Bovine Serum Albumin–Coated Gold Nanorods Enable Low‑Dose NIR‑II Photothermal Therapy
The second near‑infrared (NIR‑II, 1000–1300 nm) window offers unparalleled tissue penetration, positioning it as the ideal optical regime for imaging and photothermal therapy. Yet, crafting gold nanorods that absorb within this band while maintaining low cytotoxicity remains challenging. We engineered a series of high‑aspect‑ratio gold nanorods exhibiting robust surface‑plasmon resonance across the NIR‑II window. Coating these nanorods with bovine serum albumin (BSA) dramatically enhanced their biocompatibility without compromising optical performance. In a breast‑cancer mouse model, the BSA‑coated nanorods achieved complete tumor ablation using a modest NIR‑II laser intensity of only 0.75 W cm⁻². These results confirm that low‑dose NIR‑II photothermal therapy is feasible with large‑aspect‑ratio gold nanorods. Gold nanoparticles (AuNPs) have emerged as versatile agents in biomedical research due to their exceptional biocompatibility and low cytotoxicity. Their high X‑ray attenuation makes them attractive for computed tomography (CT) tumor imaging [1, 2], while their pronounced surface‑plasmon resonance (SPR) enables efficient photon‑to‑heat conversion for photothermal therapy (PTT) [3, 4]. AuNPs with tunable size and morphology—nanorods, nanoshells, nanocages—have been tailored for cancer ablation, particularly gold nanorods (AuNRs) whose anisotropy affords strong NIR absorption and photothermal stability [5–8]. NIR light penetrates tissues more deeply than visible light, and longer wavelengths experience reduced scattering [9]. Rod‑shaped particles also exhibit enhanced tumor permeability and prolonged circulation, boosting accumulation [10, 11]. However, conventional PTT often requires high‑power lasers, risking collateral damage [12]. The NIR‑II window offers deeper penetration and lower scattering than NIR‑I, enabling effective therapy at lower laser intensities [13–17]. Yet, producing AuNRs that absorb in NIR‑II while remaining biocompatible is difficult. Here, we report a seed‑less synthesis of AuNRs with absorption peaks at 1000–1300 nm, followed by BSA coating to reduce cytotoxicity. We evaluate their photothermal performance in vitro and in vivo, demonstrating tumor ablation in a breast‑cancer mouse model with a laser intensity as low as 0.75 W cm⁻². Gold chloride trihydrate (HAuCl₄·3H₂O, 99.9 %), hexadecyltrimethylammonium bromide (CTAB, 99 %), nitric acid (GR 65–68 %), hydrogen peroxide (30 %), sodium borohydride (NaBH₄, 97 %), silver nitrate (AgNO₃, 99.8 %), hydrochloric acid (38 %), hydroquinone (99 %), bovine serum albumin (BSA, 98 %) were sourced from Shanghai Aladdin Biological Technology Co. Ltd. RPMI 1640, Penicillin–Streptomycin, PBS, foetal bovine serum (FBS), 4T1 cells, and Cell Counting Kit‑8 (CCK‑8) were obtained from HyClone, Corning, Gibco, and Dojindo, respectively. Ultrapure water (Millipore) was used throughout.
AuNRs were synthesized via a seedless protocol. Briefly, 0.4 mL of 10 mM HAuCl₄ and 10 mL of 0.1 M CTAB were combined with 23–33 µL of 100 mM AgNO₃. Subsequent addition of 10–30 µL 1.2 M HCl and 525 µL 0.1 M hydroquinone yielded a light‑yellow growth solution. After 15 min stirring, 10–40 µL of freshly prepared 10 mM ice‑cold NaBH₄ was injected, followed by 30 s stirring and 18 h aging at room temperature. AuNR@CTAB was purified by two PBS washes.
CTAB concentration in AuNR@CTAB was adjusted to 1 mM and sonicated to dissolve fully. 3 mL of AuNR@CTAB was mixed with 3 mL 10 mg mL⁻¹ BSA and sonicated for 30 min. After centrifugation (9500 × g, 40 min), the pellet was resuspended in 6 mL 5 mg mL⁻¹ BSA, pH adjusted to 11–12 with 2 M NaOH, and stirred for ≥18 h. Final AuNR@BSA was purified by two PBS washes and stored in PBS.
