5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
Abstract
Carbon dots (CDs) are fluorescent nanomaterials prized for their tunable photoluminescence, high photothermal conversion, and excellent biocompatibility. Enhancing their quantum yield (QY) typically requires surface passivation or heteroatom doping, particularly nitrogen. In this study, we present a rapid, one‑pot microwave‑assisted synthesis that integrates polydopamine (PDA) passivation into a 5‑minute pyrolysis step, markedly simplifying the protocol compared to conventional hydrothermal methods. The resulting PDA‑functionalized CDs (CD‑PDA) were characterized by DLS, FT‑IR, UV‑Vis, and fluorescence spectroscopy. The N‑doping introduced by PDA increased the QY from <5 % to 5 % (a near‑tripling) and improved the yield by ~1.5‑fold due to enhanced nucleation via phenolic groups. CD‑PDA also displayed a 35 % photothermal conversion efficiency, surpassing many gold‑based photothermal agents. Importantly, the PDA coating retains functional groups that enable further conjugation via Michael addition or Schiff base chemistry, opening avenues for targeted drug delivery or theranostic applications.
Background
Carbon dots are a unique class of zero‑dimensional carbon nanostructures that combine sp² and sp³ hybridization with abundant π‑electrons, fostering defect sites and heteroatom incorporation that drive their light‑to‑heat or light‑to‑photon conversion processes. Since their first isolation from arc‑discharged single‑walled carbon nanotube purification in 2004, CDs have been leveraged in bioimaging, photothermal therapy (PTT), photodynamic therapy (PDT), and biosensing, owing to their adjustable fluorescence, high photothermal conversion, and inherent biocompatibility. A number of synthetic routes exist—laser ablation, acid oxidation, hydrothermal treatment, microwave pyrolysis, electrochemical oxidation, ultrasonic irradiation, and plasma methods—yet achieving high QY and robust NIR absorption remains challenging.
Incorporating nitrogen heteroatoms is a proven strategy to boost CD fluorescence. For instance, polyethylenimine (PEI)‑functionalized CDs prepared by 700 W microwave pyrolysis of glycerol and branched PEI reached a QY of 15.3 % and were applied in cell imaging and gene delivery. N‑ and P‑co‑doped CDs derived from ATP via hydrothermal treatment achieved a 9.8 % QY. Phenolic compounds such as ferulic acid have also been shown to enhance CD growth by acting as catalytic seeds.
Polydopamine (PDA), a melanin‑like polymer formed by dopamine self‑polymerization, is rich in catechol, amine, and phenolic hydroxyl groups. These functionalities make PDA an attractive passivating and catalytic agent for CDs. Building on this premise, we developed a one‑pot, 5‑minute microwave‑assisted pyrolysis protocol to synthesize PDA‑passivated CDs, evaluating their physicochemical properties, photoluminescence, photothermal performance, and cytotoxicity.
Results and Discussion
Synthesis and Characterization of CD‑PDA
CD‑PDA were fabricated by combining 6 mL of pre‑polymerized PDA (2 h at pH 8.5) with 20 mL of glycerol and subjecting the mixture to 5 min of 500 W microwave oxidation. For comparison, control CDs were produced from glycerol alone under identical conditions. The PDA‑mediated synthesis yielded ~1.5‑fold higher product mass, attributable to the phenolic groups that enhance nucleation.
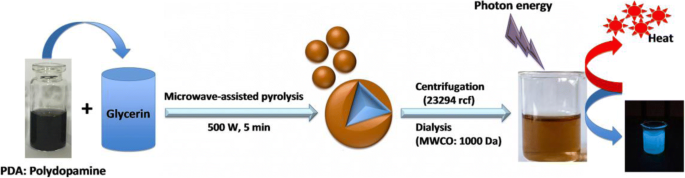
Schematic illustration of the CD‑PDA synthesis pathway.
Dynamic light scattering revealed a hydrodynamic diameter of 51.5 ± 19.5 nm for CD‑PDA, with a zeta potential of –27.5 ± 0.4 mV, indicating a negatively charged PDA coating. Control CDs exhibited 5.5 ± 2.5 nm. TEM images confirmed monodisperse spherical particles with diameters of ~25 nm for CD‑PDA and ~5 nm for CDs, reflecting the 20 nm size increase after PDA passivation.
