Dose‑Dependent Liver, Brain, and Embryonic Toxicity of Titanium Dioxide Nanoparticles in Mice
Abstract
Titanium dioxide nanoparticles (TiO₂‑NPs) are ubiquitous in food, cosmetics, and medical products, yet their safety profile remains uncertain. Using ICR mice, we examined the biodistribution, organ‑specific toxicity, and underlying mechanisms of TiO₂‑NPs delivered via intraperitoneal (i.p.) injection and oral gavage. Titanium accumulation followed a dose‑dependent pattern, with the liver showing the highest burden, followed by kidney, spleen, lung, brain, and heart. High‑dose exposure (≥150 mg kg⁻¹) led to significant organ‑body weight ratio alterations, liver and kidney functional impairment, and metabolic disturbances as reflected in serum biochemistry. In the liver, TiO₂‑NPs induced mitochondrial swelling, hepatocyte apoptosis, reactive oxygen species (ROS) overproduction, and dysregulation of antioxidant and detoxifying genes (SOD, CAT, GST, HSP70, CYP1A1). In the brain, high doses caused neuronal rupture, inflammatory infiltration, increased nitric oxide synthase activity, altered glutamate and acetylcholinesterase levels, and lipid peroxidation. Ex vivo embryo cultures exposed to ≥100 µg mL⁻¹ TiO₂‑NPs exhibited reduced crown‑rump length, head length, and increased malformation rates, with toxicity escalating with particle size. These findings underscore the organ‑specific, dose‑dependent risks of TiO₂‑NP exposure and provide a basis for human health risk assessment.
Background
Titanium dioxide nanoparticles (TiO₂‑NPs) are incorporated into a broad spectrum of consumer products, including confectionery, beverages, dairy, cosmetics, and pharmaceuticals. Reported concentrations in food can reach 0.5–9 g kg⁻¹, and products marketed as “nano‑free” often still contain TiO₂‑NPs [1–3]. While TiO₂‑NPs offer valuable functional properties, their potential to accumulate in biological tissues and induce toxicity is a growing concern [4–6]. Animal studies have demonstrated that TiO₂‑NPs can be taken up via the gastrointestinal tract or inhalation, subsequently distributing to liver, kidney, spleen, lung, heart, and brain [7–8]. In vitro, TiO₂‑NPs have elicited cytotoxicity in lymphoblastoid and hepatoma cell lines, induced oxidative stress in glial cells, and compromised neuronal viability in a dose‑dependent manner [9–12]. The mechanisms involve oxidative damage, membrane permeability alteration, DNA oxidation (8‑OHG), inflammation, and apoptosis [13–19]. Comprehensive, systematic data on TiO₂‑NP toxicity remain scarce; hence, this study seeks to elucidate its effects and mechanisms in vivo.
Methods
Chemicals and Reagents
Micro‑scale TiO₂ and 5 nm anatase TiO₂ were sourced from Sigma‑Aldrich (Shanghai). 10, 60, and 90 nm anatase TiO₂ were purchased from Run He Ltd. (Shanghai). Reagents of analytical grade were obtained from Sigma‑Aldrich (Shanghai) and Gibco (USA). TiO₂ suspensions (1 %) were sterilized, sonicated, and diluted to desired concentrations prior to use.
Animals and Models
ICR mice (22 ± 3 g, 50 % male/female) were used for organ toxicity studies; ICR females (45, 20–35 g) and males (15, 35–40 g) for embryo toxicity. All procedures were approved by the Tianjin University of Science and Technology Ethics Committee and adhered to international animal care guidelines.
Dosing Regimens
For general toxicity, mice received i.p. TiO₂‑NPs at 5, 10, 50, 100, 150, or 200 mg kg⁻¹ daily for 14 days (10 mice/group). Control animals received saline. For liver toxicity, mice were gavaged with 5, 10, or 50 mg kg⁻¹ daily for 60 days (10 mice/group); controls received 0.5 % CMC.
Titanium Quantification
Tissue samples were digested with HNO₃/H₂O₂, diluted, and analyzed by ICP‑MS. Titanium concentrations were expressed per gram of tissue.
Blood Biochemistry
Serum enzyme activities (ALT, ALP, AST, LDH, CK, etc.) were measured using a Toshiba TBA‑2000FR analyzer. Organ/body weight ratios were calculated post‑euthanasia.
Histopathology and TEM
Organs fixed in 10 % formalin were stained with hematoxylin–eosin. Liver and brain tissues were processed for transmission electron microscopy to assess mitochondrial integrity and cellular ultrastructure.
Oxidative Stress and Gene Expression
ROS levels (O₂⁻, H₂O₂, MDA) were quantified by colorimetric assays. Antioxidant enzyme activities (SOD, CAT, GPx, APX) and non‑enzymatic antioxidants (ASA/DASA, GSH/GSSG) were measured in brain homogenates. mRNA levels of antioxidant and stress‑related genes were quantified by qPCR, normalized to β‑actin.
