Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
Abstract
We synthesized an amphiphilic hyperbranched polyglycerol (HPG) modified with dodecanethiol (DS) via thiol‑ene click chemistry, producing DSHPG. Characterization by NMR, FTIR, and GPC confirmed successful modification, while DSC and TGA revealed enhanced thermal stability. Gold nanoparticles (Au NPs) were synthesized using DSHPG as both stabilizer and surface modifier. By varying the HPG molecular weight, we tuned NP diameters: 4.1 nm (Au@DSHPG‑1, HPG Mn = 1123), 9.7 nm (Au@DSHPG‑2, HPG Mn = 3826), and 15.1 nm (Au@DSHPG‑3, HPG Mn = 55 075). Transmission electron microscopy (TEM) and dynamic light scattering (DLS) confirmed these sizes. DSHPG’s long alkyl chains imparted excellent dispersion in nonpolar solvents; hydrocarbon nanofluids were prepared by dispersing the Au NPs into a series of hydrocarbons. UV‑Vis spectroscopy monitored dispersion stability, showing that over 80 % of the Au NPs remained suspended after 3 600 h.
Background
Endothermic hydrocarbon fuels are crucial in aerospace for their high energy density and regenerative cooling potential. Catalytic cracking, typically promoted by noble metal nanoparticles (NPs) such as Pt, Pd, and Au, enhances fuel conversion and heat sink properties. Prior studies have shown Pt and Pd NPs lower the cracking onset temperature of JP‑10 from 650 °C to 600 °C, while Pd NPs in decalin and aviation kerosene increase conversion, gas yield, and heat sink. Our earlier work demonstrated that Au NPs dispersed in JP‑10 exhibit higher catalytic activity than the pure fuel. However, maintaining a stable solid‑liquid suspension of NPs in hydrocarbons remains challenging.
Coating NPs with protective layers is a proven strategy to improve stability. Hyperbranched polymers, particularly hyperbranched polyglycerol (HPG), offer a globular architecture with abundant hydroxyl termini, making them ideal for hosting and stabilizing metal NPs. HPG has been successfully employed to synthesize a variety of inorganic nanocrystals with excellent biocompatibility and to act as initiators for hydrocarbon cracking. Modifying HPG with thioether groups—via thiol‑ene click chemistry—introduces soft‑metal binding sites that can further stabilize metal NPs, yet few examples of thioether‑substituted hyperbranched polymers exist.
In this work, we synthesized amphiphilic DSHPG, leveraging its long alkyl chains for solubility in hydrocarbon fuels, and used it to prepare Au@DSHPG. We systematically investigated how the HPG molecular weight influences Au NP size and evaluated dispersion stability in various solvents and hydrocarbon mixtures.
Methods
Materials
All reagents were purchased from commercial suppliers: glycidol, potassium methylate, 1,1,1‑tris(hydroxymethyl)propane (TMP), 4‑(dimethylamino)pyridine, 1,4‑dioxane, glycidyl methacrylate, benzophenone, chloroauric acid, tetraoctylammonium bromide, sodium borohydride, and various organic solvents (toluene, methanol, chloroform, diethyl ether, dichloromethane, DMSO, hexane, cyclohexane, decahydronaphthalene, 1‑dodecane, octane, 1‑tetradecane). HPG, HPG‑MA, DSHPG, and Au@DSHPG were synthesized in-house following Scheme 1.
