S,N‑Co‑Doped Graphene Quantum Dot/TiO₂ Composites for High‑Efficiency Visible‑Light Photocatalytic Hydrogen Generation
Abstract
We have synthesized S,N‑co‑doped graphene quantum dots (S,N‑GQDs) and integrated them with commercial TiO₂ (P25) via a straightforward hydrothermal route. The resulting S,N‑GQD/P25 composites exhibit markedly enhanced photocatalytic hydrogen production under 400–800 nm irradiation, achieving a 3.6‑fold increase over pure P25 without the need for noble‑metal co‑catalysts. The superior performance is attributed to the expanded visible‑light absorption of the composites and the efficient separation and transfer of photogenerated charge carriers. This study offers a scalable strategy to develop robust, metal‑free photocatalysts for solar‑to‑hydrogen conversion.
Background
Hydrogen energy represents a clean, high‑energy‑density alternative that can be produced from water and sunlight—two of the most abundant resources on Earth. While numerous semiconductors (e.g., CdS, SiC) have been investigated for photocatalytic H₂ evolution, TiO₂ remains the industry standard due to its low cost, non‑toxicity, chemical stability, and long service life. However, its wide bandgap (3.2 eV) limits activity to the UV region and rapid electron‑hole recombination further suppresses solar‑to‑hydrogen efficiency.
Strategies such as transition‑metal doping, noble‑metal deposition, and organic sensitization have been explored to overcome these limitations. Recently, carbon‑based nanomaterials—including graphene and carbon nanotubes—have been coupled with TiO₂ to enhance charge transport and reduce recombination. Nevertheless, pristine graphene’s zero bandgap and broad light absorption can block light reaching TiO₂, diminishing overall photocatalytic performance.
Graphene quantum dots (GQDs) offer a promising alternative. With lateral dimensions below 10 nm, GQDs combine the superior electronic properties of graphene with enhanced water solubility, chemical stability, and tunable optical bandgaps. Doping GQDs with heteroatoms such as nitrogen and sulfur broadens their visible‑light absorption, increases carrier mobility, and improves chemical robustness, making them attractive for photocatalytic applications.
In this work, we report a simple hydrothermal synthesis of S,N‑co‑doped GQDs and their integration with P25 TiO₂. The composites demonstrate superior hydrogen generation under UV‑visible and visible‑only irradiation, without requiring noble‑metal cocatalysts. Detailed mechanistic insights are provided through photophysical and electrochemical studies.
Methods
Synthesis of S,N‑GQDs
Citric acid (1.26 g, 6 mmol) and thiourea (1.38 g, 18 mmol) were dissolved in 30 mL DMF, stirred to form a clear solution, and then transferred to a 50 mL Teflon‑lined autoclave. The autoclave was heated to 180 °C for 8 h, followed by natural cooling. The resulting precipitate was collected by adding ethanol, centrifuged at 10,000 rpm for 15 min, and dried under vacuum.
Preparation of S,N‑GQD/P25 Composites
In a typical synthesis, 0.5 g P25 and 5 mL S,N‑GQD solution (2 mg mL⁻¹) were dispersed in 20 mL deionized water and stirred for 4 h at room temperature to form a homogeneous suspension. This mixture was then subjected to hydrothermal treatment at 150 °C for 6 h in a 40 mL Teflon‑sealed autoclave. The composites were isolated by centrifugation at 4,000 rpm for 5 min, washed with water, and dried at 50 °C overnight. To assess loading effects, composites containing 0–10 wt% S,N‑GQDs were prepared.
Characterization
Transmission electron microscopy (TEM) and high‑resolution TEM (HRTEM) were performed on a JEOL JEM‑2100 F (200 kV). X‑ray diffraction (XRD) patterns were recorded on a Rigaku D/max‑2500 diffractometer (Cu Kα, 40 kV, 300 mA). Fourier‑transform infrared (FTIR) spectra were obtained with a Nicolet 6700 spectrometer. Raman spectra were collected using a NEXUS670 Raman system. UV‑vis absorption spectra were measured on a Lambda 950 spectrophotometer.
