Carbon Quantum Dot–Platinum Porphyrin Composite: A Dual‑Function Theranostic Agent for Enhanced Photodynamic Cancer Therapy
Abstract
Photosensitizers (PSs) are notoriously hydrophobic, limiting their clinical utility in photodynamic therapy (PDT). To overcome this, we engineered a novel theranostic nanoagent, CQDs@PtPor, by electrostatic binding of a tetraplatinated porphyrin complex (PtPor) to negatively charged carbon quantum dots (CQDs). Comprehensive characterization (XRD, TEM, XPS, FTIR) confirmed the successful assembly of CQDs and PtPor into a single nanostructure. Spectroscopic analysis revealed efficient resonance energy transfer from CQDs to PtPor, resulting in markedly enhanced singlet oxygen (1O2) production. In vitro studies on HeLa cells demonstrated that CQDs@PtPor achieved superior phototoxicity compared to free PtPor while exhibiting negligible dark cytotoxicity, underscoring its potential as a safe and effective cancer therapeutic platform.
Background
PDT has emerged as a non‑invasive strategy for treating skin disorders, macular degeneration, and various cancers [1]. The therapy hinges on light‑activated PSs that generate reactive oxygen species (ROS) upon irradiation, selectively killing tumor cells while sparing healthy tissue [2–6]. Although activatable PSs such as porphyrin and phthalocyanine derivatives can serve dual roles in imaging and therapy, their clinical translation is hampered by poor aqueous solubility, prolonged cutaneous photosensitivity, limited tissue penetration, and suboptimal absorption beyond 700 nm [7–11]. Various nanocarriers—liposomes, polymeric nanoparticles, gold nanoparticles, carbon nanotubes, graphene, and carbon quantum dots (CQDs)—have been explored to improve PS delivery and performance [12–22].
CQDs, a versatile class of carbon nanomaterials, exhibit exceptional optical properties, high water solubility, low toxicity, excellent biocompatibility, and facile surface functionalization [23–27]. Their abundant surface groups enable the covalent or non‑covalent attachment of drugs, proteins, or targeting ligands, making them attractive carriers for theranostic applications [31–33]. Prior studies have demonstrated that CQDs conjugated with PSs (e.g., Ce6, ZnPc, TMPyP) can enhance fluorescence imaging and photodynamic efficacy through energy transfer mechanisms [34–36].
Recently, a tetraplatinated porphyrin complex exhibited low dark cytotoxicity but IC50 values as low as 19 nM upon 420‑nm irradiation, indicating potent anticancer activity [37]. However, its hydrophobicity and limited biocompatibility preclude clinical use. We therefore designed CQDs@PtPor to harness CQDs’ solubility and surface chemistry while retaining PtPor’s therapeutic potency. The electrostatic assembly between the positively charged PtPor and negatively charged CQDs not only improves water dispersibility but also promotes energy transfer, thereby amplifying 1O2 generation and phototoxicity.
Methods
Materials
Trans‑platinum diammine dichloride (trans‑platin) (Aladdin®), 1,3‑Diphenylisobenzofuran (DPBF) (Sigma‑Aldrich), and all solvents were purchased from Tianjin Fu Chen Chemical Reagents or Sinopharm Chemical Reagent Co., Ltd. All reagents were used as received.
Synthesis of [Trans‑PtCl(NH3)2]4‑5,10,15,20‑Tetra(4‑pyridyl)‑Porphyrin Nitrate
Trans‑platin (0.193 mmol, 58 mg) and AgNO3 (0.193 mmol, 33 mg) were dissolved in 5 mL DMF and stirred for 24 h. The resulting white AgCl precipitate was removed by centrifugation, yielding a clear solution. This was added to a suspension of 5,10,15,20‑Tetra(4‑pyridyl)porphyrin (0.487 mmol, 30 mg) in 3 mL DMF, followed by stirring at 50 °C for 48 h. After cooling, 10 mL diethyl ether was added to precipitate the red product, which was washed with methanol, dichloromethane, and ether. Drying under vacuum gave 81 mg of product (86 % yield). The structure was confirmed by 1H NMR and ESI‑MS.
Preparation of CQDs
Citric acid (0.45 g) and ethylenediamine (500 µL) were dissolved in 10 mL DI water and subjected to hydrothermal treatment at 200 °C for 5 h in a PTFE‑lined autoclave. The resulting brown‑black solution was purified by centrifugation and dialysis against DI water to obtain CQDs.
Preparation of CQDs@PtPor Composite
PtPor (20 mg in 3 mL DMSO) was dispersed in 12 mL water and slowly added to a CQDs suspension (5 mg in 15 mL H2O) under sonication. The mixture was stirred at room temperature for 24 h, then purified by centrifugation and dialyzed for 2 days. The final aqueous solution was lyophilized at 4 °C to yield the composite.