Transmission electron microscopy (TEM, Talos F200X) assessed morphology; dynamic light scattering (DLS, Zetasizer Nano ZS) measured size and zeta potential; UV–Vis spectroscopy (Shimadzu UV‑2700) recorded absorption. In vivo tumor distribution was examined by TEM of excised tumor sections post‑injection. FT‑IR and XRD analyses (Beijing Zhongke Baice Co.) confirmed BSA coating and crystalline gold, respectively.
AuNR solutions (OD = 0.5–2 at 1064 nm) were irradiated with a 1064 nm laser (0.35–1 W cm⁻²) for 30 min, and temperature changes were monitored by infrared thermal imaging (FLUKE TI25). Photostability was evaluated by repeated 10 min on/off irradiation cycles at 0.5 W cm⁻².
4T1 cells were seeded in 96‑well plates (5 × 10³ cells well⁻¹) and incubated 24 h. AuNR@CTAB or AuNR@BSA (10 µL) was added at varying concentrations, followed by 24 h incubation. CCK‑8 (10 µL) was added, incubated 40 min, and absorbance measured at 450 nm. Phototoxicity assays involved 1064 nm irradiation (0.75 W cm⁻², 10 min) before the CCK‑8 test.
Female BALB/c mice (6–8 weeks) were inoculated subcutaneously with 2 × 10⁶ 4T1 cells. When tumors reached ~100 mm³, experiments commenced. All procedures followed institutional ethical guidelines.
AuNR@BSA (200 µL) was injected via the tail vein. Blood samples were collected at 0.25, 2, 4, 6, 8, 12, 36, and 48 h, digested with HNO₃ and H₂O₂, and Au content quantified by ICP‑OES. Organs (heart, liver, spleen, lung, kidney) were harvested 24 h post‑injection, digested, and Au concentrations measured similarly.
Mice were divided into four groups: (1) AuNR@BSA, (2) AuNR@BSA + laser, (3) laser only, (4) control. AuNR@BSA (200 µL) was injected intratumorally, followed 10 min later by 1064 nm irradiation (0.75 W cm⁻², 10 min). Tumor temperature, volume, and body weight were monitored for 14 days. Tumors were excised at the study endpoint.
Statistical analyses were performed with SPSS 16.0. Data are expressed as mean ± SD; significance was set at p < 0.05.
Optimizing NaBH₄, HCl, and Ag⁺ concentrations enabled control over aspect ratio and SPR peak position. Increasing NaBH₄ blue‑shifted the peak from 1223 to 865 nm, while higher HCl red‑shifted it from 871 to 1070 nm. Reducing Ag⁺ concentration extended the SPR peak further. Final AuNRs exhibited a wine‑red color and an absorption maximum at 1064 nm (Fig. 1). This wavelength aligns with the NIR‑II window and matches available laser sources.