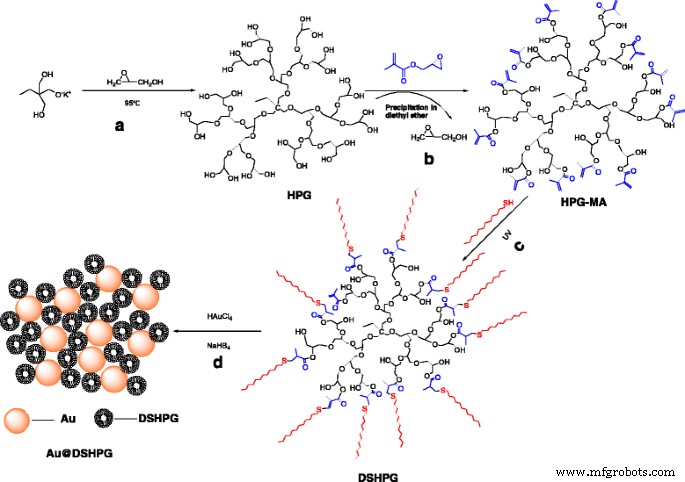
Preparation of HPG, HPG‑MA, DSHPG, and Au@DSHPG
Preparation of HPG
HPG was prepared by ring‑opening anionic polymerization of glycerol, initiated by TMP and potassium methylate. The monomer/initiator ratio and the addition of 1,4‑dioxane controlled the polymer’s number‑average molecular weight (Mn). The product was neutralized on a cation‑exchange resin, precipitated twice in acetone, and dried under vacuum at 85 °C.
Preparation of Methacrylated HPG‑MA
HPG (1 g) was reacted with glycidyl methacrylate (10 mL) in DMSO under nitrogen, catalyzed by 4‑(dimethylamino)pyridine. After 24 h, the product was precipitated in diethyl ether and dried overnight.
Preparation of DSHPG by Click Chemistry
DSHPG was synthesized by dissolving HPG‑MA in chloroform, adding 1‑dodecanethiol (2 mL) and benzophenone, and irradiating with UV for 1 h. The polymer was purified by chloroform dialysis and dried.
Preparation of Au@DSHPG
Au NPs were generated by reducing aqueous HAuCl4 with NaBH4 in toluene in the presence of DSHPG. After stirring, the mixture was allowed to rest for 48 h to ensure full transfer of Au into the polymer shell, then the solvent was evaporated, and the brown solid was dried under vacuum.
Characterization
Structural confirmation was performed by 1H and 13C NMR (Bruker 400 MHz) and FTIR (Nicolet iS10). GPC (Waters) determined Mn and dispersity. Thermal properties were measured by DSC (TA Q2000) and TGA (TA Q50). Morphology and size were examined by TEM (Philips CM‑200) and DLS (Malvern ZEN 3600). Stability of nanofluids was monitored by UV‑Vis spectroscopy (Shimadzu UV‑1750) using 1.0 cm quartz cells.
Results and Discussion
Molecular Structures of HPG, HPG‑MA, and DSHPG
1H and 13C NMR spectra (Fig. 1) confirm successful methacrylation and subsequent thiol‑ene addition. The disappearance of vinyl signals and appearance of dodecyl resonances in DSHPG demonstrate complete conversion. FTIR spectra (Fig. 2) show characteristic C=C stretching at 1562 cm‑1 for HPG‑MA and its reduction in DSHPG, further confirming the click reaction.
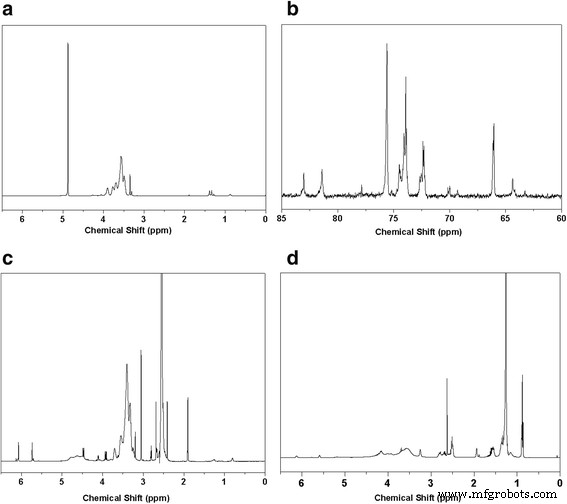
NMR characterizations of HPG, HPG‑MA, and DSHPG
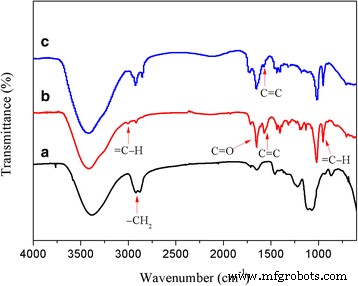
FTIR spectra of HPG, HPG‑MA, and DSHPG
Thermal Behaviors
DSC analysis (Fig. 3) shows Tg values of −37.5 °C for HPG, −20.8 °C for HPG‑MA, and a melting peak at −4.7 °C for DSHPG, indicating crystallization of the long alkyl chains. The Au@DSHPG sample lacks these peaks, suggesting that NP incorporation disrupts chain packing. TGA (Fig. 4) reveals a primary weight loss near 400 °C for Au@DSHPG, attributed to HPG core degradation, while higher‑temperature losses correspond to DSHPG shell decomposition. The Au content in Au@DSHPG‑2 is 27.2 wt %.