Quantum Yield Measurement
The CQD quantum yield (Φ) was calculated relative to quinine sulfate (Φst ≈ 54 %) using the standard comparative method. Absorbance was kept below 0.1 at 360 nm to avoid reabsorption effects.
Singlet Oxygen Generation
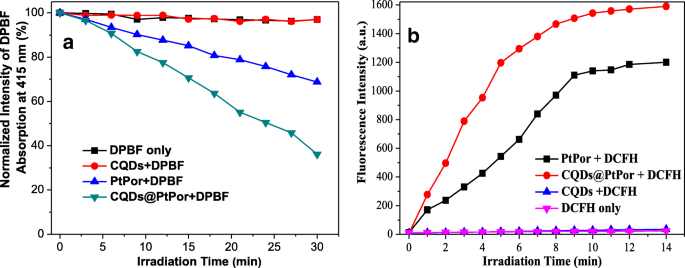
Samples (5 mg mL−1) were mixed with 10 mM DPBF in a 3 mL cuvette and irradiated with a 50 W xenon lamp (500 nm long‑pass filter, 6 mW cm−2) for up to 30 min. The decrease in DPBF absorbance at 415 nm was monitored every 3 min to quantify 1O2 production. DCFH fluorescence (λem = 525 nm) was also used as a qualitative probe.
Cytotoxicity Assays
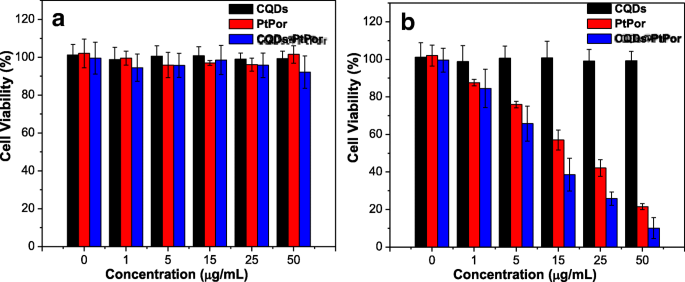
HeLa cells were cultured in DMEM with 5 % FCS, 100 U mL−1 penicillin, and 100 µg mL−1 streptomycin at 37 °C, 6 % CO2. For dark cytotoxicity, cells were incubated with varying concentrations of CQDs, PtPor, or CQDs@PtPor for 24 h, then assessed by MTT assay. For phototoxicity, cells were irradiated after 24 h incubation, and viability was again determined by MTT.
Cellular Imaging
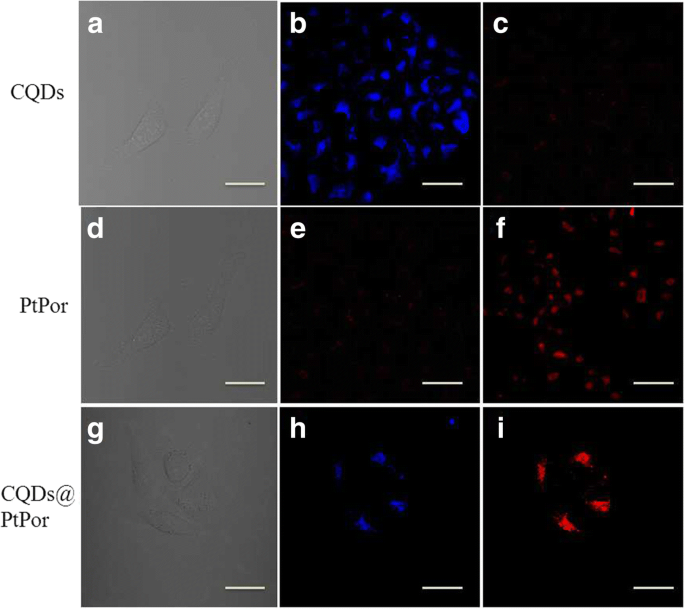
HeLa cells (5 × 104 cells mL−1) were incubated with 0.25 mg mL−1 CQDs@PtPor for 1 h, washed, and imaged by confocal microscopy (Zeiss LSM7 DUO) using a 405 nm laser.
