indispensable for early tumor diagnosis, yet its inherent lack of specificity limits clinical impact. By harnessing angiogenesis as a molecular imaging target, contrast agents can be delivered directly to neovasculature via receptor‑specific carriers. We engineered a dual‑targeted paramagnetic liposome that displays two angiogenic ligands: the αvβ3‑integrin‑binding Arg‑Gly‑Asp (RGD) motif and the neuropilin‑1‑binding heptapeptide ATWLPPR (A7R). These liposomes, with diameters below 100 nm, encapsulated the gadolinium chelate Gd‑DTPA efficiently. Their T1 relaxivity matched that of free Gd‑DTPA, while cellular uptake studies demonstrated a 50 % increase over non‑targeted liposomes and a 20 % gain over single‑targeted constructs, an effect that was abrogated by competitive peptide blockade. In A549 xenograft mice, the dual‑ligand liposomes produced a signal‑enhancement ratio (SER) three times higher than free Gd‑DTPA and 1.5 × higher than single‑targeted variants at 2 h post‑injection, with the SER declining to only 60 % of peak after 6 h. These results underscore a synergistic targeting advantage and position the αvβ3/NRP‑1 dual‑ligand liposome as a potent platform for molecular MRI of tumors.
Introduction
MRI remains the imaging modality of choice for early solid‑tumor detection due to its superior soft‑tissue contrast and non‑ionizing nature. Paramagnetic agents such as Gd‑DTPA further enhance image quality, yet the low tumor‑to‑background signal ratio hampers precise localization. Liposomes offer a biocompatible vehicle that encapsulates hydrophilic agents, protecting them from plasma interactions and extending systemic half‑life. Functionalizing liposome surfaces with peptides, antibodies, or aptamers can redirect these carriers to over‑expressed tumor antigens, thereby increasing accumulation at malignant sites.
Angiogenic receptors, notably αvβ3 integrin and neuropilin‑1 (NRP‑1), are abundantly expressed on tumor endothelium and many tumor cells, making them attractive imaging targets. However, receptor heterogeneity often limits single‑ligand strategies. Dual‑targeted delivery, whereby two distinct ligands are presented on the same nanoparticle, expands cell‑surface binding opportunities and can enhance avidity and specificity. Building on our prior work with RGD‑functionalized liposomes, we hypothesized that co‑displaying αvβ3‑integrin and NRP‑1 ligands would amplify tumor targeting and MRI contrast enhancement.
Here we report the synthesis, characterization, and preclinical evaluation of a dual‑ligand paramagnetic liposome that co‑conjugates RGD and ATWLPPR. Comparative analyses with non‑targeted, single‑ligand, and mixed‑single‑ligand liposomes were performed in vitro and in vivo to assess cellular uptake, receptor binding, and MRI performance.
Materials and Methods
Chemicals
Egg phosphatidylcholine (PC, MW 775 Da) and mPEG2000‑DSPE (MW 2788 Da) were sourced from Avanti Polar Lipids; cholesterol (MW 386 Da) was purchased from Bio Basic. Gd‑DPTA (Magnevist) was supplied by Bayer Schering Pharma. All peptides and conjugates were custom‑synthesized by Yishengyuan (Shanghai, China).
Peptides and Conjugates
Three peptides were designed: dual‑targeted P1 (GARYC‑RGD‑CFD‑ATWLPPR, MW 2435 Da), single‑targeted P2 (GARYC‑RGD‑CFDG, MW 1670 Da), and single‑targeted P3 (ATWLPPR, MW 1191 Da). Each peptide was linked to a C6‑palmitic acid (Pal) anchor via FMOC solid‑phase synthesis. HPLC purity exceeded 90 % for all products.
Liposome Preparation
Liposomes were assembled by thin‑film hydration with a PC/cholesterol/mPEG2000‑DSPE molar ratio of 1.85:1:0.15. After solvent evaporation and film formation, peptides were dissolved in DMSO (final 1 % DMSO) and incorporated into the lipid film at peptide‑to‑lipid ratios of 4.5, 3, and 2.5 μg mol⁻¹ for P1, P2, and P3, respectively. The hydrated film was extruded sequentially through 0.4 µm, 0.2 µm, and 0.1 µm membranes to yield uniform nanoparticles. Gd‑DTPA was encapsulated during hydration; unencapsulated agent was removed by ultrafiltration (100 kDa MWCO). Final preparations (Gd‑LP, P1‑Gd‑LP, P2‑Gd‑LP, P3‑Gd‑LP, and P2/P3‑Gd‑LP) were stored at 4 °C under nitrogen.