Ex Vivo Embryo Toxicity
Embryos at embryonic day 8.5 were cultured with TiO₂‑NPs (0–200 µg mL⁻¹) for 48 h, with additional time‑course studies (16, 26, 48 h). Developmental parameters (crown‑rump length, head length, yolk sac diameter, body sections) and malformation rates were assessed using the Maele‑Fabry Van score [22].
Statistical Analysis
Data were analyzed with SPSS 13; significance was set at p < 0.05. Comparisons employed Dunnett’s t‑test, ANOVA, LSD, SNK, chi‑square, or rank‑sum tests as appropriate.
Results
Tissue Distribution of Titanium
TiO₂‑NPs accumulated in liver, kidney, spleen, lung, brain, and heart in a dose‑dependent manner (Fig. 1). The liver exhibited the highest titanium burden, followed by kidney, with similar levels in spleen, lung, brain, and heart.
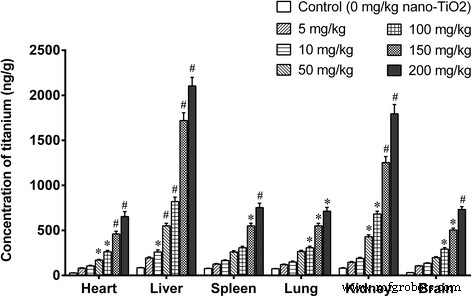
Figure 1. Titanium concentration in organs after 14‑day i.p. exposure to TiO₂‑NPs. *p < 0.05, #p < 0.01 versus control.
General Toxicity
Body weight gain was unaffected across all doses. Low doses (5–10 mg kg⁻¹) did not alter organ/body weight ratios. High doses (≥50 mg kg⁻¹) increased liver, kidney, spleen, and heart ratios while reducing lung and brain ratios (Fig. 2).
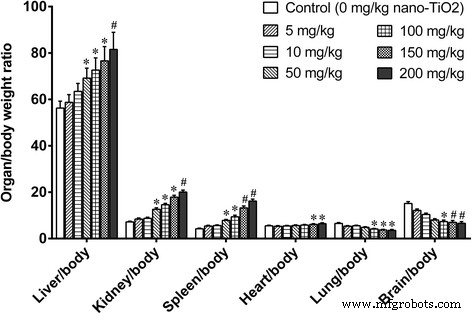
Figure 2. Organ/body weight ratios after 14‑day i.p. exposure. *p < 0.05, #p < 0.01 versus control.
High‑dose exposure (150–200 mg kg⁻¹) elevated serum liver enzymes (ALT, ALP, LAP, PChE, TBIL, TP, ALB) and renal markers (decreased UA, BUN), and increased myocardial injury markers (AST, CK, LDH, HBDH) and metabolic parameters (GLU, TG, TC, HDL). Low doses showed no significant biochemical changes (Fig. 3).
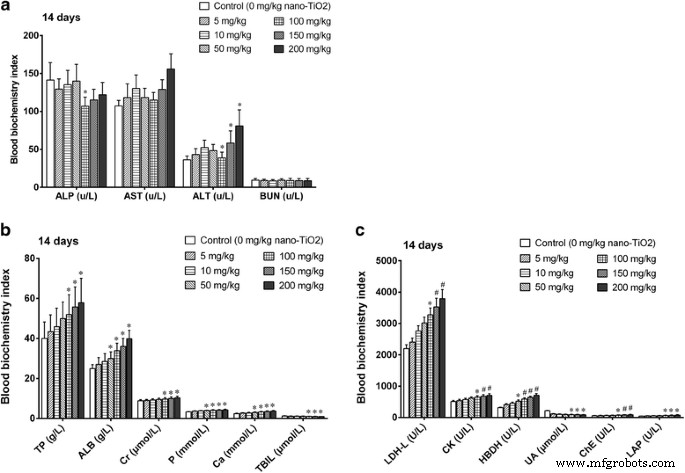
Figure 3. Serum biochemistry after 14‑day i.p. exposure. *p < 0.05, #p < 0.01 versus control.
Liver Toxicity
Histology revealed dose‑dependent vascular congestion, basophilic infiltration, ischemia, and central vein obstruction at ≥50 mg kg⁻¹ (Fig. 4). TEM showed mitochondrial swelling, chromatin condensation, and apoptotic bodies at doses as low as 5 mg kg⁻¹ gavage (Fig. 5). Low‑dose gavage did not alter ROS, MDA, or antioxidant gene expression, whereas 10–50 mg kg⁻¹ increased ROS (O₂⁻, H₂O₂, NO, MDA) and down‑regulated SOD, CAT, GSHPx, MT, GST, HSP70, P53, TF, while up‑regulating CYP1A1 (Fig. 6 & 7).