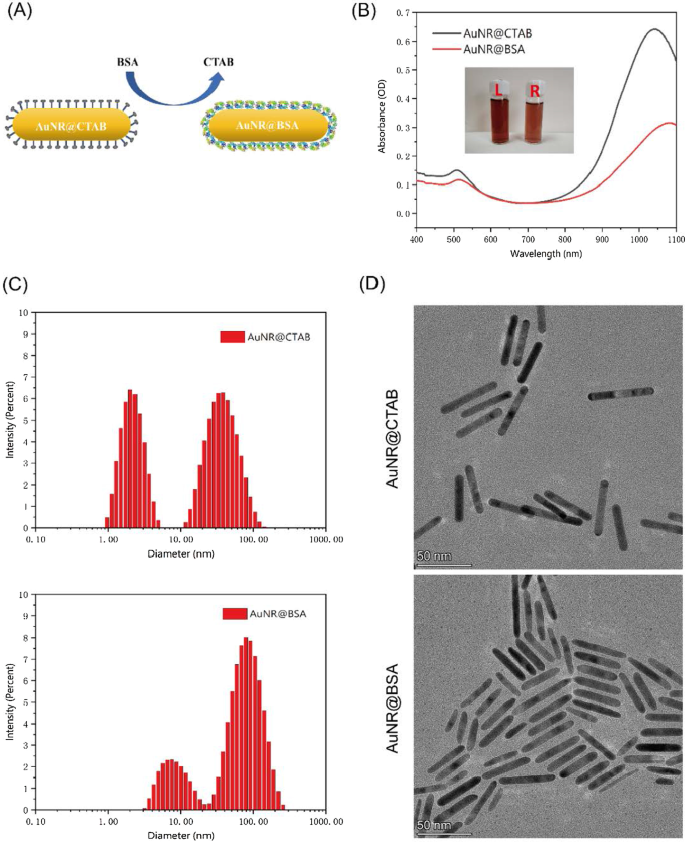
Figure 1. Photoproperties of AuNR@CTAB under varying synthesis conditions: (a) preparation scheme; (b) TEM image; (c) UV–Vis spectra versus NaBH₄; (d) versus HCl; (e) versus AgNO₃. CTAB, while effective for nanorod growth, is cytotoxic above 1–10 µM and can precipitate at low temperatures. Replacing CTAB with BSA both mitigates toxicity and improves colloidal stability. AuNR@BSA retained a similar aspect ratio but displayed a ~30 nm red‑shifted SPR peak at 1064 nm (Fig. 2). Zeta potential changed from positive (CTAB) to negative (BSA), confirming surface exchange. FT‑IR spectra revealed characteristic BSA amide bands (1649 and 1539 cm⁻¹). DLS showed hydrodynamic diameters of 8.6 nm and 89 nm for the two populations, indicating mild size increase upon BSA coating. TEM confirmed rod‑like morphology with slightly rounded ends (Fig. 2). All dimensions remained below 100 nm, favoring tumor penetration.
Figure 2. Characterization of AuNR@BSA and AuNR@CTAB: (a) synthesis; (b) UV–Vis spectra; (c) DLS size distribution; (d) TEM images. AuNR@BSA (OD ≈ 1) achieved a temperature rise from ~25 °C to 80 °C within 5 min under 0.35 W cm⁻², plateauing thereafter (Fig. 3A). Temperature scaled linearly with laser power (Fig. 3B). Increasing OD further boosted peak temperatures. Photothermal stability was confirmed over three 10‑min irradiation cycles at 0.5 W cm⁻² (Fig. 3D). Thermal images displayed peak temperatures of 85.5 °C for AuNRs versus 44.5 °C for PBS (Fig. 3E). These results underscore the superior photothermal conversion and stability of BSA‑coated nanorods in the NIR‑II range.
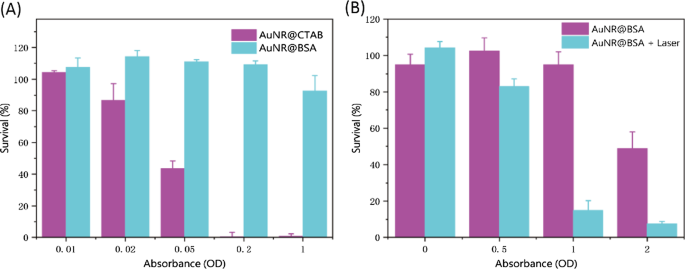
Figure 3. In vitro photothermal performance: (a) temperature vs. time; (b) temperature vs. laser power; (c) temperature vs. absorbance; (d) three‑cycle stability; (e) thermal images. AuNR@CTAB displayed significant cytotoxicity even at low concentrations (OD ≈ 0.05), whereas AuNR@BSA maintained >80 % cell viability up to OD ≈ 1 (Fig. 4A). Photothermal treatment (0.75 W cm⁻², 10 min) reduced 4T1 cell viability to ~20 % at OD ≈ 1, while control cells remained ~100 % viable (Fig. 4B). Thus, BSA coating preserves biocompatibility while enabling effective photothermal killing.