Photocatalytic Hydrogen Generation
Fifty milligrams of each photocatalyst were dispersed in 100 mL aqueous solution containing 10 mL methanol as a sacrificial agent. Hydrogen evolution was monitored by online gas chromatography. UV‑visible irradiation was supplied by a 300 W Xe lamp; a 400 nm long‑pass filter isolated the visible region.
Photoelectrochemical Measurements
Transient photocurrent responses were recorded on a three‑electrode setup: Pt counter, saturated calomel reference, and the photocatalyst coated on ITO glass as the working electrode. The working electrode was prepared by doctor‑blading a slurry of 0.05 g photocatalyst, 0.2 g PEG 20000, and 1.0 mL water onto ITO, followed by calcination at 450 °C for 30 min. The exposed area was ~2 cm², and the electrolyte was 0.5 M Na₂SO₄.
Results and Discussion
Figure 1 displays TEM images of the S,N‑GQDs and the S,N‑GQD/P25 composites. The GQDs are uniformly dispersed without aggregation; HRTEM reveals (0–110) lattice fringes with a spacing of ~0.24 nm, confirming their graphite‑like structure. AFM height profiles indicate a thickness of 0.8–1.2 nm, characteristic of few‑layer GQDs. When mixed with P25, the GQDs decorate the TiO₂ surface, as shown in Fig. 1d.
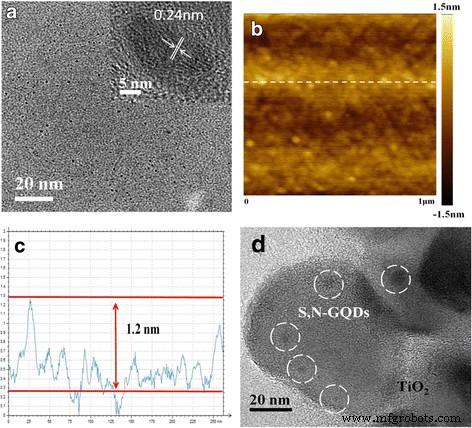
Morphology characterizations. a TEM and HRTEM images of the S,N‑GQDs. b, c An AFM image and the height profile of the S,N‑GQDs. d A TEM image of the S,N‑GQD/P25 composites
XRD patterns (Fig. 2) confirm that the composites retain the anatase/rutile mixture of commercial P25, with additional broad peaks at 25.6° corresponding to the (0 0 2) plane of graphite. The low GQD loading (<10 wt%) does not alter the TiO₂ crystal lattice.
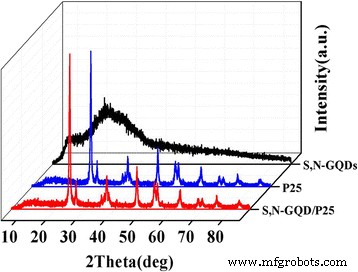
XRD patterns of the P25, S,N‑GQDs, and S,N‑GQD/P25 composites
FTIR spectra (Fig. 3) exhibit characteristic O–H, C=O, C=S, C–S, and N–H vibrations in the GQDs, while the Ti–O stretching band of TiO₂ shifts slightly in the composites, confirming interfacial bonding. Raman spectra (Fig. 4) reveal the D and G bands of the GQDs in the composites, further indicating successful loading.
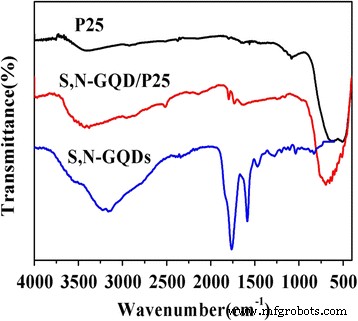
FTIR spectra of P25, S,N‑GQDs, and S,N‑GQD/P25
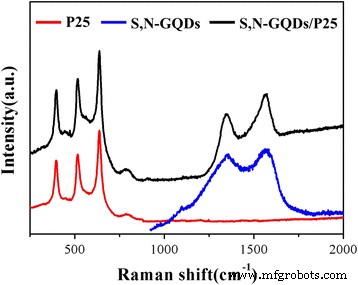
Raman spectra of P25, S,N‑GQDs and S,N‑GQD/P25
UV‑vis spectroscopy (Fig. 5) shows two absorption bands of the S,N‑GQDs at 345 and 462 nm, indicating a reduced bandgap of 2.5 eV. The composites display extended absorption up to 800 nm, far beyond pure P25, highlighting the sensitizing role of the GQDs.
