Controlled Hydrothermal Synthesis of ZnO Nanocrystals for Enhanced Inverted Polymer Solar Cells
Abstract
We report a scalable, cost‑effective hydrothermal route that delivers ZnO nanocrystals in a range of morphologies—nanorods, nanotetrapods, nanoflowers, and nanocubes—by simply adjusting solution concentration, temperature, and surfactant type. Incorporating these nanostructures into organic bulk‑heterojunction solar cells as the electron‑transport layer demonstrates that device performance is strongly morphology‑dependent, with the nanotetrapod configuration achieving the highest power‑conversion efficiency (PCE).
Background
Organic bulk‑heterojunction (BHJ) solar cells that employ n‑type inorganic metal‑oxide nanostructures for electron transport have gained traction due to their enhanced stability, low‑cost fabrication, and compatibility with solution processing [1–4]. ZnO nanocrystals stand out for their high electron mobility, optical transparency across the visible spectrum, and facile morphology control, making them ideal electron‑transport materials [5–7]. While prior studies have shown improved device metrics through nanorods, nanowalls, and nanotetrapods, the precise influence of morphology on photovoltaic performance remains debated.
In this work, we synthesize ZnO nanocrystals of distinct shapes using a straightforward hydrothermal process. By varying precursor concentration, temperature, and surfactant, we achieve nanorods, nanotetrapods, nanoflowers, and nanocubes. These nanostructures are then integrated into the active layer of inverted BHJ solar cells, and the resulting J–V characteristics reveal a clear morphology‑performance relationship. Our findings highlight that large surface area, optimal inter‑nanocrystal spacing for active‑layer infiltration, and continuous electron‑transport pathways are essential for maximizing device efficiency.
Methods
Deposition of ZnO Seed Layer
To nucleate ZnO growth on mismatched substrates, we deposit a seed layer via dip‑coating, following the protocol detailed in our previous publication [8].
Hydrothermal Growth of ZnO Nanocrystals
The ITO substrate, coated with the ZnO seed layer, is positioned upside‑down in a reaction vessel containing 40 ml of an aqueous mixture of zinc nitrate hexahydrate (Zn(NO3)2·6H2O) and hexamethylenetetramine (HMTA) at equal molarity. A chosen surfactant—either polyethylenimine (PEI) or sodium citrate—is added to tailor growth. The sealed vessel is then maintained at a constant temperature for a set duration. After growth, the ZnO nanocrystals are harvested, rinsed with deionized water, and dried at ambient conditions. For a detailed procedure, see reference [8].
Fabrication of the Solar Cells [9]
We first spin‑coat a thin PCBM layer (20 mg ml−1 in dichloromethane, 1000 rpm, 30 s) onto the ZnO nanocrystals. This intermediate layer enhances infiltration of the active polymer into the nanocrystal matrix. Next, a P3HT:PCBM blend (10 mg ml−1 P3HT, 16 mg ml−1 PCBM in chlorobenzene) is spin‑coated (1000 rpm, 30 s) and baked at 225 °C for 1 min to remove solvent and promote polymer penetration. A PEDOT:PSS hole‑transport layer (4000 rpm, 40 s) is then applied and annealed at 130 °C for 15 min, yielding ~35 nm thickness. Finally, a 100 nm Al cathode is thermally evaporated, and the device is annealed at 130 °C for 20 min under nitrogen. The complete architecture is illustrated in Fig. 1.
Device architecture of the organic bulk heterojunction solar cell
Characterization
Field‑emission scanning electron microscopy (FE‑SEM; Hitachi FE‑S4800) examines the ZnO nanocrystal morphology. Photovoltaic performance is evaluated using a Keithley 2400 source‑meter under AM 1.5G illumination (100 mW cm−2). All measurements are performed in triplicate to ensure reproducibility.
Results and Discussion
By tuning the hydrothermal parameters, we successfully produced ZnO nanorods, nanotetrapods, nanoflowers, and nanocubes. The most compelling evidence of morphological control comes from the patterned ZnO nanorod array fabricated on a TiO2 ring template derived from a self‑assembled polystyrene monolayer [11]. Figures 2a and 2b display the top view and 45° tilt of this array, grown from a 0.05 M Zn(NO3)2·6H2O/HMTA solution at 80 °C for 3 h. The rods exhibit a uniform 380 nm diameter and a 200 nm spacing, providing a short, continuous electron‑transport path and ample space for active‑layer infiltration.
