Harnessing Reactive Oxygen Species Toxicity for Advanced Tumor Therapy: Nanoparticle Strategies and Clinical Outlook
Abstract
Reactive oxygen species (ROS) possess a dual nature: essential for cellular signaling yet lethal at elevated concentrations. By deliberately elevating ROS in tumors, nanomedicine can selectively trigger oxidative stress that leads to cancer cell death. This review surveys recent nanotechnologies that convert ROS toxicity into therapeutic tools, emphasizing photodynamic, chemodynamic, and radiation‑enhanced strategies.
Introduction
ROS—including singlet oxygen (¹O₂), superoxide anion (O₂⁻·), hydroxyl radical (·OH), and hydrogen peroxide (H₂O₂)—originate primarily from mitochondrial electron transport. While low‑level ROS support normal physiology, excessive ROS inflict DNA, protein, and lipid damage, accelerating tumor initiation and progression. Conversely, heightened ROS can overwhelm antioxidant defenses, causing apoptosis or necrosis in malignant cells.
Understanding the balance between ROS generation and scavenging is crucial for designing nanosystems that precisely modulate oxidative stress within the tumor microenvironment (TME). Recent advances in ROS‑based nanotheranostics offer a promising platform to exploit this balance for selective cancer eradication.
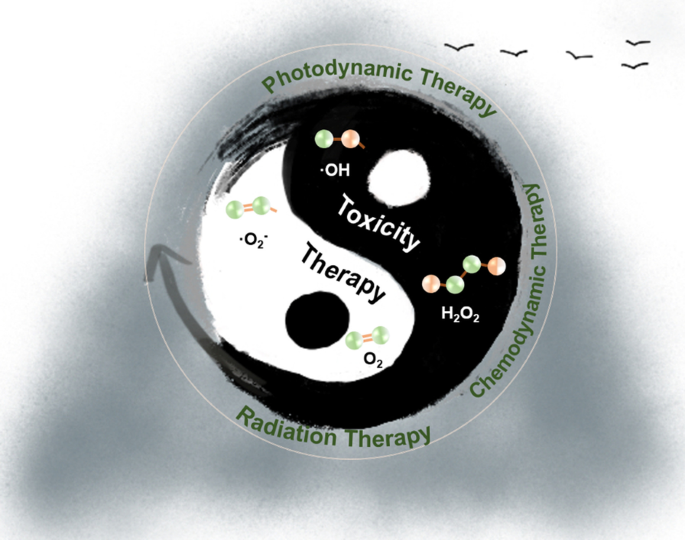
Schematic representation of transforming ROS toxicity into therapeutic advantage.
Fight Fire with Fire
The cytotoxic potential of ROS can be harnessed by nanomedicine to amplify oxidative stress selectively within tumors. Key advantages of ROS‑based nanotherapies include tunable ROS production, controlled delivery, and minimal off‑target effects. We review three primary modalities that exploit ROS: photodynamic therapy (PDT), chemodynamic therapy (CDT), and radiation therapy (RT).
Photodynamic Therapy
PDT relies on a photosensitizer (PS), light, and oxygen to generate cytotoxic ROS, primarily ¹O₂. Conventional PSs often suffer from poor tissue penetration and low quantum yields under clinically relevant wavelengths. Nanomaterials address these limitations by acting as light harvesters, oxygen carriers, and PS carriers.
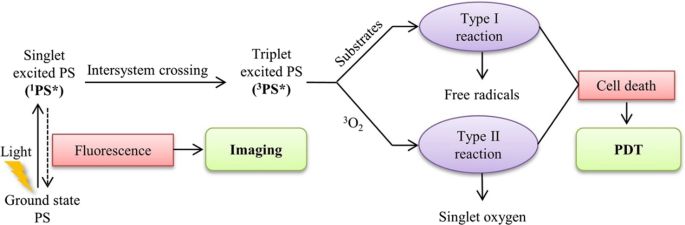
Schematic drawing of a typical photodynamic reaction.
Examples include black phosphorus nanosheets (BP NSs) that generate ¹O₂ under 660 nm NIR light, and BP‑PEI/AuNP hybrids that combine photothermal and photodynamic effects for synergistic tumor suppression. Gold nanostructures—nanorods, cluster‑luminogens, and thiol‑coated nanoclusters—serve as self‑sufficient PSs, achieving tumor ablation without organic sensitizers. These advances demonstrate the feasibility of nanomaterial‑mediated PDT with enhanced depth, efficiency, and safety.
Chemodynamic Therapy
CDT employs Fenton or Fenton‑like reactions between transition metal ions and endogenous H₂O₂ to produce highly reactive ·OH radicals. Because tumors contain elevated H₂O₂ and an acidic microenvironment, metal‑based nanocarriers can release Fe²⁺ or other catalytic ions selectively within the TME, triggering ROS generation and cell death.
