FePO4 Nanoparticles: Biocompatible Carriers for Doxorubicin Delivery, RNA Stabilization, and Enhanced Anticancer Activity
FePO4 nanoparticles combine biocompatibility, high bioavailability, magnetic responsiveness, and superior sensory properties, making them ideal for food fortification, biomedical imaging, and drug delivery. In this study, we engineered FePO4 as a carrier for the anticancer agent doxorubicin (DOX), achieving an optimal drug loading of 26.81 ± 1.0 %. The resulting FePO4-DOX particles exhibited uniform size distribution, concentration‑dependent biocompatibility (>70 % viability up to 80 µg/mL), and a dramatic 10‑fold increase in cytotoxicity compared to free DOX, with enhanced selectivity for cancer cells. Moreover, FePO4 nanoparticles stabilized RNA and preserved mRNA translation activity, underscoring their potential as multifunctional delivery platforms for therapeutic nucleic acids. These findings highlight the versatility of FePO4 particles for safe drug and RNA delivery, and their promise in food fortification applications. Among inorganic nanomaterials, iron‑based nanoparticles (Fe‑NPs) stand out for their magnetic properties, endogenous metabolism, and FDA‑approved applications in contrast imaging, drug delivery, and hyperthermia. FePO4 nanoparticles, in particular, exhibit excellent biocompatibility, high oral bioavailability, and minimal organoleptic impact, making them attractive for iron‑deficiency anemia prevention through food fortification. Their uptake in intestinal cells occurs primarily via divalent metal transporter‑1 (DMT‑1), ensuring efficient absorption. Beyond imaging, FePO4 offers a chemically simple, scalable synthesis route that yields monodisperse particles with controlled surface charge, enabling robust drug loading and nucleic acid stabilization. This work explores FePO4 as a drug‑delivery vehicle for DOX and as a stabilizer for RNA, aiming to demonstrate a single platform that addresses both therapeutic and nutritional needs. FePO4 was synthesized via a straightforward co‑precipitation of ammonium phosphate and iron(III) nitrate, followed by dispersion in DSPE‑PEG‑COOH to prevent aggregation. Dynamic light scattering revealed a hydrodynamic diameter of 175 ± 5 nm with a polydispersity index of 0.15 ± 0.01, indicating excellent size homogeneity. Zeta potential measurements showed a negative surface charge of −19.1 ± 8 mV, which reduces protein opsonization and prolongs circulation time. FTIR spectra confirmed the presence of PO43− groups and absence of precursor residues, verifying product purity. MTT assays on mouse osteosarcoma K7M2 and fibroblast NIH/3T3 cells demonstrated concentration‑dependent viability, with >70 % cell survival up to 80 µg/mL, confirming the biocompatibility of FePO4 NPs within a therapeutically relevant window. Characterization of FePO4 nanoparticles: a Size distribution; b Zeta potential; c FTIR spectra; d Biocompatibility across cell lines (mean ± s.d.; n = 3–6). DOX was incorporated via co‑incubation with Fe(NO3)3 followed by precipitation with (NH4)3PO4, yielding a loading efficiency of 26.81 ± 1.0 %. The Fe3+–DOX complex forms during this step, as confirmed by a