Enhanced Gene Delivery Using Gold Nanoparticles Functionalized with Chitosan, N‑Acylated Chitosan, and Chitosan Oligosaccharide
Abstract
Gold nanoparticles (AuNPs) are increasingly used in biomedical research due to their unique optical properties, high density, and ease of surface modification. Here, we present a non‑viral gene delivery platform that combines AuNPs with three chitosan‑derived polymers: native chitosan (CO), N‑acylated chitosan (Acyl‑CO), and chitosan oligosaccharide (COS). Two synthetic routes were explored: a conventional chemical reduction with sodium borohydride and a green one‑pot synthesis where COS simultaneously reduces and stabilizes AuNPs. The resulting nanocomposites were fully characterized by UV‑Vis spectroscopy, ζ‑potential, FTIR, and transmission electron microscopy (TEM). Agarose gel electrophoresis confirmed robust plasmid DNA (pDNA) complexation, and transfection efficiency in HEK‑293 cells was evaluated via β‑galactosidase activity and EGFP fluorescence. Transfection rates of 27 %, 33 %, and 60 % were achieved with CO‑AuNPs, Acyl‑CO‑AuNPs, and COS‑AuNPs, respectively, demonstrating that the green, COS‑based formulation offers superior gene delivery performance while avoiding toxic reducing agents.
Background
Gene therapy relies on the delivery of therapeutic genetic material into target cells. While viral vectors provide high transfection efficiencies, they suffer from immunogenicity, limited payload capacity, and production complexity. Non‑viral carriers, in contrast, are safer, easier to produce, and more stable during storage. Chitosan, a naturally occurring polysaccharide derived from chitin, has emerged as a promising non‑viral vector because of its positive charge, low toxicity, and ability to protect DNA from enzymatic degradation. However, native chitosan is poorly soluble at physiological pH and highly viscous in dilute acid solutions, limiting its biomedical application. Chemical modifications such as N‑acylation increase hydrophobicity and tune the degree of deacetylation, while oligomerization (COS) reduces viscosity and enhances cellular uptake due to shorter chain lengths.
Gold nanoparticles, with their tunable size, surface plasmon resonance, and strong affinity for amine, thiol, and cyanide groups, are ideal candidates for building multifunctional nanocarriers. Cationic polymers like chitosan can be grafted onto AuNPs to create positively charged surfaces that electrostatically bind plasmid DNA, facilitating cellular internalization. Moreover, the high density of AuNPs promotes rapid cellular uptake, and their optical properties enable real‑time monitoring of delivery.
Previous studies have demonstrated that chitosan‑AuNP conjugates can achieve higher transfection efficiencies than liposomes or polymeric nanoparticles. Yet, most syntheses rely on toxic reducing agents such as sodium borohydride or cetyltrimethylammonium bromide (CTAB). In this work, we introduce a green synthesis route that uses COS as both reducing and stabilizing agent, eliminating hazardous chemicals while preserving the favorable physicochemical properties of the nanocomplexes.
Methods
Nanocomposite Synthesis
Three distinct AuNP formulations were prepared (Fig. 2). CO‑AuNPs and Acyl‑CO‑AuNPs were synthesized via chemical reduction with NaBH₄, while COS‑AuNPs were produced by a green one‑pot method where COS simultaneously reduces Au³⁺ and caps the nanoparticles.
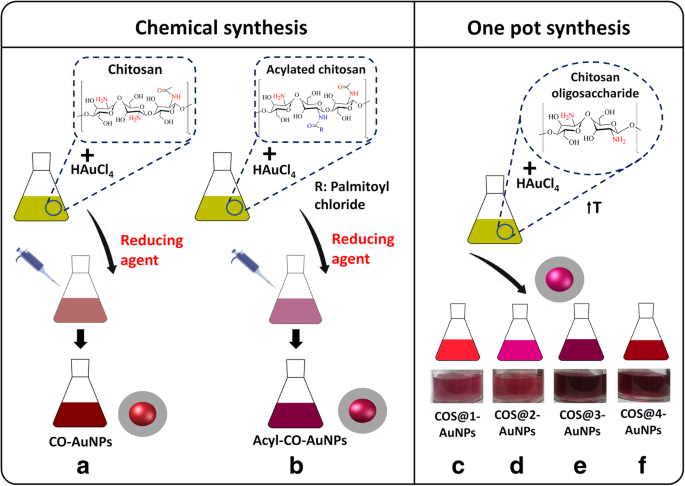
Schematic diagram for each synthesis: a–b chemical synthesis for CO‑AuNPs and Acyl‑CO‑AuNPs; c–f green synthesis for COS‑AuNPs.