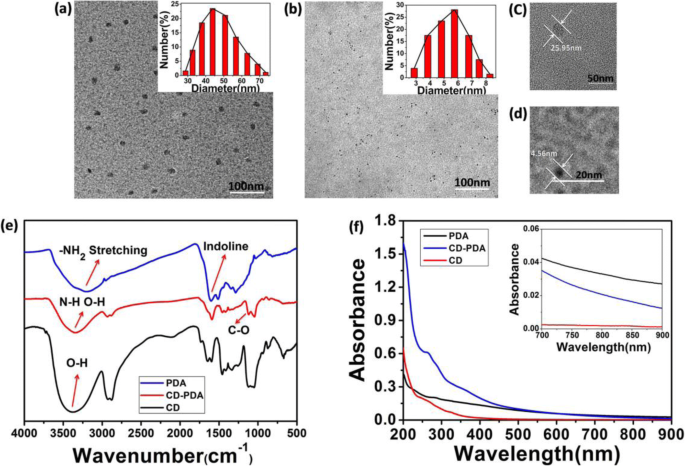
Morphology, FT‑IR spectra, and UV‑Vis spectra of CD‑PDA and CDs.
FT‑IR spectra displayed characteristic catechol O–H stretches (~3400 cm⁻¹) and aromatic C=C bands (~1600 cm⁻¹), confirming PDA integration. New peaks at 1642, 1588, and 1640 cm⁻¹ corresponded to C=O, N–H, and C–N vibrations, evidencing successful surface passivation. UV‑Vis spectra showed broad absorption from 200 to 900 nm, especially in the NIR region, a hallmark of PDA’s photothermal capability. CD‑PDA exhibited peaks at 274 and 370 nm, while CDs peaked at 260 and 330 nm; the bathochromic shift reflects amidogen introduction from PDA.
Photoluminescence of CD‑PDA
CD‑PDA’s emission peak shifted from 450 to 500 nm as the excitation wavelength varied from 350 to 420 nm, demonstrating tunable fluorescence. Fluorescent microscopy and macroscopic UV imaging (365 nm) confirmed stronger emission relative to CDs. The QY, measured against quinine sulfate (54 % QY), was 5 %, nearly triple the control CDs. Fluorescence intensity remained stable over 2100 s of 365 nm irradiation, indicating robust photoluminescence.
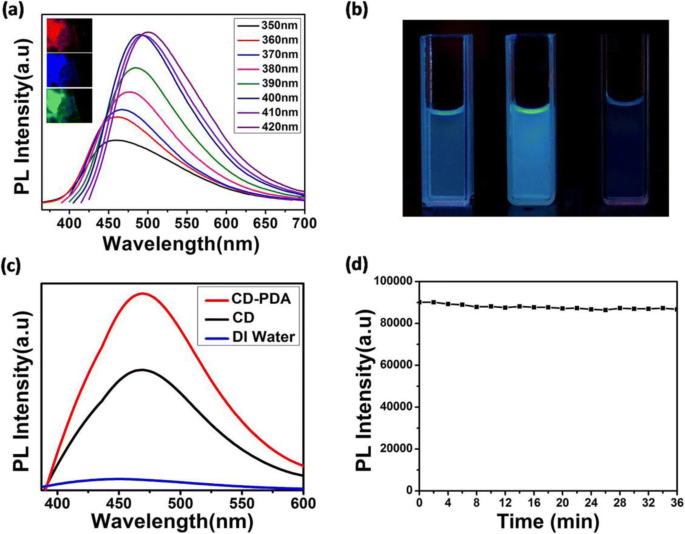
Optical properties of CD‑PDA and CDs.
Optimization studies revealed that 2‑h dopamine pre‑polymerization yielded maximum fluorescence, while varying PDA concentrations (3–9 mg mL⁻¹) produced a bell‑shaped response. Acidic conditions (pH 5) further enhanced fluorescence, suggesting favorable performance in tumor microenvironments.
Photothermal Performance and Cytotoxicities of CD‑PDA
Photothermal Efficacy
Under 808 nm, 2 W cm⁻² irradiation for 10 min, CD‑PDA (200 µg mL⁻¹) raised the temperature by 27 °C, comparable to PDA alone (30 °C) and significantly higher than CDs (7.5 °C). Concentration‑dependent heating curves confirmed a direct correlation between CD‑PDA load and temperature rise, with a 35 % photothermal conversion efficiency—surpassing gold nanorods (22 %).
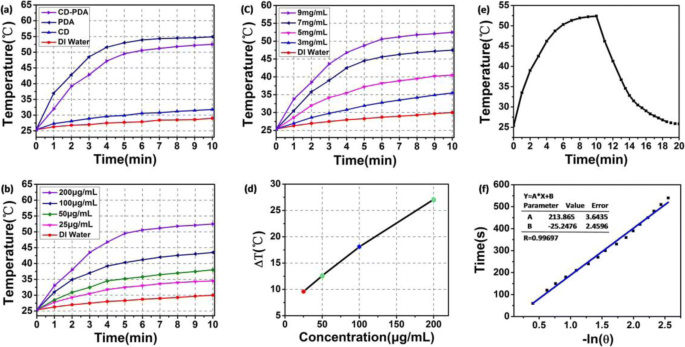
Photothermal conversion properties of CD‑PDA and CDs.