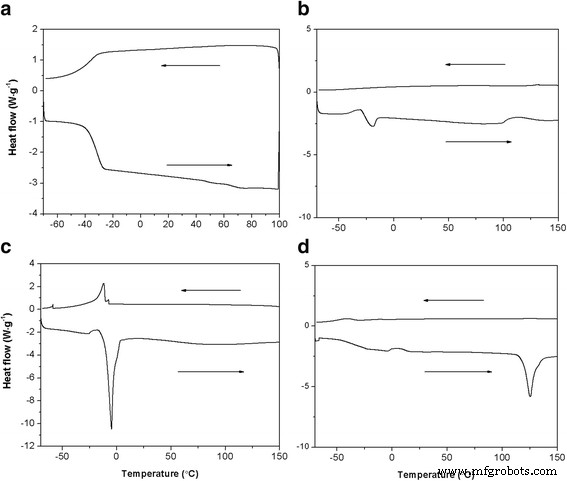
DSC curves for HPG, HPG‑MA, DSHPG, and Au@DSHPG‑2
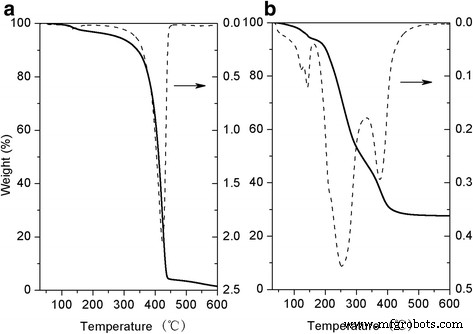
TGA and DTG curves for HPG and Au@DSHPG‑2
Size Control of Au@DSHPG
By adjusting the HPG Mn from 1 123 to 55 075 (Table 1), we achieved precise control over Au NP diameter: 4.1 nm (Au@DSHPG‑1), 9.7 nm (Au@DSHPG‑2), and 15.1 nm (Au@DSHPG‑3). TEM images (Fig. 5) confirm these sizes, and DLS data corroborate the trend. The relationship demonstrates that larger HPG cores provide a more spacious encapsulation environment, enabling growth of larger NPs. The multi‑polymer stabilization model—where multiple HPG chains coordinate a single NP—explains the observed size dependence.