Liposome Characterization
Dynamic light scattering measured particle size and zeta potential; transmission electron microscopy (TEM) confirmed morphology. Gadolinium loading was quantified by ICP‑OES.
Measurement of T1 Relaxivity
T1‑weighted images were acquired on a 3.0 T system (Philips/GE). Samples ranging from 1 × 10⁻³ to 1 × 10 mM Gd/L were prepared in PBS. An inversion‑recovery spin‑echo (STIR) sequence (TI = 200–9000 ms, TR = 10 000 ms, TE = 7.6 ms, FOV = 2 × 2 cm², matrix = 320 × 320, slice = 5 mm) was used to determine R1 values. Relaxivity (r₁) was calculated from (R₁obs – R₁m)/C.
Cell Lines and Culture
A549 (human adenocarcinoma) and HUVECs (human umbilical vein endothelial) were obtained from the Cancer Institute of Tongji University. Cells were cultured in DMEM supplemented with 10 % neonatal bovine serum, 100 U mL⁻¹ penicillin, and 100 µg mL⁻¹ streptomycin at 37 °C, 5 % CO₂, until 80–90 % confluence.
Cellular Uptake and Competitive Binding
Cells were incubated with Gd‑liposomes (10 mM Gd) for 4 h at 37 °C. After washing, cells were digested in nitric acid and Gd content measured by ICP‑OES. In competitive assays, free peptides (P1, P2, or P3) were co‑incubated with liposomes to block receptor binding.
MRI Capability of Detection In Vivo
Female BalB/c nude mice (4 weeks) received subcutaneous A549 inoculations (1 × 10⁴ cells). When tumors reached 50–100 mm³, mice were randomized (n = 5 per group) and anesthetized with urethane. Baseline T2‑weighted scans identified tumors, followed by T1‑weighted acquisitions before and at 0.5, 1, 2, 4, and 6 h post‑contrast. SER was calculated from mean signal intensities within tumor and hind‑limb muscle ROIs.
Statistical Analysis
Data were expressed as mean ± SD. One‑way ANOVA with post‑hoc testing (SPSS 22.0) determined significance; p < 0.05 was considered significant.
Results
Liposome Characterization
All formulations exhibited spherical, uniform morphology under TEM, with diameters below 100 nm and zeta potentials ranging from –15 to –60 mV. Mean sizes were: Gd‑LP 87.8 ± 0.9 nm, P1‑Gd‑LP 103.5 ± 1.2 nm, P2‑Gd‑LP 89.9 ± 1.5 nm, and P3‑Gd‑LP 89.9 ± 1.2 nm.
T1 Relaxivity of Dual-Targeted Liposome
Gd‑DTPA displayed the highest r₁ values among all groups, yet no statistically significant difference (p > 0.05) was observed relative to liposomal formulations (Fig. 1). This indicates that lipid encapsulation preserves the intrinsic relaxivity of Gd‑DTPA.
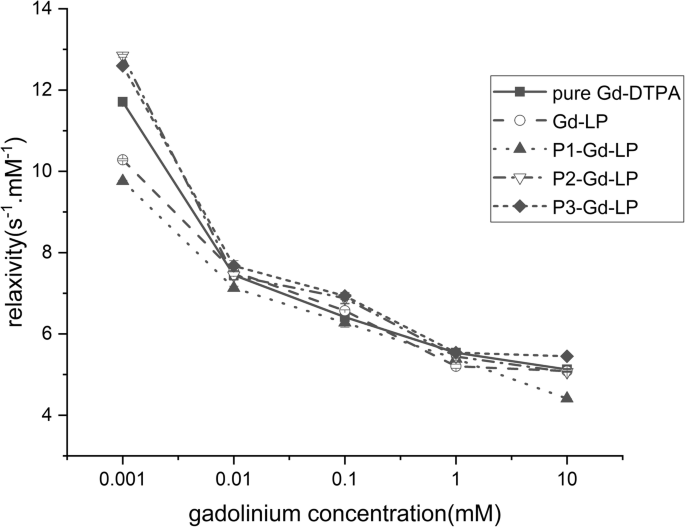
T1 relaxivity (s−1 mM−1) of pure Gd‑DTPA, Gd‑LP, P1‑Gd‑LP, P2‑Gd‑LP, and P3‑Gd‑LP measured at varying Gd concentrations (mM). Data represent mean ± SD (n = 3), p > 0.05.