In Vitro Cell Viability
MTT assays on HeLa cells demonstrated dose‑dependent viability. CD‑PDA maintained >80 % viability up to 100 µg mL⁻¹, outperforming CDs and matching PDA’s profile. Upon 808 nm irradiation (5 min), CD‑PDA reduced cell viability to 30 % at 200 µg mL⁻¹, confirming potent photothermal killing while retaining low dark toxicity.
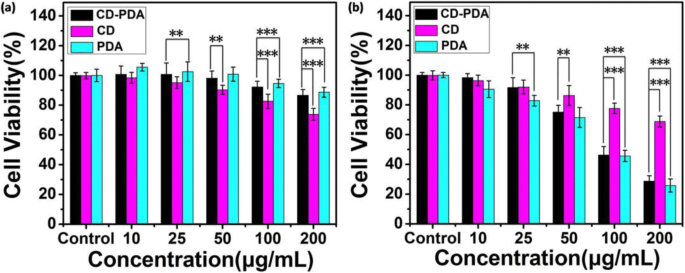
In vitro cytotoxicity of CD‑PDA, CDs, and PDA.
Conclusions
We have established a streamlined, 5‑minute microwave‑assisted protocol to synthesize PDA‑passivated CDs. The resulting CD‑PDA exhibit a 5 % QY, a 35 % photothermal conversion efficiency, and enhanced yield due to PDA‑induced nucleation. In vitro studies reveal excellent biocompatibility and significant photothermal therapeutic effect, with the PDA layer offering avenues for further functionalization via Michael addition or Schiff base chemistry.
Methods/Experimental
Materials
All reagents were analytical grade. Dopamine hydrochloride (Sigma‑Aldrich), quinine sulfate (Fluka), glycerol (>99 %), Tris buffer (10 mM, pH 8.5), DMSO (>99.8 %), dialysis membranes (MWCO 1000 Da), MTT, trypsin, and antibiotics (Beyotime) were used without further purification. HeLa cells were obtained from ATCC. DMEM and FBS were sourced from HyClone and Biological Industries, respectively.
Instrumentation and Characterization
FT‑IR (Nicolet 380), UV‑Vis‑NIR (Perkin‑Elmer Lambda 750), fluorescence (Tecan Infinite 200PRO), DLS (Malvern Nano‑ZS), TEM (FEI Tecnai G), and a domestic 500 W microwave oven (Galanz) were employed for analysis.
Preparation of CD‑PDA and CDs
Dopamine (50 mg) was dissolved in 10 mL Tris (pH 8.5) and stirred at room temperature for 2 h to form PDA. CD‑PDA synthesis involved mixing 6 mL of this PDA with 20 mL glycerol, followed by 5 min microwave oxidation at 500 W, then dialysis (48 h, MWCO 1000 Da) and lyophilization. CDs were prepared similarly but without PDA.
Fluorescence Quantum Yield Determination
QY was measured using quinine sulfate (0.1 M H₂SO₄, QY = 54 %) as a standard. Samples were diluted to an optical density <0.02 at 350 nm, and integrated fluorescence (380–700 nm) was compared against the standard using the formula F = Fₛₜ(GradX/GradST)(RX²/RST²).
Photothermal Measurements
Samples (200 µg mL⁻¹) were placed in quartz cells and irradiated with an 808 nm, 2 W cm⁻² laser. Temperature was recorded every minute for 10 min; cooling curves were obtained post‑irradiation. Photothermal efficiency was calculated following established protocols.
Cell Culture
HeLa cells were cultured in DMEM (high glucose) supplemented with 10 % FBS and antibiotics at 37 °C, 5 % CO₂.
MTT Assay
Cells (2 × 10⁴ cells well⁻¹) were incubated with varying concentrations of CD‑PDA (10–100 µg mL⁻¹) for 24 h, followed by MTT (5 mg mL⁻¹) for 4 h. Absorbance at 490 nm was measured, and viability was expressed as a percentage relative to untreated controls.
Nanomaterials
- Bovine Serum Albumin–Coated Gold Nanorods Enable Low‑Dose NIR‑II Photothermal Therapy
- High‑Efficiency, Excitation‑Independent Blue‑Emitting Carbon Dots with Tunable Photoluminescence
- Fast Microwave‑Synthesis of Pyridinic‑Rich N,S‑Co‑Doped Carbon Quantum Dots with Superior Photoluminescence and Catalase‑Mimicking Activity
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Rapid, One‑Pot Synthesis of Nitrogen‑Doped Microporous Carbon Spheres for Superior Symmetric Supercapacitor Performance
- Carbon Quantum Dot–Platinum Porphyrin Composite: A Dual‑Function Theranostic Agent for Enhanced Photodynamic Cancer Therapy
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Carbon Dots: Pioneering Next-Generation Nanothermometers – A Comprehensive Review
- Targeted Carbon Nanotube Photothermal Therapy Plus Checkpoint Inhibition: A New Strategy for Metastatic Breast Cancer