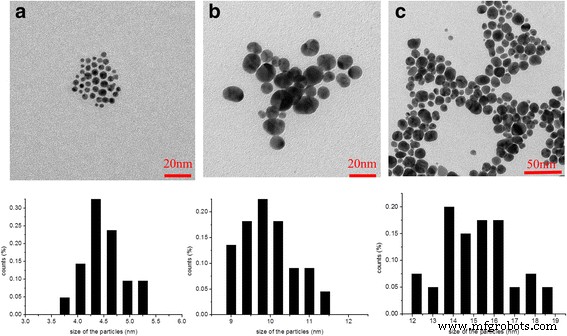
TEM image and size distribution of Au@DSHPG (a: Au@DSHPG‑1, b: Au@DSHPG‑2, c: Au@DSHPG‑3)
Dispersion Ability in Solvents
UV‑Vis spectroscopy of saturated Au@DSHPG‑2 solutions (Fig. 6) shows superior dispersion in nonpolar solvents (toluene, diethyl ether) compared to polar solvents (acetonitrile, water). Mixing toluene with acetonitrile in varying ratios further demonstrates that decreasing solvent polarity enhances NP dispersion (Fig. 7–8). Solubility coefficient calculations (Eq. 1) predict the best miscibility with toluene, corroborating experimental observations. These results confirm that DSHPG confers strong lipophilicity, enabling Au NP stability in hydrocarbon environments.
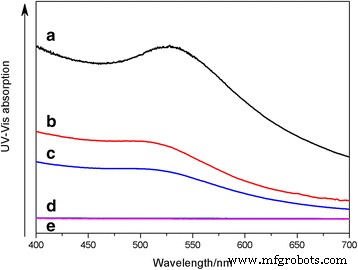
UV‑Vis absorption of saturated Au@DSHPG‑2 solutions (a: toluene, b: diethyl ether, c: ethyl acetate, d: acetonitrile, e: water)
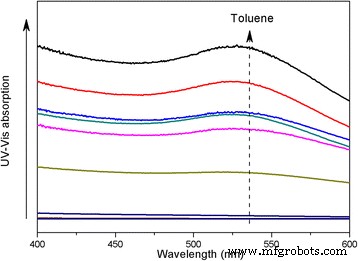
UV‑Vis absorption of Au@DSHPG‑2 in toluene/acetonitrile mixtures
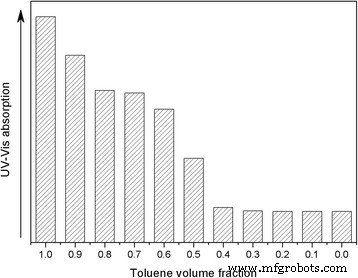
Absorbance at 521 nm vs. toluene fraction
Long‑Term Stability in Hydrocarbons
Au@DSHPG‑2 was dispersed in hexane, cyclohexane, octane, decalin, dodecane, and tetradecane. UV‑Vis monitoring over 3 600 h (Fig. 9) shows that relative NP concentration remains above 80 % in all hydrocarbons, with minor declines only in dodecane and tetradecane. The DSHPG shell slowly swells and releases NPs, but the process is sluggish, resulting in exceptional long‑term stability.
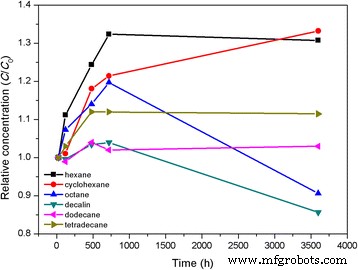
Relative NP concentration in hydrocarbons over 3 600 h
Conclusions
We demonstrated that hyperbranched polyglycerol with tunable Mn can be functionalized with dodecanethiol to produce DSHPG, which effectively stabilizes Au NPs and allows precise size control via core molecular weight. DSHPG‑modified Au NPs exhibit excellent dispersion in nonpolar solvents and maintain high stability (> 80 %) in a range of hydrocarbon fuels for over 3 600 h. These hydrocarbon‑based nanofluids hold promise for enhancing heat sink performance in catalytic cracking processes.
Nanomaterials
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- FePO4 Nanoparticles: Biocompatible Carriers for Doxorubicin Delivery, RNA Stabilization, and Enhanced Anticancer Activity
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Green Synthesis of Gold and Silver Nanoparticles with Platycodon Saponins from Platycodi Radix
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Green Synthesis of Gold Nanoparticles from Artemisia capillaris, Portulaca oleracea, and Prunella vulgaris Extracts: Antioxidant Activity and Cytotoxicity Evaluation
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy



