High‑Efficiency, Excitation‑Independent Blue‑Emitting Carbon Dots with Tunable Photoluminescence
Abstract
We report the synthesis of blue‑luminescent carbon dots (CDs) by a simple hydrothermal route that exhibit an unprecedented excitation‑independent photoluminescence (PL) at low concentrations. Upon dilution, the emission maximum blue‑shifts from 480 nm to 443 nm, while the PL intensity increases markedly. Spectroscopic analysis indicates two distinct emissive pathways: (i) intrinsic sp2 carbon core emission that dominates at low concentrations and is excitation‑independent, and (ii) surface‑state‑driven emission from highly polar nanoclusters that is excitation‑dependent at high concentrations. These findings advance the fundamental understanding of CD photophysics and open new avenues for tunable, high‑quantum‑yield fluorescent applications.
Background
Carbon dots, a subclass of carbon‑based nanomaterials, have attracted considerable attention due to their small size (<10 nm), excellent photostability, biocompatibility, and environmental benignity [1]. Their versatile optical properties have been exploited in drug delivery [2], fluorescent inks [3], sensors [4], optoelectronics [5], photocatalysis [6], and light‑emitting devices [7]. CD synthesis typically employs hydrothermal treatment, electrochemical oxidation, or microwave assistance [8]. A hallmark of CDs is their excitation‑dependent emission, wherein the PL peak shifts with the excitation wavelength due to size distribution, hetero‑atom doping, solvent polarity, surface defects, and passivation states [9]. However, reports of excitation‑independent behavior remain scarce, limiting the development of tunable optical devices. Here we demonstrate that, by controlling the dilution of a freshly prepared CD solution, the emission peak undergoes a pronounced blue‑shift while simultaneously achieving excitation independence at low concentrations. This phenomenon is attributed to the dissociation of nanoclusters into individual CDs, thereby reducing surface‑state contributions and revealing the intrinsic core emission.
Methods
Reagents and Chemicals
Citric acid monohydrate (99.5 %) was sourced from SCR (Shanghai, China) and ethylenediamine was purchased from Tianzheng Reagent (Tianjin, China). Deionized water (18.25 MΩ cm) was obtained from a laboratory water purifier. All reagents were used as received.
Preparation of Carbon Dots
Aqueous CD solutions were prepared by dissolving 1.0507 g citric acid and 335 µL ethylenediamine in 10 mL deionized water. The mixture was stirred, transferred to a Teflon‑lined autoclave, and heated at 150 °C for 5 h. After cooling to room temperature, the yellowish CD solution was diluted (1 mL to 5–400 mL water) to study concentration effects; the solution gradually turned colorless with increasing dilution.
Characterization
Photoluminescence (PL) measurements were performed on a Hitachi F4500 fluorescence spectrophotometer. Absorption spectra were recorded on a Shimadzu UV-3101PC spectrometer. Fourier‑transform infrared (FTIR) spectra were acquired with a Bruker VERTEX spectrometer. TEM imaging used a FEI Tecnai G2 20S‑twin; dynamic light scattering (DLS) was carried out on a Malvern Zetasizer Nano ZS; X‑ray diffraction (XRD) on a Bruker D8; Raman spectroscopy on a LabRAM HR Evolution (Horiba) with 532 nm excitation; X‑ray photoelectron spectroscopy (XPS) on a PHI 5000 Versa Probe; and atomic force microscopy (AFM) on a MultiMode scanning probe microscope.
Results and Discussion
Transmission electron microscopy (TEM) confirmed the formation of uniform, spherical CDs with an average diameter of 3.6 nm (Fig. 1a). High‑resolution TEM revealed lattice fringes of 0.295 nm, corresponding to the (002) plane of graphitic carbon, indicating a crystalline sp2 core (Fig. 1b). XRD patterns showed a broad peak at 20.24°, characteristic of graphitic interlayer spacing, while Raman spectra lacked distinct G and D bands, supporting the coexistence of crystalline graphitic cores and amorphous sp3 regions (Fig. S2).
