High‑Performance Polyelectrolyte Microcapsules Encoded with PEGylated Quantum Dots for Advanced Bioimaging
Abstract
Fluorescent imaging is a cornerstone of modern diagnostics, enabling real‑time visualization of molecular, cellular, and tissue dynamics. Semiconductor nanocrystals—quantum dots (QDs)—combine size‑tunable emission, broad absorption, and superior photostability, making them ideal fluorophores for labeling biomolecules and encoding microparticles. In this study, we present a streamlined, solvent‑free method for incorporating water‑soluble, PEG‑functionalized CdSe/ZnS QDs into the multilayer membrane of polyelectrolyte microcapsules via layer‑by‑layer deposition. The resulting hollow capsules exhibit uniform size (mean 4.45 ± 0.65 µm), robust surface charge control, and a 590 nm fluorescence peak identical to the free QDs. Flow cytometry and fluorescence microscopy confirm that the capsules form a homogeneous population with high signal intensity (~10⁴ PE units), enabling straightforward detection in standard laboratory instruments. These PEGylated QD‑encoded microcapsules hold promise as bright, photostable bioimaging and theranostic agents that can be further functionalized for stimulus‑responsive delivery.
Introduction
Targeted delivery of therapeutics and high‑resolution imaging demand carriers that can be precisely tracked in vivo. Polyelectrolyte microcapsules, assembled through electrostatic layer‑by‑layer deposition, offer a versatile platform for incorporating functional agents, including magnetic, metallic, and fluorescent nanoparticles. Quantum dots, with their narrow, symmetric emission spectra that scale with particle size, enable multiplexed detection using a single excitation wavelength. Their photostability and quantum yields (~40 %) rival organic dyes, while their larger extinction coefficients confer higher brightness per particle.
Traditional fluorescent labeling of microcapsules typically relies on covalent conjugation or physical mixing of dyes, which can suffer from leaching and photobleaching. Embedding QDs within the capsule membrane presents a more robust optical encoding strategy, provided the QDs remain water‑soluble and stably anchored. Here we report the synthesis of carboxyl‑terminated PEGylated CdSe/ZnS QDs, their integration into a multilayer polyelectrolyte shell, and the comprehensive characterization of the resulting fluorescent microcapsules.
Methods
Aim, Design, and Study Setting
Fabrication of QD‑Encoded Polyelectrolyte Microcapsules
CdSe/ZnS core/shell QDs (590 nm peak) were synthesized by Dr. P. Samokhvalov (NRNU MEPhI, Moscow) and purified through repeated chloroform extraction and cysteine precipitation. The QDs were subsequently PEGylated using a thiol‑terminated PEG12‑COOH derivative (1:4.6 mass ratio) and purified by gel filtration. Hydrodynamic diameter (18.7–23.3 nm) and ζ‑potential (−26.7 ± 0.8 mV) were measured by DLS and laser Doppler micro‑electrophoresis (Zetasizer Nano ZS).
Layer‑by‑layer deposition was performed on calcium carbonate (CaCO₃) microparticles. Five initial polyelectrolyte layers (PAH/PSS) were assembled, followed by adsorption of the PEGylated QDs onto the PAH surface. Additional PAH/PSS layers capped the QDs, and the CaCO₃ core was removed by 0.2 M EDTA (pH 6.5). The resulting hollow microcapsules were surface‑modified with bovine serum albumin (BSA) by incubation in 50 mM phosphate buffer (pH 7.4, 1 % BSA) at 4 °C for 12 h. Final suspensions were washed five times and stored at 4 °C in the dark.
Optical and Fluorescence Microscopy
Microcapsule morphology and size distribution were examined using an Axio Observer 3 (Zeiss) with a 40×/0.55 lens. Fluorescent imaging employed an HBO 100 mercury illuminator, FITC long‑pass filter set, and a 100×/1.30 oil objective (Immersol 518F). Sectioned samples (150 nm) were prepared by epoxy embedding and ultramicrotomy (Leica EM UC6). Fluorescence of sections was recorded with a TexasRed filter set (560/40 nm excitation, 630/75 nm emission).
Fluorescence Characterization
Emission spectra of free and encapsulated QDs were recorded on an Infinite 200 PRO multimodal plate reader (Tecan) using 480 nm excitation. Samples were centrifuged (2630 × g, 20 min) before measurement.
Flow Cytometry
Samples (10⁶ beads/mL) were analyzed on an FACSCanto II (BD) with a 488 nm laser. Forward (FSC), side scatter (SSC), and PE (585/42 nm) signals were recorded. Data were processed in FACSDiva.
