Nanopillar‑Induced Modulation of Actin Cytoskeleton and Focal Adhesions in U2OS Cells
Abstract
Background
We investigate how arrays of polymer nanopillars on glass influence the actin cytoskeleton and focal adhesion (FA) organization in U2OS osteosarcoma cells. Our analysis reveals three distinct cellular regimes—full substrate contact, peripheral adhesion, and suspension on pillars—determined by pillar density and geometry. Dense arrays induce peripheral FAs and a ring‑shaped actin cortex, mirroring the response of cells on compliant substrates.
Results
Using electron‑beam lithography, we fabricated SU‑8 nanopillar arrays with pitches of 500–2000 nm and heights of 500–1000 nm. High‑resolution Airyscan imaging of U2OS cells expressing LifeAct‑GFP and TagRFP‑vinculin quantified >400 cells and >7700 FAs across >400 images. Dense pillars (pitch 1000 nm, height 1000 nm) caused cells to float atop the array, with FAs concentrated at the periphery; sparse arrays (pitch 2000 nm, height 1000 nm) allowed full basal adhesion. Quantitative metrics showed significant differences in FA number, area, and peripheral localization at 24 h, which disappeared by 48 h.
Conclusion
High‑throughput nanofabrication, advanced microscopy, and rigorous image analysis provide a robust platform to dissect cell–nanostructure interactions. Our findings demonstrate that tailoring nanopillar geometry can steer actin architecture and FA distribution, offering a route to engineer culture substrates that bias specific cellular functions.
Graphic Abstract
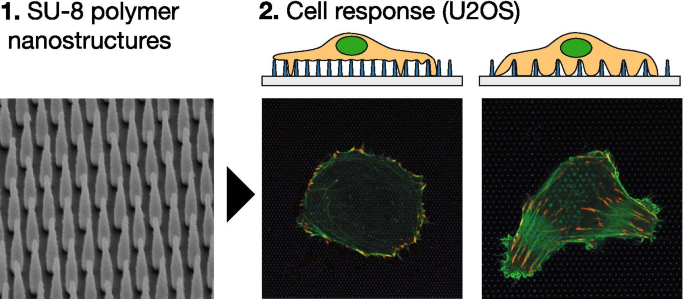
Background
In vivo, cells experience a complex 3D extracellular matrix (ECM) that delivers both biochemical cues and mechanical stiffness, regulating morphogenesis, homeostasis, and differentiation. ECM composition varies across tissues, influencing cell behavior.
To better mimic physiological conditions, researchers increasingly use 3D or semi‑3D culture systems. Compared with flat surfaces, cells on 3D matrices exhibit altered viability, proliferation, differentiation, and morphology, underscoring the importance of dimensionality in cell culture studies.
While true‑3D hydrogels (e.g., collagen, Matrigel) recapitulate the bulk ECM, 2.5D surfaces—flat glass decorated with nanostructures—provide precise, reproducible topography while remaining compatible with optical microscopy. Such surfaces enable controlled studies of membrane curvature, ligand spacing, and mechanical compliance.
Cell–ECM adhesion is mediated by focal adhesions, multiprotein complexes that link integrins to the actin cytoskeleton. Vinculin, a key FA component, anchors F‑actin, modulates filament bundling, nucleation, and signaling. Disruption of vinculin alters cell shape, adhesion strength, and force transmission.
Nanopillar arrays have been shown to influence cell morphology and FA distribution. Dense arrays can lift cells off the substrate, while sparse arrays allow basal contact. Prior work on gallium phosphide and silicon nanocolumns demonstrates density‑dependent FA number and morphology. However, systematic, quantitative studies on polymer nanostructures and their impact on actin and FA organization remain limited.
Results
SU‑8 nanopillar arrays were fabricated with controlled pitches (500–2000 nm) and heights (500–1000 nm). Arrays were classified as dense or sparse based on observed cell adhesion behavior. Live‑cell imaging captured ≥100 cells per condition at 24 h and 48 h, producing >400 high‑resolution images and 20 3D datasets.