Materials
Gold(III) chloride trihydrate (HAuCl₄·3H₂O), sodium borohydride (NaBH₄), low‑molecular‑weight chitosan (50–190 kDa, 75 % deacetylation), and COS (5 kDa) were purchased from Sigma‑Aldrich. All glassware was cleaned with aqua regia for 24 h and rinsed with Milli‑Q water to remove metal traces.
CO‑AuNPs
CO (2 mg mL⁻¹) was dissolved in 1 % acetic acid and stored overnight. The solution was filtered (0.22 µm) and mixed with 10 mM HAuCl₄ (1 mL) under vigorous stirring for 30 min. NaBH₄ (100 mM, 0.4 mL) was added dropwise; the color changed from yellow to wine‑red, indicating nanoparticle formation. Stirring continued for 2 h.
Acyl‑CO‑AuNPs
Acylation of low‑molecular‑weight chitosan was performed with caproyl chloride following a modified protocol. The resulting gel was precipitated with acetone, washed with methanol, and dried. The gel (0.33 % in 0.1 M HCl) was mixed with 10 mM HAuCl₄ (1 mL) and stirred for 1 h. NaBH₄ (100 mM, 0.4 mL) was then added, producing a pink‑red solution that turned fully colored within 2 h of stirring.
COS‑AuNPs
For the green synthesis, COS (100 – 200 mg) was added to HAuCl₄ (0.003 – 0.017 wt %) and stirred at 80 °C for 60 min. Four variants were prepared, differing in COS amount and gold concentration (Table 1). Nanoparticles were purified by centrifugation (12 000 g, 30 min).
Characterization
FTIR
FTIR spectra (Perkin Elmer Spectrum One) confirmed the presence of amide bands and the successful acylation of Acyl‑CO. The degree of substitution (DS) was calculated as 75 % using the ratio of absorbances at 1651 cm⁻¹ and 3350 cm⁻¹.
ζ‑Potential
Zeta potential measurements (Malvern Nano Z) revealed positive surface charges (43–55 mV) for all composites, confirming their suitability for DNA binding. Post‑complexation values dropped, indicating successful plasmid adsorption.
UV‑Vis
UV‑Vis spectra (400–700 nm) displayed localized surface plasmon resonance (LSPR) peaks at 534 nm (CO‑AuNPs), 507 nm (Acyl‑CO‑AuNPs), and 533–536 nm (COS‑AuNPs). Spectra remained unchanged after 150 days, evidencing colloidal stability.
TEM
TEM images (JEOL JEM‑1010) revealed spherical nanoparticles with diameters ranging from 3 to 15 nm. Size distributions were narrower for COS‑AuNPs, reflecting superior monodispersity.
Complex Formation and DNA Binding
Plasmids pSV‑β‑Gal (6.82 kb) and pIRES2‑EGFP (5.3 kb) were purified via alkaline lysis. DNA (200–400 ng) was mixed with each nanocomposite in DMEM, and agarose gel electrophoresis (0.8 % agarose, 90 V, 60 min) demonstrated complete DNA retention in the wells, confirming electrostatic complexation.
Cell Transfection
Cell Culture
HEK‑293 cells were seeded at 12 000 cells per well in 96‑well plates, reaching ~90 % confluence after 24 h.
EGFP Transfection
Complexes (15 µL, 200 ng DNA) were added to cells for 2 h, followed by 48 h incubation in complete DMEM. GFP expression was quantified by fluorescence microscopy (Carl Zeiss Axio Vert.A1).
β‑Galactosidase Transfection
After 24 h incubation, cells were fixed and incubated with X‑gal solution. Blue staining, indicative of β‑galactosidase activity, was observed by bright‑field microscopy (Motic AE2000). LipofectAMINE™ 2000 served as a positive control.
Cell Viability
Trypan blue exclusion (0.2 %) assessed viability 2 h post‑transfection. Concentration ranges were 0.1–3 mM for CO‑AuNPs and Acyl‑CO‑AuNPs, 0.5 mM for COS@3‑AuNPs and COS@4‑AuNPs, and 0.1 mM for COS@1‑AuNPs and COS@2‑AuNPs.