Materials
Carboxylated PEG12‑HS (Thermo Fisher), PAH (Mw ≈ 15 kDa, Sigma‑Aldrich), PSS (Mw ≈ 70 kDa, Sigma‑Aldrich), PAA (Mw ≈ 15 kDa, Sigma‑Aldrich), CaCl₂, Na₂CO₃, EDTA, BSA, and all reagents were purchased from Sigma‑Aldrich. Solutions were prepared with MilliQ water (18.2 MΩ cm).
Statistical Analysis
Data were analyzed with MS Excel 2007 and Origin Pro 2015; results are reported as mean ± SD from three independent experiments.
Results and Discussion
Development of QD‑Encoded Polyelectrolyte Microcapsules
The layer‑by‑layer strategy offers a solvent‑free, biocompatible route to encapsulate QDs within a controllable polyelectrolyte shell. Calcium carbonate particles first acquire a PAH layer, followed by alternating PSS and PAH layers to build a 10‑layer shell. The PEGylated QDs, carrying a negative surface charge, are then adsorbed onto the terminal PAH layer and sealed with additional polyelectrolyte layers. Removal of the CaCO₃ core by EDTA yields a hollow capsule; a final BSA coating confers biocompatibility and reduces non‑specific aggregation.
Figure 1 illustrates the layering sequence, ζ‑potential evolution, and a representative fluorescence micrograph of the final capsules.
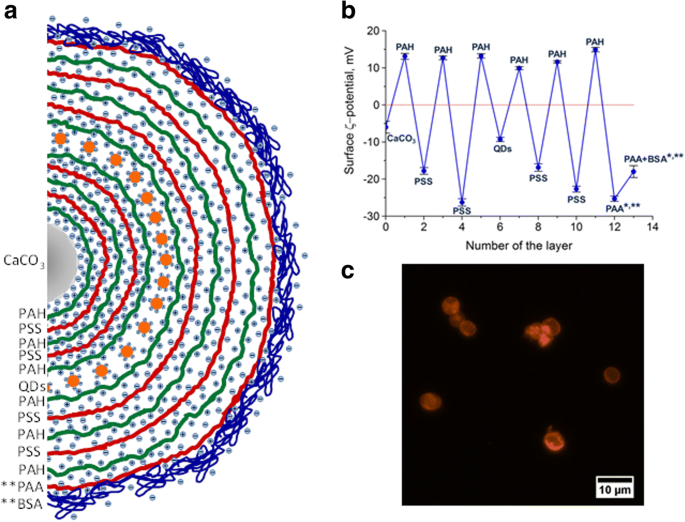
The design and structure of the polyelectrolyte microcapsules encoded with quantum dots (QDs): a Schematic of layer arrangement. b ζ‑potential changes during assembly and QD incorporation. c Fluorescent microphotograph of PEGylated QD‑encoded microcapsules. * ζ‑potentials after core removal; ** after BSA coating.
Scanning electron microscopy of cross‑sections confirms the hollow architecture and uniform distribution of QDs within the membrane (Figure 5).
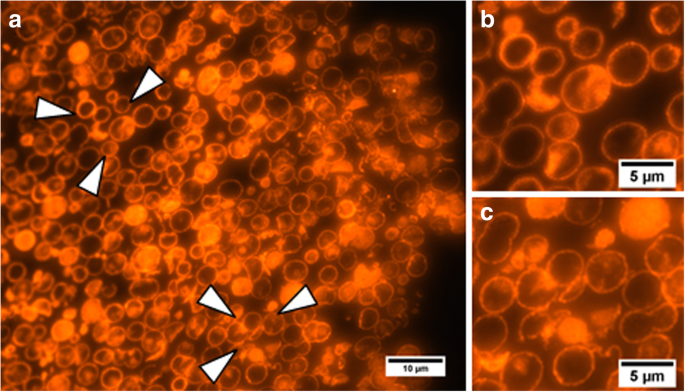
Microphotographs of sections of QD‑encoded microcapsules. Arrows in (a) point to the areas magnified in (b) and (c).
Dynamic light scattering shows a narrow size distribution with a mean diameter of 4.45 ± 0.65 µm (Figure 2), comparable to red blood cells and suitable for intravenous delivery without breaching the blood‑tissue barrier. In regions of enhanced permeability (e.g., tumors), these capsules can extravasate, enabling targeted imaging.