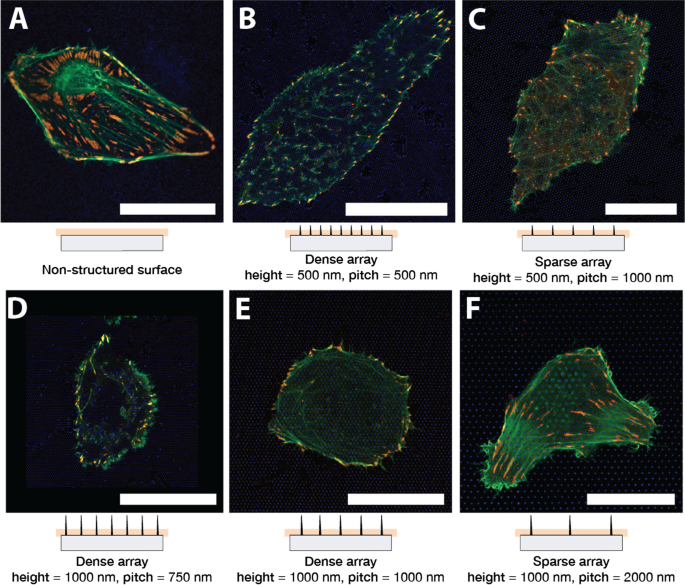
Representative U2OS cells on glass (A) and nanopillar arrays (B–F) after 24 h. LifeAct‑GFP (green) marks F‑actin; TagRFP‑vinculin (red) marks FAs. Scale bar 25 µm.
Cells on sparse arrays (pitch 2000 nm) displayed flat‑like morphology with basal FAs and F‑actin fibers extending to the substrate. In contrast, dense arrays (pitch 500–1000 nm, height 1000 nm) caused cells to float above the pillars; F‑actin formed a ring around the cell periphery, and FAs were confined to peripheral contact sites. Z‑stack imaging confirmed that actin networks in dense arrays resided above the pillar base, while in sparse arrays actin and FAs co‑localized at the substrate interface.
Quantitative analysis of >7700 FAs revealed significant differences at 24 h: dense arrays exhibited fewer, smaller FAs with a higher peripheral fraction, whereas sparse arrays and flat glass had more centrally located FAs. By 48 h, FA number, area, and distribution converged across conditions, suggesting a transient remodeling phase.
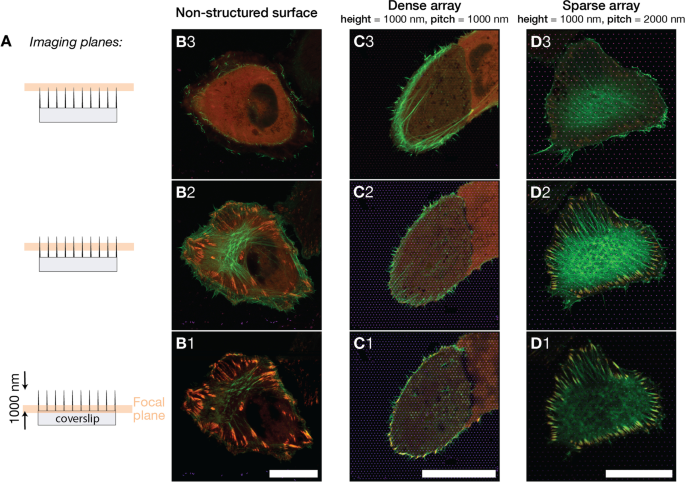
Multi‑plane Airyscan images: (A) focal planes at 0.0 µm, 0.4 µm, 0.8 µm; (B–D) merged LifeAct‑GFP and TagRFP‑vinculin on glass, 1000 nm, and 2000 nm arrays. Scale bar 25 µm.
Figure 3 summarizes cell morphology metrics (area, circularity, aspect ratio) across surfaces. Significant area reduction was observed at 24 h for dense arrays but not at 48 h. Circularity and aspect ratio remained comparable after 48 h.