Results and Discussions
Nanocomposite Synthesis and Characterization
The green COS‑AuNPs were produced without hazardous reagents, demonstrating the feasibility of a fully biocompatible synthesis route. UV‑Vis spectra (Fig. 3) confirmed the formation of AuNPs, with LSPR peaks consistent across all formulations. Stability tests after 150 days showed no spectral shift, indicating excellent colloidal robustness.
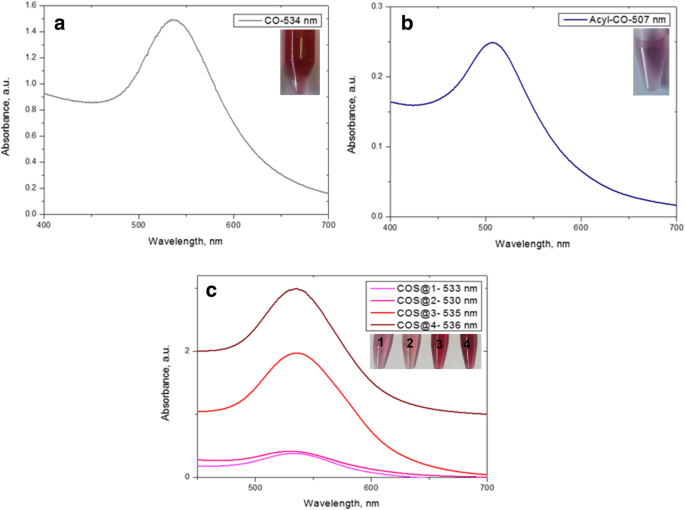
UV‑Vis spectra of the nanocomposites. LSPR maxima: CO‑AuNPs 534 nm, Acyl‑CO‑AuNPs 507 nm, COS‑AuNPs 533–536 nm.
Stability
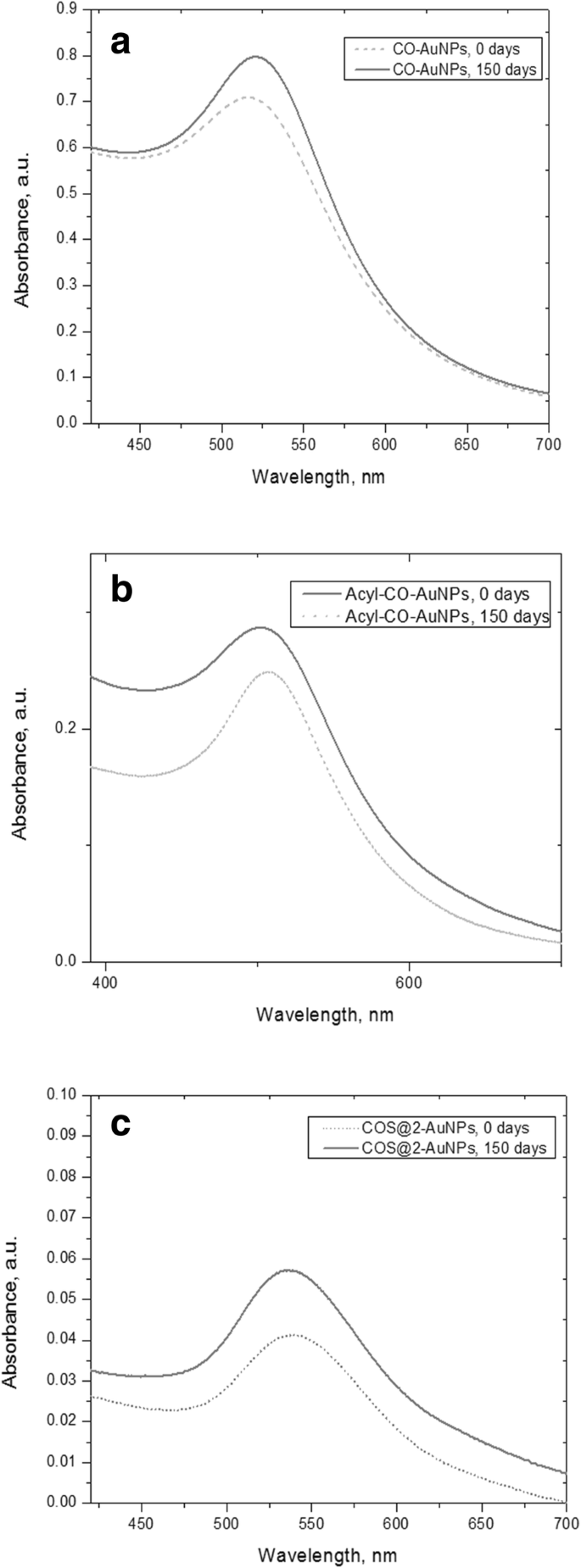
UV‑Vis spectra after 150 days: CO‑AuNPs (a), Acyl‑CO‑AuNPs (b), COS‑AuNPs (c).