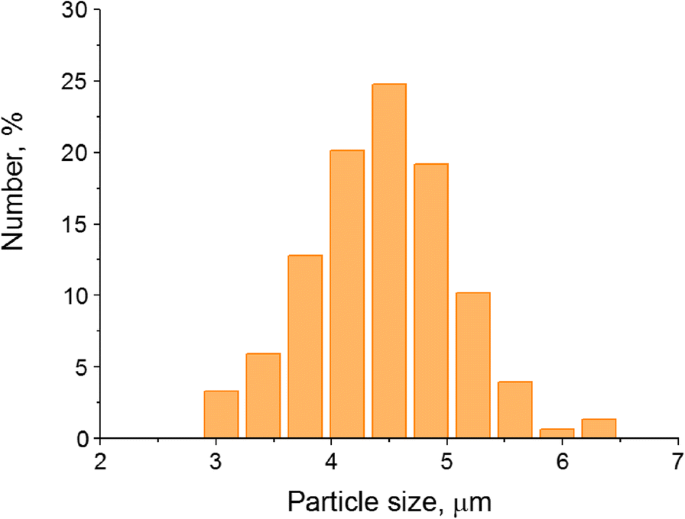
Size distribution of the polyelectrolyte microcapsules optically encoded with quantum dots (n = 600).
Fluorescence and Structural Characteristics
Emission spectra of the encapsulated capsules retain the 590 nm peak of the free QDs (Figure 3), indicating that the polyelectrolyte matrix does not quench fluorescence. Flow cytometry reveals a clear separation between QD‑encoded (orange) and placebo (gray) capsules in the PE channel, with fluorescence intensity ~10⁴ PE units, while FSC/SSC distributions overlap, confirming identical size and granularity (Figure 4).
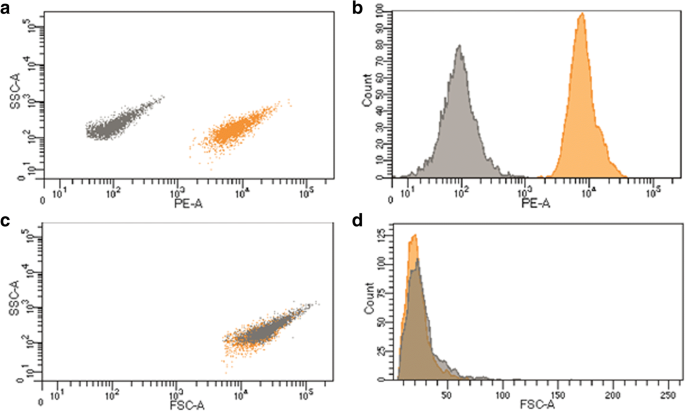
Detectability of QD‑encoded microcapsules by flow cytometry: a Dot‑plot (SSC‑PE). b PE histogram. c Dot‑plot (SSC‑FSC). d FSC histogram. 2500 events per sample.
The high photostability of QDs, combined with a quantum yield of ~40 %, yields a brightness that matches or exceeds that of conventional organic dyes while avoiding photobleaching. The ability to encode multiple colors by size tuning offers virtually limitless spectral multiplexing.
Conclusions
We have demonstrated a reproducible, solvent‑free fabrication route for PEGylated QD‑encoded polyelectrolyte microcapsules with uniform size, stable surface charge, and bright fluorescence. The capsules are readily detectable by standard flow cytometers and confocal microscopes, making them strong candidates for in vitro and in vivo bioimaging. Future work will explore stimulus‑responsive functionalization and integration into theranostic platforms.
Abbreviations
- BSA:
Bovine serum albumin
- EDTA:
Disodium ethylenediaminetetraacetate
- PAA:
Polyacrylic acid
- PAH:
Polycation poly(allylamine hydrochloride)
- PEG:
Polyethylene glycol
- PSS:
Polyanion poly(sodium 4‑styrenesulfonate)
- QD:
Quantum dot
- TOPO:
Trioctylphosphine oxide
Nanomaterials
- Semiconductor Nanoparticles: Properties, Synthesis, and Applications
- Synergistic Electrical Enhancement in Epoxy Composites with Carbon Nanotubes and Graphite Nanoplatelets
- Optimizing ZnO Film Morphology and Properties via Interfacial Layer Design
- Engineering Fluorescent Polyelectrolyte Microcapsules with Quantum Dot Encoding for Advanced Theranostic Applications
- GeSiSn Thin Films with Nanoislands: Strain‑Driven Morphology, Superstructure Control, and Mid‑IR Photoluminescence
- How Nanoparticle Size and Agglomeration Influence Interfacial Strength and Tensile Performance in Polymer Nanocomposites
- Hydrothermal Synthesis of 19 nm Sc₂O₃:Er³⁺,Yb³⁺ Nanoparticles with 4× Superior Red Upconversion Luminescence
- Hierarchical ZSM‑5 Zeolites with Exceptional Mesoporosity and Catalytic Performance: A Novel Soft‑Template Approach
- Antimicrobial Performance of PBAT Nanocomposites and Composites Incorporating Copper, Cu/Cu₂O Nanoparticles, and Copper Sulfate
- Developing a Precise Lysosomal pH Sensor Using Self-Decomposing SiO2 Nanoparticles