color shift from yellow to brown and unchanged fluorescence emission (590 nm). Hydrodynamic size remained 187 ± 7 nm, while the surface charge shifted to −8.89 ± 5 mV, reflecting the cationic nature of the bound DOX. Cytotoxicity assays revealed a 10‑fold reduction in IC50 values compared to free DOX (NIH/3T3: 0.248 µM vs. 2.61 µM; K7M2: 0.107 µM vs. 1.01 µM). Importantly, the FePO4-DOX nanoparticles maintained >70 % viability at the equivalent FePO4 concentrations, indicating that the enhanced cytotoxicity arises from Fe3+-DOX complexation rather than nanoparticle toxicity. The selective killing of cancer cells over fibroblasts was also amplified, with a 19 % difference in viability at 0.1 µM DOX equivalent. Doxorubicin loading efficiency and physicochemical changes in FePO4-DOX: a Loading efficiency across formulations; b Color transition; c Emission spectra; d Size distribution; e Zeta potential (mean ± s.d.; n = 3). Cytotoxicity of FePO4-DOX versus free DOX in NIH/3T3 and K7M2 cells: a,b Cell viability after 48 h; c,d IC50 comparison (mean ± s.d.; n = 6). Confocal microscopy of K7M2 cells revealed negligible uptake at 0.5–1 h, followed by pronounced intracellular fluorescence at 3 h, comparable to free DOX. The time‑dependent internalization profile demonstrates that FePO4-DOX nanoparticles can effectively deliver DOX into cancer cells within a clinically relevant timeframe, while the magnetic properties of FePO4 enable potential theranostic applications. Time‑course internalization of FePO4-DOX versus free DOX in K7M2 cells (0.5, 1, 3 h). Red fluorescence indicates successful nanoparticle uptake. FePO4 nanoparticles protect model RNA from hydrolysis and nucleases, as evidenced by intact bands on RNA agarose gel electrophoresis (RAGE) after incubation in water and serum. In contrast, copper and carbon nanotube nanoparticles accelerated degradation. Functional assays using luciferase‑encoding mRNA demonstrated that FePO4 preserved translation efficiency, delivering a two‑fold increase in luminescence relative to control, whereas copper nanoparticles reduced expression. These results underscore FePO4 as a robust RNA stabilizer that maintains biological activity, positioning it as a candidate for mRNA‑based therapeutics and vaccines. RNA stabilization and translation: a Degradation assay in water; b Stability in serum; c Luciferase expression with FePO4 versus copper nanoparticles. FePO4 nanoparticles, synthesized via a simple co‑precipitation and PEGylation strategy, form monodisperse particles (~175 nm) with a stable negative surface charge. They exhibit high biocompatibility (>70 % viability up to 80 µg/mL) and efficient DOX loading (26.8 %). FePO4-DOX nanoparticles deliver enhanced cytotoxicity (10‑fold IC50 reduction) and increased selectivity for cancer cells. The particles also stabilize RNA against hydrolysis and preserve mRNA translation activity, validating their dual role as drug and nucleic acid carriers. These attributes make FePO4 nanoparticles a compelling platform for therapeutic delivery and for fortifying foods with iron and bioactive compounds. FePO4 nanoparticles were produced by mixing equimolar solutions of ammonium