Figure 4. Cytotoxicity and photothermal toxicity: (a) cell viability vs. concentration; (b) viability with/without laser. AuNR@BSA exhibited a half‑life of ~1.5 h in circulation (Fig. 5A). After 24 h, predominant accumulation was observed in liver and spleen, consistent with reticuloendothelial system uptake (Fig. 5B). These findings suggest potential for liver‑spleen‑targeted applications.
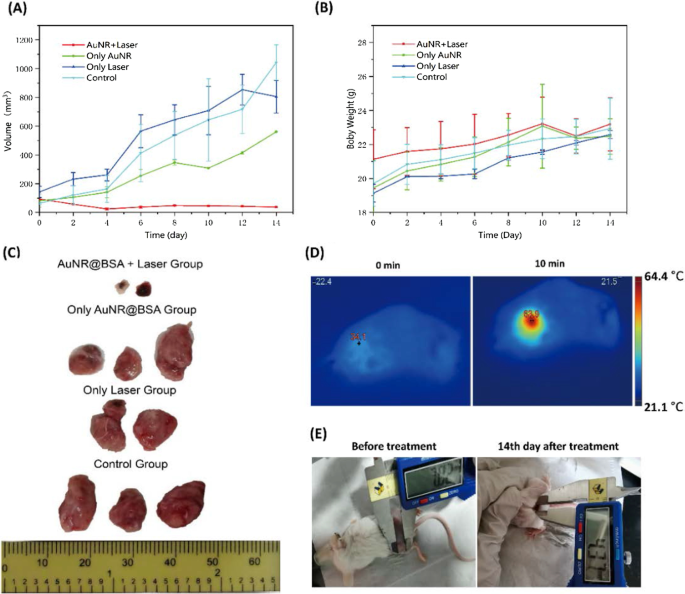
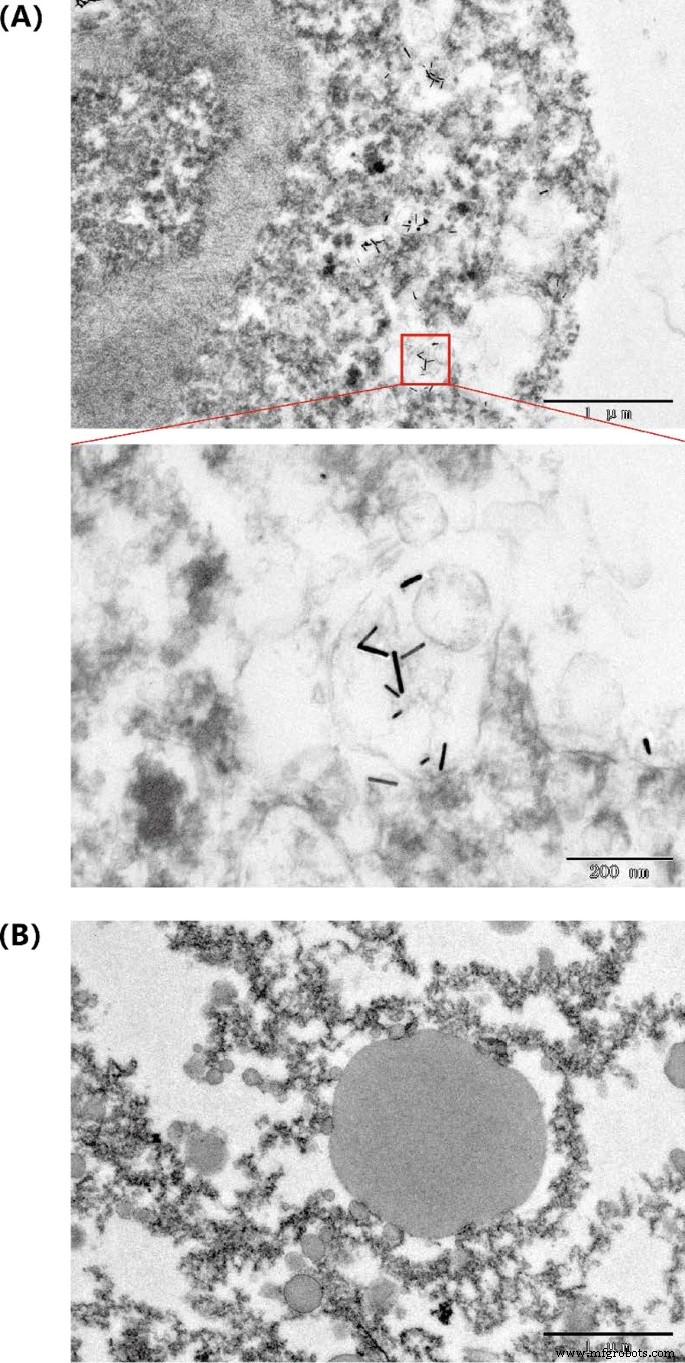
Figure 5. Biodistribution: (a) Au concentration in blood over time; (b) organ distribution at 24 h. Intratumoral injection of AuNR@BSA followed by 1064 nm irradiation (0.75 W cm⁻², 10 min) raised tumor temperature to ~64 °C, while control groups remained near body temperature (Fig. 6D). Over 14 days, the AuNR@BSA + laser group showed complete tumor ablation with no rebound growth; tumors in control groups progressed rapidly (Fig. 6A, B). Post‑mortem analysis confirmed absence of residual tumor tissue (Fig. 6C). TEM of treated tumors revealed intact AuNRs, attesting to in vivo photostability (Fig. 7).
Figure 6. In vivo treatment outcomes: (a) tumor volume; (b) body weight; (c) tumor photographs; (d) infrared thermography; (e) post‑treatment images. Figure 7. TEM of AuNR@BSA in tumor tissue: (a) treated; (b) untreated. We have synthesized BSA‑coated gold nanorods with a 1064 nm SPR peak, combining high photothermal efficiency with enhanced biocompatibility. In vivo studies demonstrated rapid tumor ablation in a breast‑cancer mouse model using low‑dose NIR‑II laser irradiation. The nanorods exhibit favorable biodistribution, with significant liver and spleen uptake, and maintain photostability in the tumor microenvironment. These attributes position AuNR@BSA as a promising platform for NIR‑II photothermal therapy, potentially in combination with immunotherapy or chemotherapy for personalized, safe, and effective cancer treatment.