Results
Preparation and Characterization of CQDs@PtPor
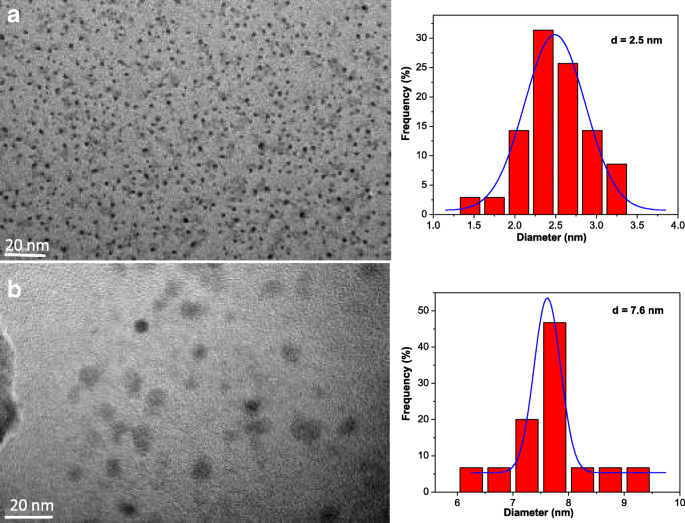
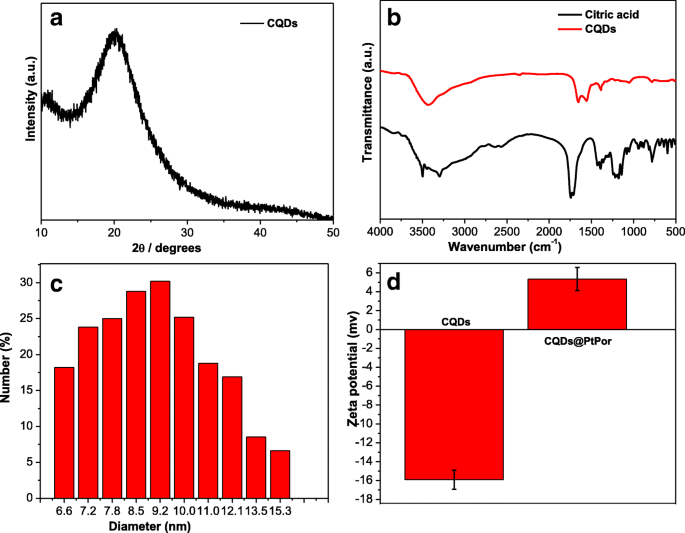
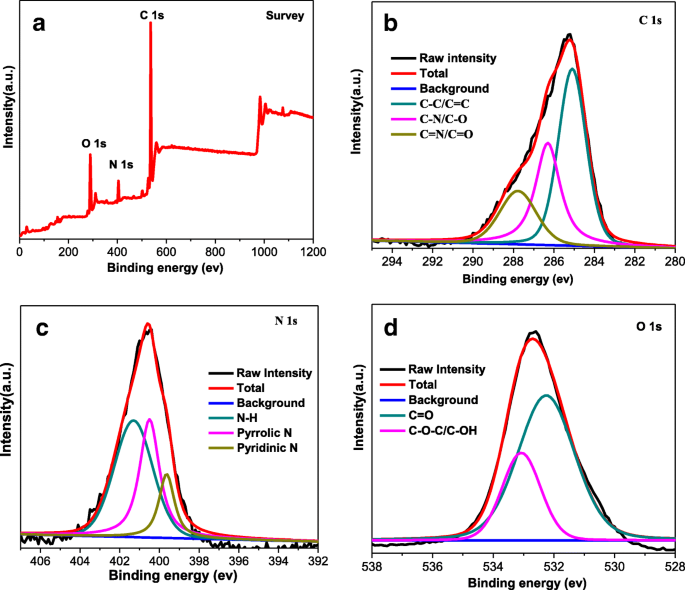
Transmission electron microscopy (TEM) revealed uniform, monodisperse particles: CQDs averaged 2.5 nm, while CQDs@PtPor averaged 7.6 nm, reflecting PtPor adsorption (Fig. 1). XRD patterns displayed a broad peak at ~23°, indicative of amorphous carbon structure. FTIR spectra confirmed the presence of hydroxyl, amino, and amide groups (Fig. 2b). Dynamic light scattering (DLS) measured a mean hydrodynamic diameter of 9.2 nm for CQDs@PtPor, corroborating TEM data. Zeta potential shifted from −15.6 mV (free CQDs) to +4.5 mV after PtPor conjugation, confirming electrostatic binding (Fig. 3d).