FTIR
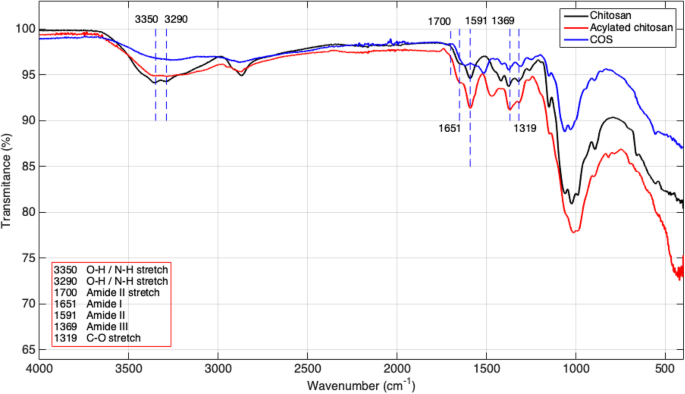
FT‑IR spectra of chitosan, Acyl‑chitosan, and COS. Key bands: 1651 cm⁻¹ (amide I), 1591 cm⁻¹ (amide II).
ζ‑Potential
Table 2 lists the ζ‑potential values: CO‑AuNPs 43.3 mV, Acyl‑CO‑AuNPs 40.2 mV, COS‑AuNPs 46.3–55.3 mV. All values remained positive after DNA binding, confirming complexation.
TEM
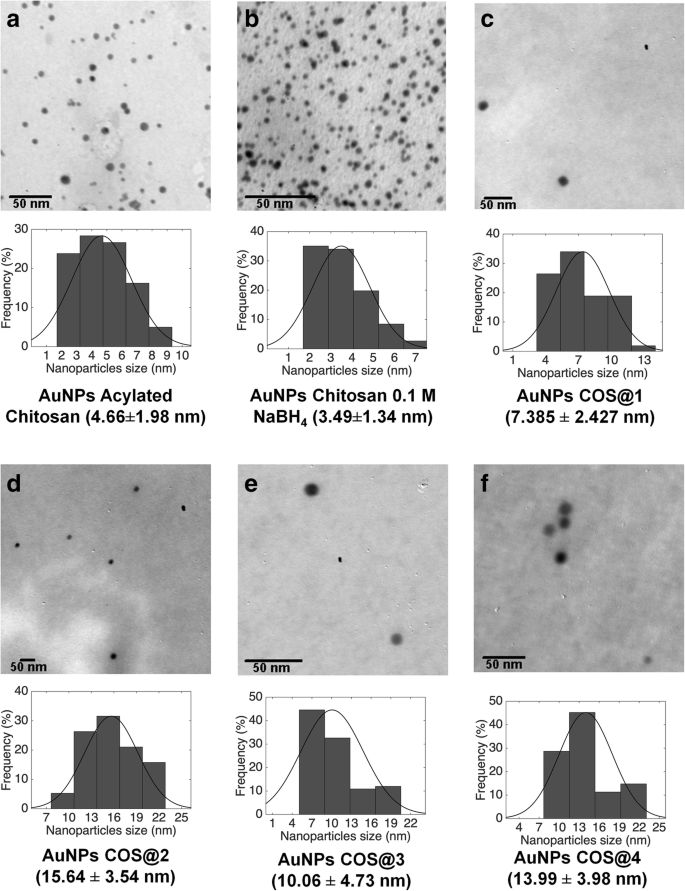
TEM micrographs: (a) Acyl‑CO‑AuNPs 4.7 nm, (b) CO‑AuNPs 3.5 nm, (c‑f) COS‑AuNPs 7.4–15.6 nm.
DNA Complexation
Agarose gels (Fig. 7) showed complete DNA retention in all nanocomplexes, whereas free plasmid migrated to the bottom. COS‑AuNPs displayed the strongest binding, retaining up to 350 ng of plasmid before saturation.