The dataset underlying this study is available from the corresponding author upon reasonable request. All data generated or analyzed during this study are included in this published article.
Abstract
Introduction
Materials and Methods
Materials
Preparation of AuNR@CTAB
Preparation of AuNR@BSA
Characterization
Photothermal Performance
In Vitro Cytotoxicity
Animal Studies
In Vivo Biodistribution and Circulation
Photothermal Treatment Efficiency
Data Analysis
Results and Discussion
Synthesis and Characterization of AuNR@CTAB
Synthesis and Characterization of AuNR@BSA
In Vitro Photothermal Effect
In Vitro Cytotoxicity and Photothermal Toxicity
In Vivo Biodistribution and Circulation
In Vivo Photothermal Treatment
Conclusion
Availability of data and materials
Nanomaterials
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Antibody‑Functionalized Silica‑Coated Gold Nanorods for Rapid Diagnosis and Photothermal Treatment of Cryptococcus neoformans
- Shape‑Dependent Uptake of Gold Nanostructures in Glioblastoma Cells: From Synthesis to Cellular Interaction
- Seed‑Mediated Synthesis of Tunable‑Aspect‑Ratio Gold Nanorods for Enhanced Near‑Infrared Photoacoustic Imaging
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Targeted MoSe2 Nanodots in Albumin Nanospheres: A Biocompatible Dual‑Modality Agent for Synergistic Photothermal Radiotherapy
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Efficient One-Step Green Synthesis of Multifunctional Gold Nanoparticles for Targeted Tumor Imaging and Therapy