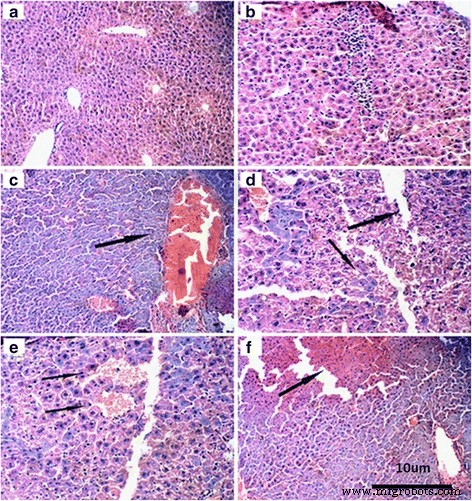
Figure 4. Liver histology at various TiO₂‑NP doses.
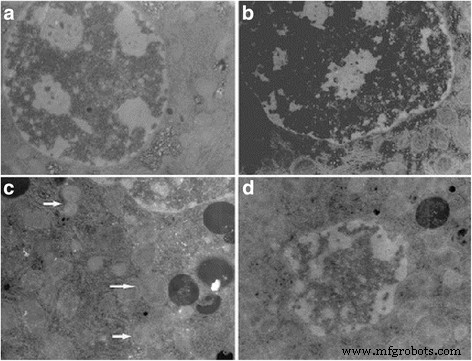
Figure 5. TEM of hepatocytes after gavage exposure.
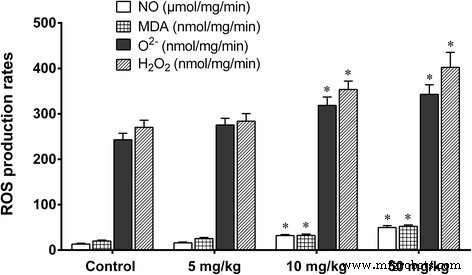
Figure 6. ROS production and MDA levels in liver.
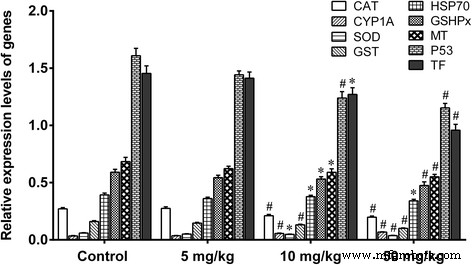
Figure 7. Relative expression of liver antioxidant and stress genes.
Brain Toxicity
High doses (≥100 mg kg⁻¹) reduced brain/body weight ratios and increased brain titanium levels (Fig. 1). Histology showed neuronal rupture, cracking, and inflammatory infiltration at 150–200 mg kg⁻¹ (Fig. 8). Oxidative parameters increased, while antioxidant enzyme activities and non‑enzymatic antioxidants decreased. Nitric oxide synthase activities and NO levels rose; acetylcholinesterase activity and glucose decreased (Fig. 9 & 10).
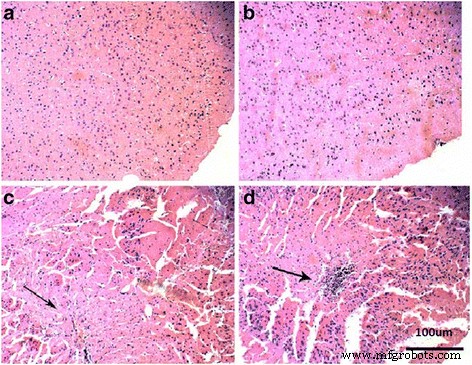
Figure 8. Brain histology at various TiO₂‑NP doses.
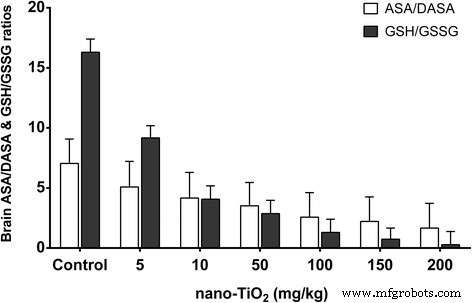
Figure 9. ASA/DASA and GSH/GSSG ratios.