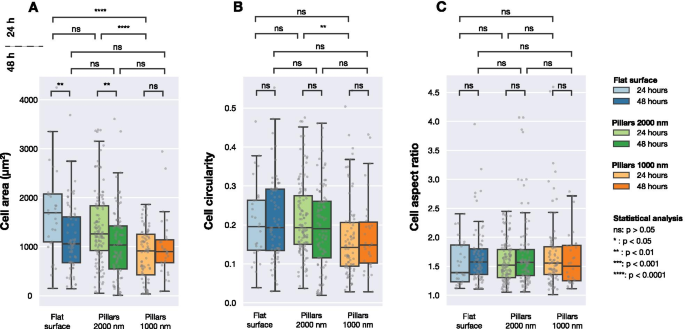
Cell area, circularity, and aspect ratio for U2OS cells on glass, 1000 nm, and 2000 nm arrays at 24 h and 48 h. Median and interquartile ranges are shown; Mann–Whitney tests indicate significance.
Figure 4 presents FA statistics. At 24 h, dense arrays had fewer FAs and smaller total FA area per cell. The FA area fraction (FA area ÷ cell area) was also lower for dense arrays. These differences dissipated by 48 h.
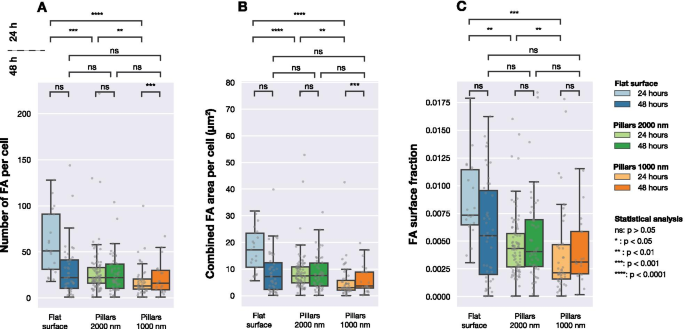
FA number, total FA area per cell, and FA area fraction for each surface. Statistical comparisons use Mann–Whitney tests.
FA localization analysis (Figure 6) shows that FAs on dense arrays are positioned closer to the cell edge, whereas on flat glass and sparse arrays they are more centrally distributed. Normalization to the maximum cell radius confirms this trend, highlighting a peripheral FA bias induced by pillar density.
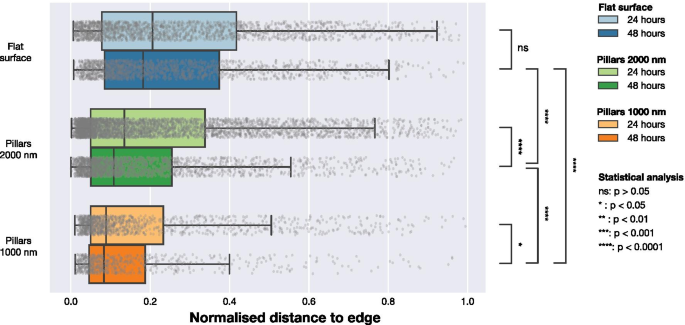
Normalized FA distances to the nearest cell edge for glass, 1000 nm, and 2000 nm arrays at 24 h and 48 h. Mann–Whitney tests assess distribution differences.
Discussion
Our study demonstrates that nanopillar geometry dictates actin architecture and FA organization in U2OS cells. Dense pillars lift cells off the substrate, creating a soft‑substrate‑like environment where FAs cluster peripherally and actin forms a cortical ring. Sparse pillars preserve basal adhesion and central FA distribution.
These observations align with reports of softer substrates inducing peripheral FAs and ring‑shaped actin. The transient differences at 24 h suggest that cells initially adjust to the topography before reaching a steady state by 48 h.
FAs mediate traction forces that shape stress fibers; conversely, actin tension influences FA maturation. Our data support a bidirectional coupling between cytoskeleton and adhesion complexes on nanostructured surfaces, extending previous findings on fibroblasts and silicon nanocolumns.
Future work should explore how these morphological changes affect signaling pathways and functional outputs such as migration, proliferation, and differentiation. The platform presented here offers a scalable route to engineer culture substrates that bias specific cellular phenotypes.