Cellular Uptake and Competitive Binding
Dual‑targeted liposomes delivered 50 % more Gd to both A549 and HUVECs than non‑targeted liposomes, and 20 % more than single‑ligand variants (Fig. 2a). Mixed‑single‑ligand liposomes (P2/P3‑Gd‑LP) showed significantly lower uptake than the dual‑targeted construct. Competitive binding with free P1, P2, or P3 reduced Gd accumulation to near non‑targeted levels, confirming receptor‑mediated uptake (Fig. 2b–d).
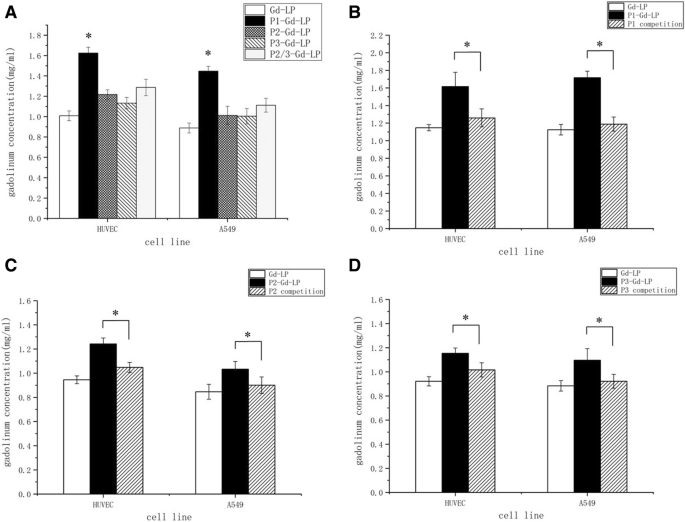
a Cellular uptake of Gd‑LP, P1‑Gd‑LP, P2‑Gd‑LP, P3‑Gd‑LP, and P2/P3‑Gd‑LP in A549 cells and HUVECs. b–d Competitive inhibition of P1‑, P2‑, and P3‑conjugated liposomes by corresponding free peptides. *p < 0.05 versus other groups.
MR Image Analysis
In vivo MRI demonstrated that dual‑targeted liposomes produced the highest SER across all time points (Fig. 4). At 2 h post‑injection, SER reached 3 × higher than free Gd‑DTPA and 1.5 × higher than single‑ligand liposomes. The SER plateaued between 2 and 6 h, declining to only 60 % of its peak value after 6 h, indicating prolonged tumor retention.

MR images of tumor-bearing mice before and after injection of different contrast agents at various time points. a pure Gd‑DTPA. b Gd‑LP. c P1‑Gd‑LP. d P2‑Gd‑LP. e P3‑Gd‑LP.
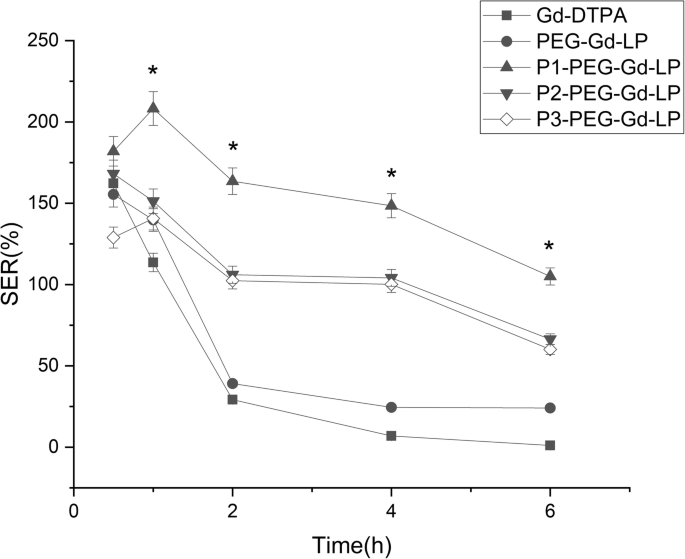
SER values at 0.5, 1, 2, 4, and 6 h post‑injection for each formulation (n = 3). *p < 0.05 for P1‑Gd‑LP versus other groups.
Discussion
Sub‑100 nm liposomes leverage the enhanced permeability and retention (EPR) effect, prolonging systemic circulation and facilitating tumor accumulation. Our dual‑ligand liposomes retained the physicochemical integrity of Gd‑DTPA, as evidenced by unchanged T1 relaxivity and minimal impact on particle size or surface charge.