Notable systems include pH‑responsive Fe₅C₂@Fe₃O₄ nanoparticles that combine MRI contrast with ROS production, and Fe‑MOF catalysts that simultaneously deplete glutathione and generate ·OH. Copper‑MOF nanoparticles loaded with chlorin e6 exploit hypoxia‑responsive release, depleting GSH and promoting ·OH generation for combined chemodynamic and sonodynamic therapy. These studies underscore the versatility of CDT and its potential to overcome the limitations of light‑dependent approaches.
Radiation Therapy
RT induces DNA damage via ROS generated by ionization of water. Tumor hypoxia and DNA repair pathways often limit RT efficacy. Nanoparticle radiosensitizers enhance ROS production and impede repair mechanisms.
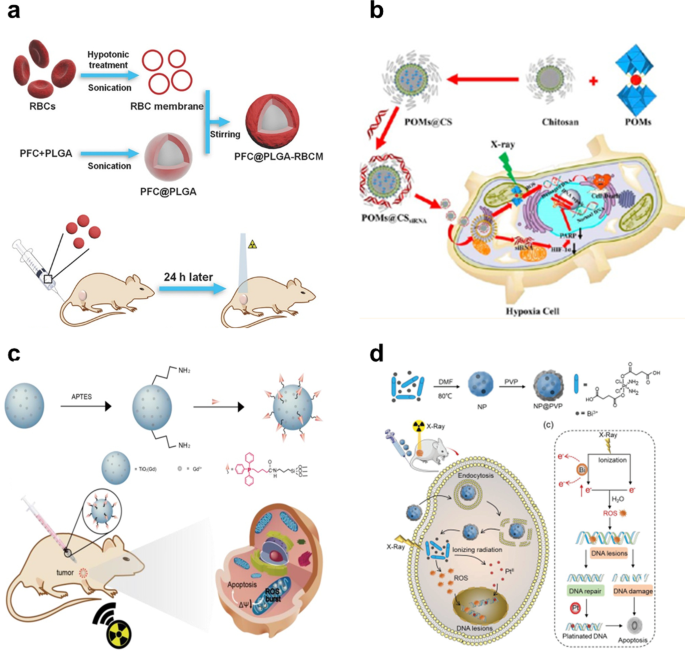
Illustrations of various nanoparticle‑mediated radiosensitization approaches.
Oxygen‑laden perfluorocarbon cores coated with red‑blood‑cell membranes alleviate hypoxia, while Gd‑W₁₀@CS nanoparticles degrade GSH and inhibit DNA repair via HIF‑1α siRNA. Mitochondria‑targeted Gd‑doped TiO₂-TPP nanoparticles and Bi‑containing bismuth–cisplatin prodrug platforms further boost ROS generation and enhance DNA damage, achieving superior tumor control at lower radiation doses.
Conclusions and Outlook
Transforming ROS toxicity into therapeutic benefit hinges on precise control of ROS levels within tumors. Nanoparticle platforms—whether for PDT, CDT, or RT—offer tunable delivery, targeted activation, and synergistic effects. Challenges remain, including scalable synthesis, long‑term biocompatibility, and comprehensive mechanistic understanding. Future research should focus on multifunctional, stimuli‑responsive systems that combine ROS generation with imaging, drug delivery, and immune modulation to achieve durable, systemic cancer eradication.
Availability of data and materials
Not applicable.
Abbreviations
- ROS:
Reactive oxygen species
- 1O2:
Singlet oxygen
- O2-:
Superoxide anion
- ·OH:
Hydroxyl radical
- H2O2:
Hydrogen peroxide
- PDT:
Photodynamic therapy
- CDT:
Chemodynamic therapy
- RT:
Radiation therapy
- PSs:
Photosensitizers
- O2:
Oxygen
Nanomaterials
- Horseradish Peroxidase‑Encapsulated Hollow Silica Nanospheres for Intracellular ROS Sensing
- Enhanced Reactive Oxygen Species Generation by GdVO4:Eu³⁺ Nanoparticles and Methylene Blue Complexes under UV–Vis and X‑ray Irradiation
- Synergistic Antioxidant and Anti‑Amyloid Action of Chlorogenic Acid–Selenium Nanoparticles in Vitro
- Hydrophilic HBP‑Modified rGO Enables Dual pH/NIR‑Triggered DOX Release for Synergistic Chemo‑Photothermal Tumor Therapy
- Improving HIFU Cancer Therapy Using Sonodynamic N2O Microbubbles
- Nanoparticle Toxicity in Biomedicine: Reactive Oxygen Species Impact
- Advances in Thermosensitive Hydrogels for Targeted Tumor Therapy
- Fine-Tuning Running Shoes for Peak Performance
- Advanced Ceramics: Benefits, Challenges, and Industrial Applications
- Why On-Site Oxygen Generation Is a Game-Changer for Industrial Efficiency