Conclusions
By combining high‑throughput nanofabrication, advanced Airyscan imaging, and quantitative image analysis, we identified three distinct U2OS cell regimes on polymer nanopillar arrays: full substrate contact, peripheral adhesion, and suspension. Pillar density and height govern actin organization and FA localization, with dense arrays mimicking the mechanical cues of compliant substrates.
These insights enable rational design of 2.5D culture platforms to direct cell behavior, potentially benefiting tissue engineering, drug screening, and mechanobiology research.
Methods
Fabrication of Nanostructures and Sample Mounting
Glass coverslips (24 mm × 24 mm, #1.5, 170 µm thick) were cleaned, plasma‑treated, and coated with SU‑8 2001 (500 nm or 1000 nm thick). Fluorescent SU‑8 was prepared by adding 100 µg mL⁻¹ of Oxazine 170, Rhodamine 800, or Coumarin 102. After spin‑coating, samples were baked, coated with conductive polymer (AR‑PC 5091), and exposed to a 100 kV EBL system to create pillar arrays (pitch 500–2000 nm, height 500–1000 nm). Post‑exposure development, oxygen plasma treatment, and platinum/palladium sputter coating completed the fabrication. Samples were mounted with the pillars facing upwards in 35 mm petri dishes for cell culture.
Cell Culture and Transfection
U2OS cells were grown in DMEM + 10% FBS at 37 °C, 5% CO₂. For experiments, 15,000 cells per well were seeded onto nanostructured or flat surfaces. After 6 h, cells were transfected with Lipofectamine 2000 using 0.5 µg plasmid each of pCMV‑LifeAct‑GFP and pTAG‑RFP‑vinculin. Transfection complexes were incubated for 20 min at room temperature before addition to wells. Media were refreshed 18 h post‑transfection.
Microscopy
Live imaging was performed on a Zeiss LSM 800 Airyscan with a 63×/1.4 NA oil objective. Images were acquired at 34 nm xy resolution and 180 nm z step size. A standard raster scan protocol ensured consistent data acquisition across all arrays.
Image Analysis
Images were processed with a custom Python 3 script (CZIfile, SciPy, scikit‑image). Background subtraction, median filtering, and Multi‑Otsu thresholding isolated vinculin‑rich FAs. Cell outlines were derived from LifeAct‑GFP intensity. Geometric descriptors (area, circularity, aspect ratio) were calculated; FAs smaller than 0.05 µm² or with extreme aspect ratios were excluded. The shortest Euclidean distance from each FA centroid to the cell edge was computed and normalized by the maximum cell radius.
Statistical Analysis
Non‑parametric Mann–Whitney tests compared distributions; p‑values ≥ 0.05 were considered non‑significant (ns). Significance levels: * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
Abbreviations
- DMEM
Dulbecco’s Modified Eagle’s Medium
- EBL
Electron Beam Lithography
- ECM
Extracellular Matrix
- EDTA
Ethylenediaminetetraacetic Acid
- FA
Focal Adhesion
- FBS
Fetal Bovine Serum
- GFP
Green Fluorescent Protein
- HMDS
Hexamethyldisilazane
- NP
Nanopillar
- RFP
Red Fluorescent Protein
- SEM
Scanning Electron Microscopy
Nanomaterials
- Special‑Purpose Batteries: From Mercury Standards to Fuel, Solar, and Chemical Detection Cells
- Solar Cells: From Early Experiments to Modern Photovoltaics – Technology, Production, and Future Outlook
- Controlled Hydrothermal Synthesis of ZnO Nanocrystals for Enhanced Inverted Polymer Solar Cells
- Atomic Force Microscopy of Glass Transition and Adhesion in Thin Polystyrene Films
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Dye‑Sensitized Solar Cells: Fundamentals, Advances, and Commercial Outlook
- Probing the Electrical Response of Double‑Sided Polymer Surface Nanostructures
- Unveiling Surface-Related Excitons and Lasing in CdS Nanostructures
- Enhanced Photocatalytic and Antibacterial Performance of Annealed Tin Oxide Nanostructures: A Molecular Docking Study
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