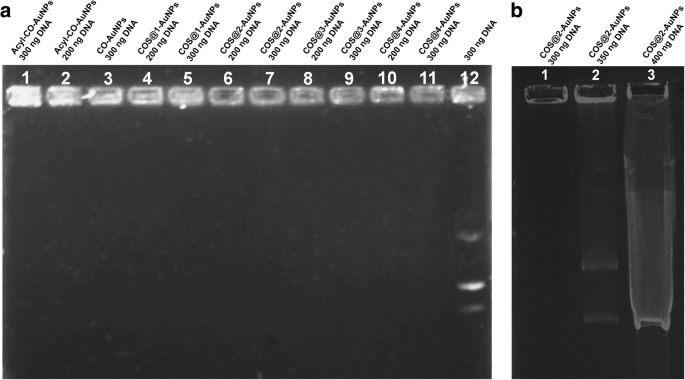
Agarose gel electrophoresis of pDNA complexes.
Transfection Efficiency
In HEK‑293 cells, COS‑AuNPs achieved the highest transfection rates: 60 % with β‑galactosidase and 48 % with EGFP. Acyl‑CO‑AuNPs and CO‑AuNPs reached 33 % and 27 % respectively. The superior performance of COS‑AuNPs is attributed to their higher chitosan/gold ratio and optimal particle size.
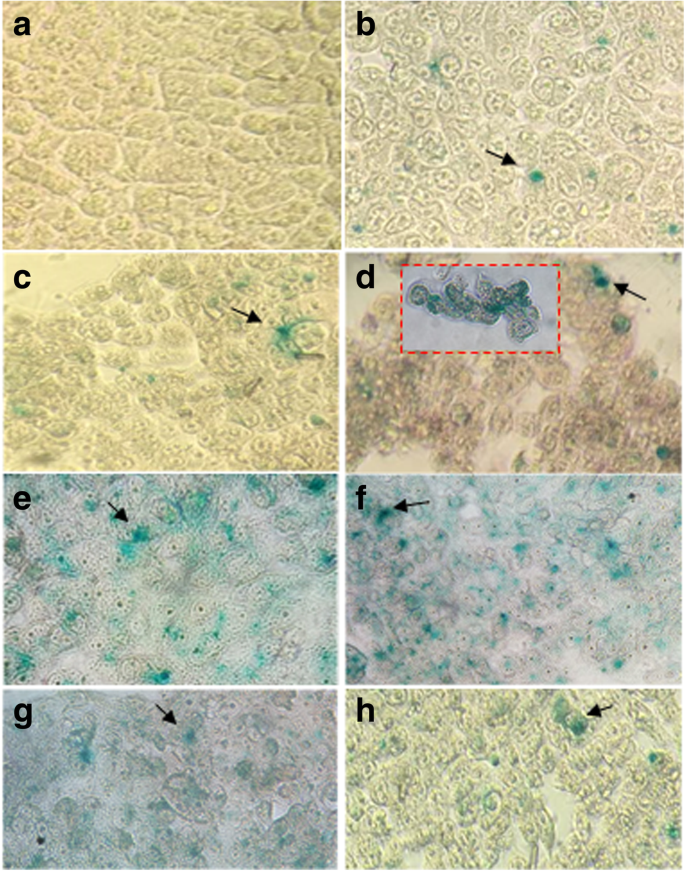
Transfection assays in HEK‑293 cells.
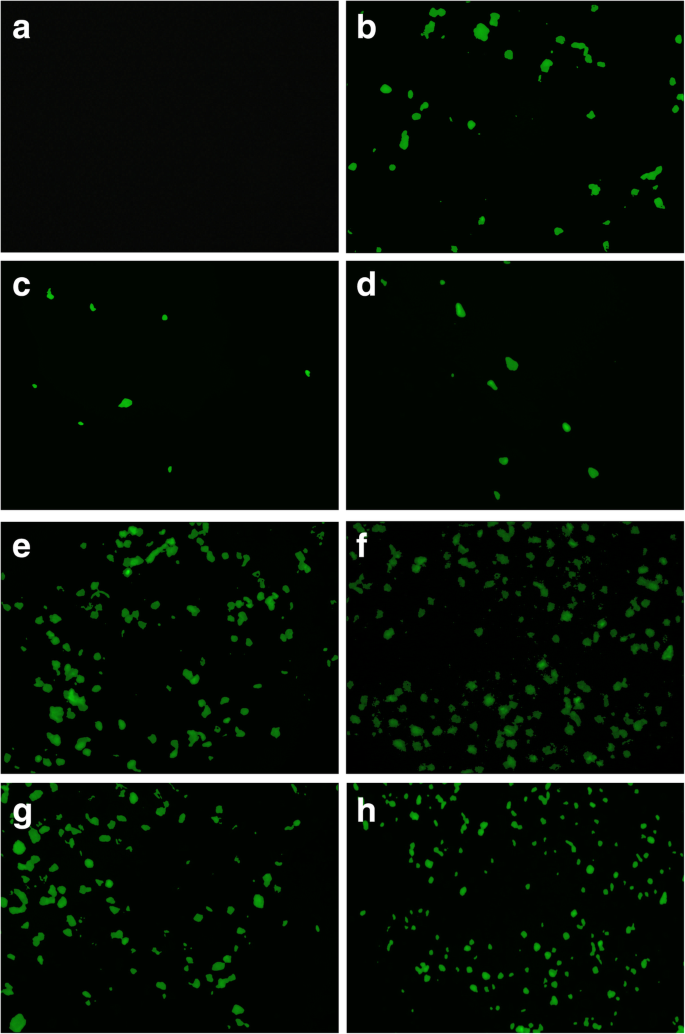
EGFP fluorescence imaging.