Angiogenesis is a hallmark of tumor progression, with αvβ3 integrin and NRP‑1 playing pivotal roles in neovessel formation. While monomeric ATWLPPR exhibits moderate affinity for NRP‑1, its combination with the high‑affinity RGD sequence creates a heterodimeric ligand that targets both receptors simultaneously. This dual engagement increases binding avidity, particularly in heterogeneous tumor microenvironments where receptor expression varies among cell subpopulations.
Our in vitro competition studies confirm that cellular uptake of dual‑targeted liposomes is receptor‑specific: blockade of either αvβ3 or NRP‑1 dramatically reduces Gd accumulation. The superior in vivo SER profiles suggest that the dual‑ligand design not only enhances initial targeting but also sustains retention within the tumor vasculature and interstitium.
Comparative work by Wu et al. demonstrated that a PET tracer bearing RGD‑ATWLPPR achieved higher tumor uptake than single‑ligand analogues. Unlike radionuclide probes, our paramagnetic liposomes can carry high payloads of Gd‑DTPA, offering amplified contrast without ionizing radiation.
These findings support the potential of αvβ3/NRP‑1 dual‑targeted liposomes for early, non‑invasive MRI of diverse solid tumors, providing a platform that can be adapted for therapeutic payloads or multimodal imaging.
Conclusions
We successfully engineered a dual‑ligand paramagnetic liposome that co‑displayed RGD and ATWLPPR, encapsulating Gd‑DTPA without compromising its relaxivity. The dual‑targeted system demonstrated enhanced cellular uptake, receptor‑specific binding, and markedly improved MRI contrast in a tumor xenograft model. These results highlight the promise of αvβ3/NRP‑1 dual‑targeted liposomes as a versatile tool for early tumor detection and potential therapeutic delivery.
Abbreviations
- ATWLPPR:
Ala-Thr-Trp-Leu-Pro-Pro-Arg
- BBN:
Bombesin
- C6:
6‑Aminohexanoic acid
- CT:
Computed tomography
- DMEM:
Dulbecco’s Modified Eagle Media
- DMSO:
Dimethyl sulphoxide
- FMOC:
Fluorenylmethoxy carbonyl
- FOV:
Field of view
- Gd-DTPA:
Gadolinium‑diethylenetriamine pentaacetic acid
- HPLC:
High‑performance liquid chromatography
- HUVEC:
Human umbilical vein endothelial cell
- ICP‑OES:
Inductively coupled plasma optical emission spectrometer
- mPEG2000‑DSPE:
N‑(carbonyl‑methoxypolyethylene glycol‑2000)-1,2‑distearoyl‑sn‑glycero‑3‑phosphoethanolamine
- MRI:
Magnetic resonance imaging
- NRP1:
Neuropilin‑1
- Pal :
Palmitic acid
- PBS:
Phosphate buffered saline
- PC:
Phosphatidylcholine
- PET:
Positron emission tomography
- RGD:
Arg‑Gly‑Asp
- ROIs:
Regions of interest
- S/N:
Signal‑to‑noise
- SER:
Signal enhancement ratio
- SI:
Signal intensity
- STIR:
Inversion recovery spin‑echo
- TE:
Echo time
- TEM:
Transmission electron microscope
- TR:
Repetition time
- VEGF‑R:
Vascular endothelial growth factor receptor
Nanomaterials
- Magnetic Resonance Imaging (MRI): Principles, Evolution, and Clinical Applications
- PEG‑Coated Gold Nanostars: A Safe, High‑Contrast Agent for CT Imaging with Rapid Renal Clearance
- Heparanase‑Targeted Magnetic Gold Nanoparticle Probe Enhances MRI Detection of Tumor Metastasis
- Tuning Microwave Resonance in FeCoBSi Stripe‑Patterned Films: Thickness‑Dependent Magnetic Characterization
- HER2-Targeted Magnetic Nanosensitizer Enhances In Vivo MRI for HER2-Positive Cancers
- Magnetite Nanocluster-Based Theranostic Agents for T2‑Weighted MRI and pH‑Responsive Doxorubicin Delivery
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Magnetic Graphene Field‑Effect Transistor Biosensor for Ultra‑Sensitive Single‑Strand DNA Detection
- Dual‑Target Magnetic Nanoparticles Enable High‑Purity Lymphatic Endothelial Cell Isolation and Dual‑Modality Imaging in Colorectal Cancer
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer