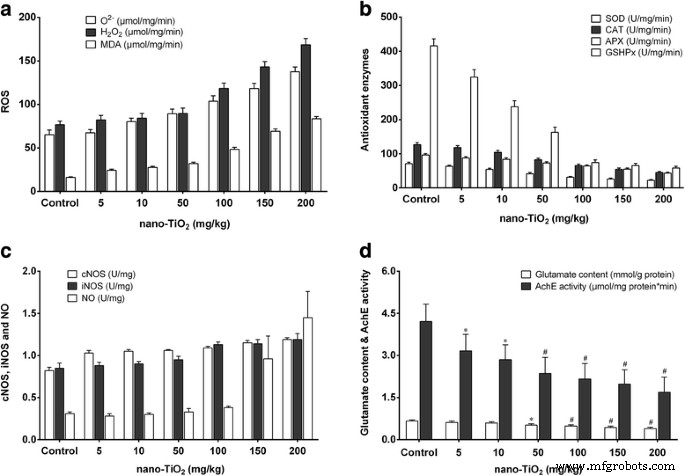
Figure 10. ROS, antioxidant enzymes, NO signaling, glutamate, and acetylcholinesterase changes.
Embryonic Toxicity
Ex vivo embryos exposed to ≥100 µg mL⁻¹ TiO₂‑NPs displayed reduced crown‑rump length, head length, and increased malformation rates, with toxicity escalating with particle size (Table 2). Time‑course studies showed significant developmental delays and malformations after 24–48 h exposure to 200 µg mL⁻¹, irrespective of size (Table 3).
Discussion
Our data confirm that TiO₂‑NPs accumulate in multiple organs in a dose‑dependent manner. High‑dose exposure leads to hepatocellular injury, renal dysfunction, and metabolic disturbances, consistent with prior reports [7, 23–28]. Oxidative stress, evidenced by increased ROS and MDA and decreased antioxidant defenses, appears central to hepatotoxicity. Gene expression analysis supports a disruption of detoxification (SOD, CAT, GST), stress response (HSP70, P53), and xenobiotic metabolism (CYP1A1) pathways.
In the brain, TiO₂‑NPs induce lipid peroxidation, neuronal damage, and inflammatory responses, accompanied by dysregulated nitric oxide signaling and neurotransmitter imbalance. These findings align with neurotoxicity reported for other metal oxide nanoparticles [24, 26, 37, 40].
Embryonic cultures revealed dose‑ and size‑dependent developmental toxicity, echoing in vivo studies that demonstrate reproductive and developmental hazards of TiO₂‑NPs [46–48, 49, 51]. The trend of greater toxicity with larger particles may reflect enhanced cellular uptake or altered surface reactivity.
Collectively, TiO₂‑NPs pose organ‑specific risks that are strongly dose‑dependent. Their capacity to generate oxidative stress, impair antioxidant defenses, and alter gene expression underlies the observed hepatotoxicity, neurotoxicity, and developmental toxicity.
Conclusions
Ingested TiO₂‑NPs distribute to heart, brain, spleen, lung, and kidney, causing liver, kidney, and cardiac dysfunction, glucose and lipid metabolic disturbances, and neurotoxicity via oxidative stress and gene dysregulation. Embryonic development is impaired in a dose‑ and size‑dependent manner. These findings underscore the need for stringent risk assessment of TiO₂‑NP exposure in humans.
Abbreviations
- ALB
- Albumin
- ALP
- Alkaline phosphatase
- ALT
- Alanine aminotransferase
- AST
- Aspartate aminotransferase
- BUN
- Blood urea nitrogen
- CK
- Creatine kinase
- cNOS
- Constitutive nitric oxide synthases
- HBDH
- Hydroxybutyrate dehydrogenase
- ICR
- Imprinting control region
- iNOS
- Inducible nitric oxide synthase
- LAP
- Leucine aminopeptidase
- LDH
- Lactate dehydrogenase
- Nano‑TiO₂
- Nanoscale titanium dioxide
- PChe
- Butyrylcholinesterase
- ROS
- Reactive oxygen species
- TBIL
- Total bilirubin
- TP
- Total protein
- UA
- Uric acid
Nanomaterials
- Titanium Dioxide (TiO₂) Market Update – Q1 2021 Prices, Supply & Regional Outlook
- PEG‑PCCL Nanoparticles: Low‑Toxicity, Sustained Paclitaxel Delivery and Enhanced Anti‑Tumor Efficacy in Hepatocarcinoma Models
- Hybrid TiO₂ Nanocomposite Coating Achieves 80% Diffuse Reflectance and Suppressed Specular Glare
- Prenatal Exposure to Titanium Dioxide Nanoparticles Alters Maternal Gut Microbiota and Elevates Fasting Blood Glucose in Rats
- Controlling Silver Nanoparticle Morphology via CTAB‑Capped Seeds and Aging Time
- Comparative Toxicity of PEG-Coated Cobalt Ferrite Nanoparticles and Nanospheres
- Developing a Precise Lysosomal pH Sensor Using Self-Decomposing SiO2 Nanoparticles
- Gold Nanoparticle-Drug Interactions Cause Acute Kidney Damage in Mice: A Size-Dependent Toxicity Study
- A Novel Analytical System for Assessing Silver Nanoparticle Physicochemical Properties in Human Skin
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications