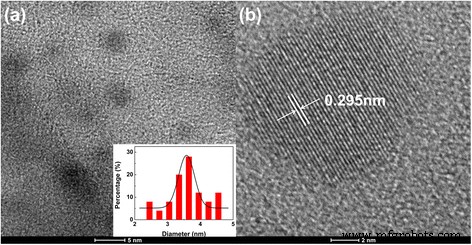
TEM and HRTEM images of as‑prepared CDs. a TEM image (inset shows size distribution). b HRTEM of a representative CD showing the crystalline core.
The as‑prepared aqueous CD solution exhibited a yellowish color and bright blue fluorescence under 365 nm UV excitation (Fig. 2a). UV‑vis absorption displayed a π→π* band at 243 nm and an n→π* transition at 345 nm, indicative of C=C and C=O functional groups. FTIR spectra confirmed abundant surface oxygenated moieties (C–O–C, C=O, –OH, and –NH), while XPS analysis quantified elemental composition and chemical states: C 1s peaks at 284.56, 285.66, 287.7 eV (C=C/C–C, C–O, C=O); N 1s peaks at 399.7 and 400.7 eV (pyrrolic‑like and graphitic‑like N); O 1s peaks at 531.55 and 532.31 eV (C=O, C–OH/C–O–C). These data collectively confirm a graphitic core decorated with oxygen‑containing surface groups (Fig. 2b).
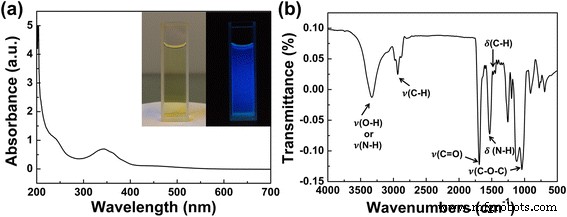
UV‑vis absorption and FTIR spectra of as‑prepared CDs. a Absorption (inset: photographs under natural light and 365 nm). b FTIR spectrum.
In a highly concentrated solution (1 mL → 25 mL water), the CDs exhibited classic excitation‑dependent PL: the emission peak red‑shifted with increasing excitation wavelength from 330 to 480 nm, peaking at 481 nm under 420 nm excitation (Fig. 3a). Using quinine bisulfate (QY = 0.56) as a reference, the quantum yield (QY) reached 74.8 %, attesting to the high radiative efficiency of these CDs. Importantly, the PL intensity increased markedly as the concentration decreased (Fig. 3c), a trend attributable to reduced collisional and self‑absorption quenching in dilute solutions.
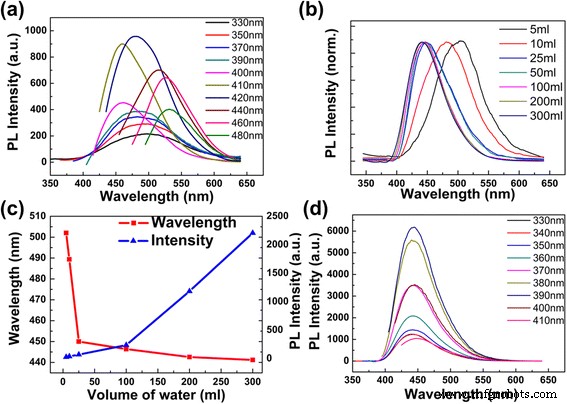
PL spectra of CDs at varying dilution. a PL of 1 mL CDs with 5 mL water (pH 10.41). b Normalized emission vs. dilution at 330 nm excitation. c PL intensity and peak shift vs. added water volume. d Emission of CDs diluted 300 mL water.
Dilution beyond 300 mL water yielded a single, concentration‑independent emission peak at 443 nm that remained invariant across excitation wavelengths (Fig. 3d). The maximum intensity was observed under 390 nm excitation. Adjusting the pH of the diluted solutions (10.2–12.08) did not alter the emission peak or intensity, confirming that concentration—not pH—governs the observed behavior.