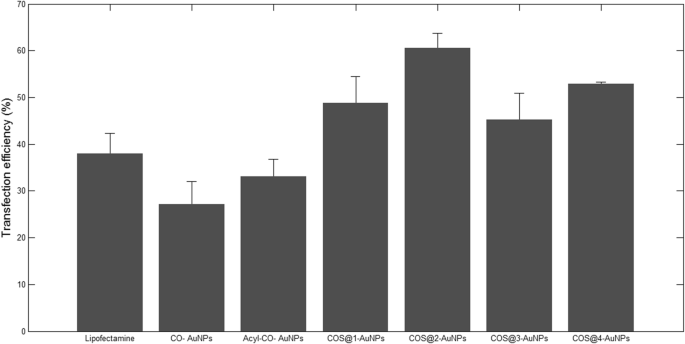
Transfection efficiency percentages.
Cell Viability
Trypan blue exclusion revealed high viability (>90 %) for COS‑AuNPs, whereas LipofectAMINE™ 2000 and CO‑AuNPs showed lower survival rates (61 % and 78 %). COS‑AuNPs therefore combine high transfection efficiency with minimal cytotoxicity.
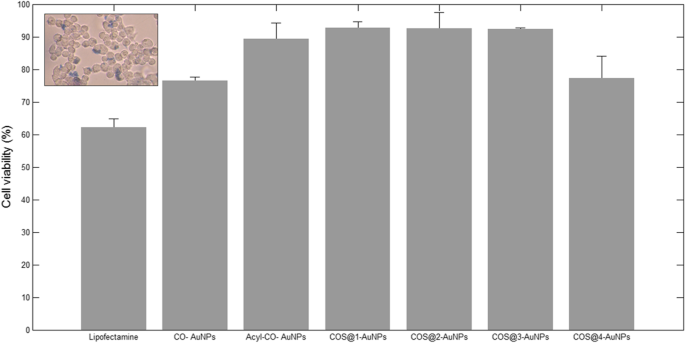
Cell viability percentages.
Conclusions
We have successfully fabricated gold nanoparticle–chitosan conjugates via both conventional and green routes. The COS‑AuNPs, synthesized without toxic reagents, displayed superior transfection efficiency (up to 60 %) and excellent cytocompatibility (>93 %). Their narrow size distribution and positive surface charge enable robust plasmid binding and efficient cellular uptake. These findings position COS‑AuNPs as a promising, environmentally friendly gene delivery platform suitable for further preclinical development.
Availability of Data and Materials
All datasets supporting the conclusions of this study are included within the main text.
Abbreviations
- Acyl-CO:
Acylated chitosan
- AuNPs:
Gold nanoparticles
- CO:
Chitosan
- COS:
Chitosan oligosaccharide
- DMEM:
Dulbecco’s modified Eagle’s medium
- FTIR:
Fourier‑transform infrared
- PBS:
Phosphate‑buffered saline
- pDNA:
Plasmid deoxyribonucleic acid
- TEM:
Transmission electron microscopy
Nanomaterials
- Plasmonic Nanoparticles: Harnessing Surface Plasmons for Advanced Photothermal Applications
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
- Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Al₂O₃ and SiO₂ Nanoparticles with Ultrasound Significantly Reduce Water Supercooling
- Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
- Shape-Dependent Cytotoxicity & Cellular Uptake of Green Tea‑Reduced Gold Nanoparticles in Cancer Cells
- Biotin‑Functionalized Redox‑Sensitive Chitosan Nanoparticles Coated with Phycocyanin for Targeted Curcumin Delivery