Photophysical Properties
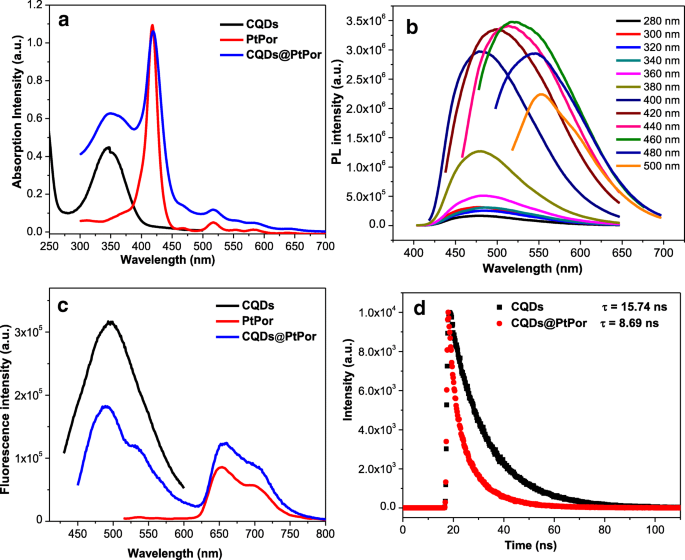
UV‑Vis spectra displayed characteristic CQD absorption (~360 nm) and porphyrin Soret/Q bands (~425, 520, 580 nm). CQDs exhibited excitation‑dependent photoluminescence (PL) with emission shifting from 460 to 552 nm across 280–500 nm excitation, and a quantum yield of 36 %. In the composite, PL intensity at 500 nm decreased while emission at 660 nm increased, evidencing efficient fluorescence resonance energy transfer (FRET). Fluorescence lifetime measurements confirmed a shortened donor lifetime in CQDs@PtPor, further supporting energy transfer (Fig. 4).
Enhanced Singlet Oxygen Production
DPBF degradation experiments revealed that CQDs@PtPor produced significantly more 1O2 than free PtPor, while CQDs alone showed negligible activity (Fig. 5a). DCFH fluorescence corroborated these findings, with CQDs@PtPor yielding the strongest signal (Fig. 5b).
Phototoxicity in HeLa Cells
Dark cytotoxicity assays showed >90 % viability at 50 µg mL−1 for all samples (Fig. 6a). Phototoxicity assays, however, demonstrated a dose‑dependent reduction in viability: CQDs@PtPor at 50 µg mL−1 achieved only 8 % survival, outperforming PtPor (18 %) and CQDs (90 %) (Fig. 6b). This superior efficacy is attributed to the enhanced 1O2 generation mediated by CQDs.
Cellular Uptake and Stability
Confocal imaging revealed that CQDs@PtPor localized predominantly in the cytoplasm of HeLa cells, with blue CQD fluorescence overlapping red PtPor emission, indicating stable complex formation post‑uptake (Fig. 7).
Discussion
The pronounced FRET from CQDs to PtPor, evidenced by reduced donor emission and shortened lifetime, directly translates into higher 1O2 production and enhanced phototoxicity. The small size (~10 nm) of CQDs@PtPor facilitates tumor accumulation via the enhanced permeability and retention (EPR) effect, while the improved water dispersibility and biocompatibility reduce systemic toxicity. The modular nature of the electrostatic assembly allows easy substitution of different PSs or targeting ligands, positioning CQDs@PtPor as a versatile platform for personalized PDT.
Conclusions
We have successfully fabricated a CQDs@PtPor nanoagent that combines the superior optical properties of CQDs with the potent anticancer activity of a tetraplatinated porphyrin. The composite exhibits high aqueous stability, low dark toxicity, efficient energy transfer, and markedly enhanced photodynamic efficacy in vitro. These attributes make CQDs@PtPor a promising candidate for future clinical translation in cancer therapy.
Change History
- Initial submission.
Abbreviations
- CA
- Citric acid
- CQDs
- Carbon quantum dots
- DPBF
- 1,3‑Diphenylisobenzofuran
- EDA
- Ethylenediamine
- MTT
- 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PDT
- Photodynamic therapy
- PSs
- Photosensitizers
- TEM
- Transmission electron microscopy
- UV
- Ultraviolet
- XPS
- X-ray photoelectron spectroscopy
- XRD
- X-ray diffraction
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Hydrothermal Synthesis of Blue‑ and Green‑Emitting Carbon Quantum Dots from Tofu Wastewater
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- High‑Performance Dye‑Sensitized Solar Cells Using Screen‑Printed Multi‑Walled Carbon Nanotube Counter Electrodes
- Efficient Colon Cancer Gene Therapy Using Cationic DOTAP–PEG‑PCL Micelles for siRNA Delivery
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Eco-Friendly Microplasma Synthesis of Yellow-Emitting Carbon Quantum Dots for Cancer Cell Imaging and Photocatalytic Inactivation
- Targeted Carbon Nanotube Photothermal Therapy Plus Checkpoint Inhibition: A New Strategy for Metastatic Breast Cancer
- CoFe₂O₄ Quantum Dots: Dual Photothermal/Photodynamic Therapy for NSCLC—Inducing Apoptosis via PI3K/AKT Modulation
- Liposomal Nanomedicine for Targeted Cancer Drug Delivery: Enhancing Efficacy and Safety