phosphate (16 mg/mL) and iron(III) nitrate (8 mg/mL) under constant stirring, followed by centrifugation and washing to remove residual precursors. The resulting precipitate was dispersed in DSPE‑PEG‑COOH (10 % w/w) to generate stable nanoparticles. Size and zeta potential were measured by DLS; chemical identity was confirmed by FTIR. DOX (100 µg) was added to Fe(NO3)3 or (NH4)3PO4 solutions before precipitation to achieve complexation with Fe3+. The resulting FePO4-DOX particles were washed thrice and quantified by fluorescence spectroscopy (excitation 490 nm, emission 595 nm). Loading efficiency was calculated as ((DOX in NPs – DOX in supernatant)/initial DOX) × 100. Mouse osteosarcoma K7M2 and fibroblast NIH/3T3 cells were seeded at 10,000 cells/well in 96‑well plates, treated with FePO4 or FePO4-DOX (20–600 µg/mL) for 48 h, and viability assessed by MTT (550 nm). Cell lines were also exposed to free DOX (0.05–5 µM) for comparative cytotoxicity. For confocal imaging, K7M2 cells (12,000 cells/well) were treated with 5 µg/mL DOX or FePO4-DOX for 0.5, 1, and 3 h, fixed, stained with DAPI, and imaged on a Zeiss LSM‑700. DOX fluorescence (560 nm) was used to track intracellular uptake. Torula yeast RNA (1 mg/mL) was incubated with 20 µg/mL of Cu, FePO4, Ag, CNT, or ZnO nanoparticles at 37 °C and monitored over time by RAGE. For serum stability, RNA was incubated in 10 % FBS/DMEM. Luciferase‑encoding mRNA was translated in rabbit reticulocyte lysate with or without 20 µg/mL FePO4 or Cu nanoparticles, and luminescence was measured with a Biotek Synergy H1 reader. Data represent at least three independent experiments, expressed as mean ± s.d. Viability data were derived from six technical replicates. The datasets supporting this study are available from the corresponding author upon reasonable request.Abstract
Introduction
Results and Discussion
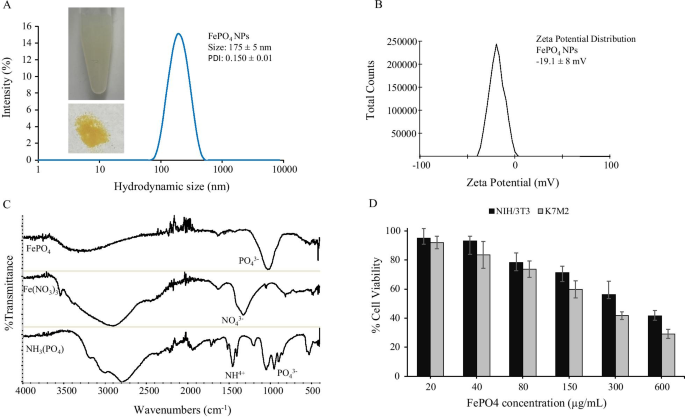
FePO4 Nanoparticle Synthesis, Characterization, and Biocompatibility
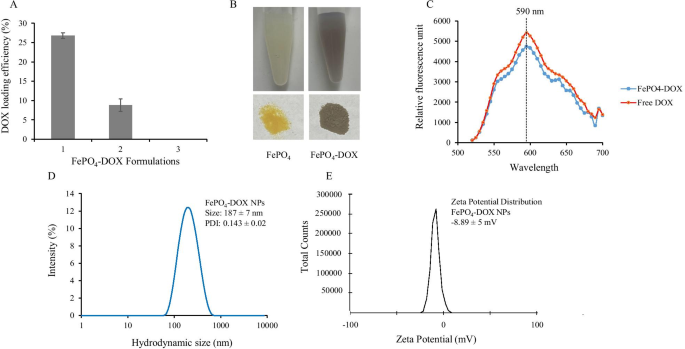
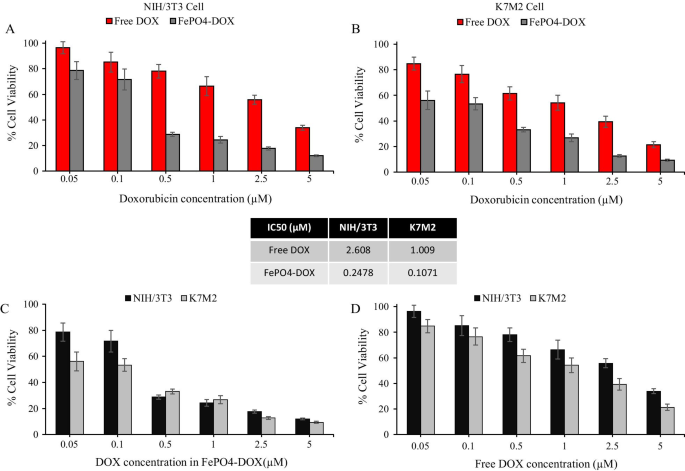
Doxorubicin Loading and Enhanced Cytotoxicity of FePO4-DOX
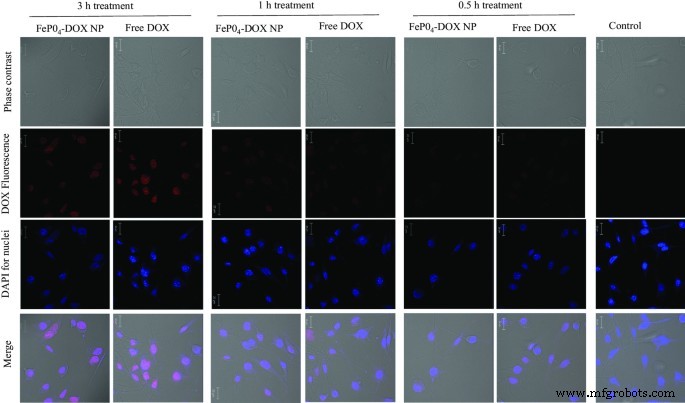
Cellular Internalization of FePO4-DOX Nanoparticles
RNA Stabilization and Functional Expression

Conclusion
Methods
Synthesis and Characterization of FePO4 Nanoparticles
Doxorubicin Loading
Biocompatibility and Cytotoxicity Assays
Cellular Internalization
RNA Stability and Expression
Statistical Analysis
Availability of Data and Materials
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Optimizing Sb-Loaded Palygorskite Nanoparticles for Superior Catalytic Hydrogenation of p-Nitrophenol
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
- Zinc Oxide Nanoparticles: Antimicrobial Properties, Mechanisms, and Applications
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Biotin‑Functionalized Redox‑Sensitive Chitosan Nanoparticles Coated with Phycocyanin for Targeted Curcumin Delivery
- Green Synthesis of Silver Nanoparticles with Yeast Extract: Controlled Morphology and Enhanced Antibacterial Activity
- Quantitative Study of PLGA Nanoparticle Uptake in Laryngeal Cancer and Immune Cells to Improve Drug Delivery