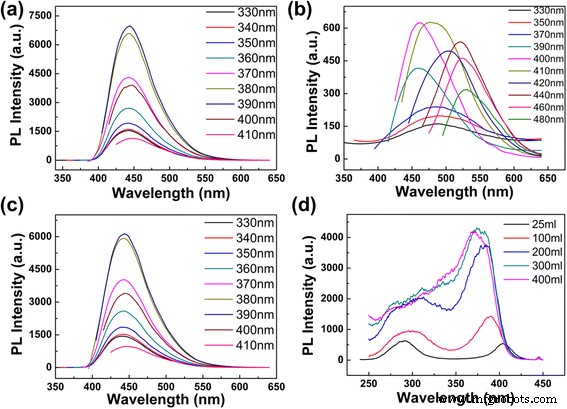
Emission and excitation spectra under different dilution and pH. a 300 mL water (pH 10.41). b 5 mL water (pH 12.08). c 300 mL water (pH 12.08). d Excitation spectra at 445 nm.
Excitation spectra of CDs at varying concentrations revealed two distinct excitation peaks: a 290 nm peak that diminishes with dilution, and a 400 nm peak that becomes more pronounced and blue‑shifts to 370 nm (Fig. 4d). Fluorescence lifetime measurements of a 1 mL → 25 mL diluted sample yielded an average lifetime of 11.85 ns, with bi‑exponential decay components (5.11 ns, 35.08 % and 13.28 ns, 64.92 %). These multiple lifetimes reflect heterogeneous emissive centers, likely arising from surface states and core states (Fig. S7).
Dynamic light scattering (DLS) and atomic force microscopy (AFM) analyses demonstrated that high‑concentration CDs form nanosized clusters (~34 nm hydrodynamic diameter) that dissociate into individual particles (~15 nm) upon dilution (Fig. S8, S9). This aggregation–disaggregation process modulates the surface polarity: clustered CDs exhibit highly polar surfaces (rich in –CO and –OH groups), giving rise to excitation‑dependent, longer‑wavelength emission; individual CDs possess lower surface polarity, allowing intrinsic sp2 core emission that is excitation‑independent and blue‑shifted (Fig. 5).
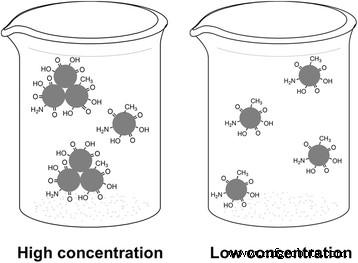
Illustration of concentration‑dependent emissive behavior. Left high‑concentration clusters; Right dispersed individual CDs.
Conclusions
We have successfully synthesized blue‑emitting CDs via a hydrothermal route that display exceptional aqueous dispersibility, a high quantum yield of 74.8 %, and a concentration‑tunable PL profile. The emission peak shifts from 480 to 443 nm as the solution is diluted, culminating in an excitation‑independent, short‑wavelength peak at 440 nm in dilute conditions. This dual‑emission mechanism—core‑dominated at low concentration and surface‑state‑dominated at high concentration—provides a versatile platform for designing tunable fluorescent probes and optoelectronic devices.
Abbreviations
- AFM:
Atomic force microscopy
- CDs:
Carbon dots
- DLS:
Dynamic light scattering
- FTIR:
Fourier transformed infrared
- PL:
Photoluminescence
- TEM:
Transmission electron microscopy
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- Fast Microwave‑Synthesis of Pyridinic‑Rich N,S‑Co‑Doped Carbon Quantum Dots with Superior Photoluminescence and Catalase‑Mimicking Activity
- Highly Efficient Magnetic Carbon Microspheres for Reusable Sulfonamide Removal from Water
- Hydrothermal Synthesis of Blue‑ and Green‑Emitting Carbon Quantum Dots from Tofu Wastewater
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Post‑Treatment Strategies to Modulate Photoluminescence of Nitrogen‑Doped Carbon Dots: Solvent, Reduction, and Metal‑Induced Enhancements
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Carbon Dots: Pioneering Next-Generation Nanothermometers – A Comprehensive Review
- Bright, Multicolor N‑Doped Carbon Dots from Ascorbic Acid and Phenylenediamine: Tunable Emission via Solvent Polarity and pH
- Stable Carbon‑Dot Frameworks with Superior Antibacterial Efficacy