The nanotetrapod array (Fig. 2c,d) was synthesized under similar conditions (0.025 M, 50 °C, 6 h) but with 0.1 ml PEI added, which promotes axial growth while suppressing radial expansion [12]. Each tetrapod, composed of 3–7 nanorods, offers a significantly larger surface area than the single‑rod array.

a Top view and b 45° tilt of the ZnO nanorod array. c Top view and d 45° tilt of the ZnO nanotetrapod array
Figure 3 presents SEM images of the nanoflower and nanocube morphologies, fabricated via a two‑step hydrothermal route. Initial nanorods (0.025 M, 85 °C, 3 h) serve as templates for secondary growth. Nanoflowers are formed in a 0.0075 M Zn(NO3)2·6H2O / 0.0075 M sodium citrate solution at 95 °C for 12 h, producing unordered, densely packed “petal” structures with ~30 nm inter‑petal gaps. Nanocubes emerge in a 0.0075 M Zn(NO3)2·6H2O / 0.015 M sodium citrate solution at 95 °C for 6 h, yielding ~150 nm cubic particles that are isolated from one another.

Top view of a the ZnO nanoflowers and b the ZnO nanocubes. The inset of Fig. 3a is a zoom‑in of a single nanoflower.
Four distinct ZnO nanocrystal morphologies were incorporated into identical inverted BHJ devices (Fig. 1). For each morphology, five devices were fabricated on the same ITO substrate; only the top three devices (within 3% PCE deviation) were considered for reporting. The resulting J–V curves are shown in Fig. 4, and key performance metrics are summarized in Table 1.
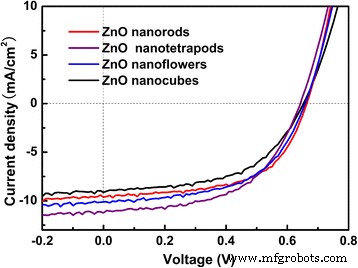
J–V characteristics of the organic bulk heterojunction solar cells with different ZnO nanostructures
The nanotetrapod device achieved the highest PCE of 3.96%, followed closely by the nanorod (3.71%) and nanoflower (3.69%) devices, while the nanocube configuration yielded 3.25%. The superior performance of the nanotetrapod is attributable to its extensive surface area (~300 nm inter‑crystal spacing) that facilitates both light absorption and efficient electron extraction. In contrast, the nanorod array, with lower surface area, exhibits reduced dye loading and consequently a lower short‑circuit current density (JSC). The nanoflower structure, despite its high surface area, suffers from tight inter‑petal spacing (<50 nm), hindering active‑layer infiltration and resulting in a slightly lower JSC. Finally, the isolated nanocube morphology interrupts electron transport pathways, further diminishing JSC and overall efficiency.
Conclusions
We have demonstrated that a simple, scalable hydrothermal process can produce ZnO nanocrystals with controlled morphologies—nanorods, nanotetrapods, nanoflowers, and nanocubes—by tuning precursor concentration, temperature, and surfactant. When integrated as the electron‑transport layer in inverted BHJ solar cells, morphology critically influences device performance. Optimal results arise from a large, accessible surface area, sufficient inter‑nanocrystal spacing for active‑layer infiltration, and an uninterrupted electron‑transport network.
Abbreviations
- HMTA:
Hexamethylenetetramine
- ITO:
Indium‑tin‑oxide
- JSC:
Short‑circuit current density
- J-V:
Current density–voltage
- P3HT:
Poly(3‑hexylthiophene)
- PCBM:
[6]-Phenyl‑C61-butyric acid methyl ester
- PCE:
Power‑conversion efficiency
- PEDOT:PSS:
Poly(3,4‑ethylenedioxythiophene):poly(styrene sulfonate)
- PEI:
Polyethylenimine
- SEM:
Field emission scanning electron microscopy
- VOC:
Open‑circuit voltage
Nanomaterials
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- How Polymers Are Synthesized: From Raw Materials to Advanced Additives
- Nanopillar‑Induced Modulation of Actin Cytoskeleton and Focal Adhesions in U2OS Cells
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Electrodeposited SnO₂ Thin Films on FTO: A Scalable Electron Transport Layer for High‑Efficiency Planar Perovskite Solar Cells
- Dye‑Sensitized Solar Cells: Fundamentals, Advances, and Commercial Outlook
- Enhanced Performance of CsPbI₂Br Perovskite Solar Cells via ZnCl₂–MnCl₂ Doping
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- High-Performance MoIn₂S₄@CNT Counter Electrodes for Dye‑Sensitized Solar Cells
- Ultra‑High Efficiency Silicon Cells with Inverted‑Pyramid Passivation: A Photovoltaic Breakthrough