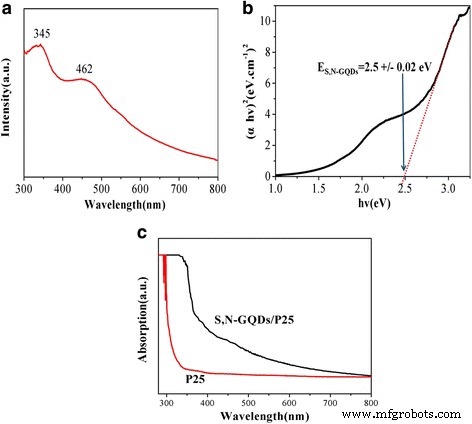
UV-vis measurements. a The UV-vis absorption spectrum of the S,N‑GQDs. b The corresponding Tauc plot of the S,N‑GQDs. c The UV-vis absorption spectra of the P25 and S,N‑GQD/P25
Photocatalytic hydrogen production (Fig. 6) demonstrates that 3 wt% S,N‑GQD loading yields the highest rate (5.7 µmol h⁻¹), 3.6 times that of pure P25. Excessive loading (>5 wt%) reduces activity, likely due to light scattering and reduced active surface area. Visible‑light only experiments (Fig. 7) confirm that the composites are active under 400–800 nm illumination, whereas pure P25 shows negligible activity.
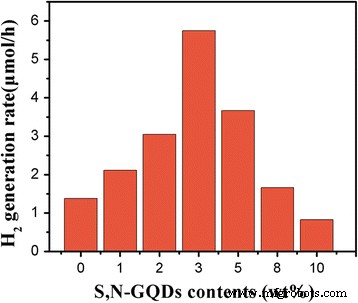
Photocatalytic H₂ generation rates of pure P25 and S,N‑GQD/P25 composites with different amounts of S,N‑GQDs under UV–vis light
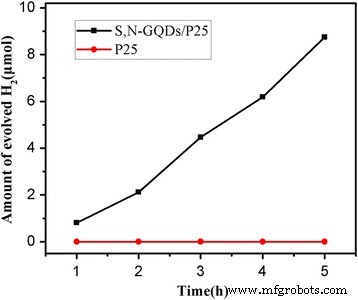
Photocatalytic H₂ generation rates of pure P25 and S,N‑GQD/P25 composites (3 wt.% S,N‑GQD) under visible light
Stability tests (Fig. 8) show that the 3 wt% composite retains >95% of its activity over three consecutive cycles, indicating excellent durability.
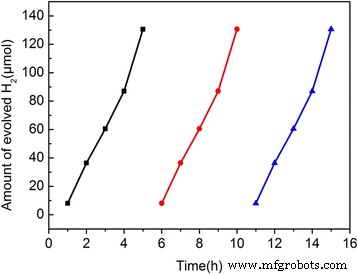
Three repeat cycle experiments of 3 wt% S,N‑GQDs/TiO₂
Transient photocurrent measurements (Fig. 9) reveal that the composite delivers nine‑fold higher photocurrent density than P25 alone, confirming efficient charge separation. The S,N‑GQDs act as an electron reservoir and a light‑absorbing sensitizer, facilitating rapid electron transfer to TiO₂’s conduction band.
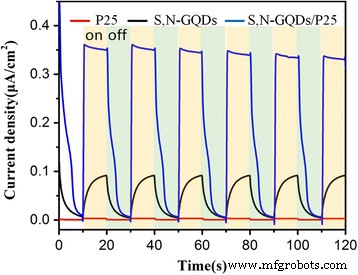
The transient photocurrent response of P25, S,N‑GQDs, and S,N‑GQD/P25 composites under UV–vis light irradiation
Photoluminescence (PL) spectra (Fig. 10) show quenching of TiO₂ emission upon GQD loading, indicating suppressed recombination. The quenching efficiency increases with GQD content, supporting the role of GQDs in charge‑carrier separation.
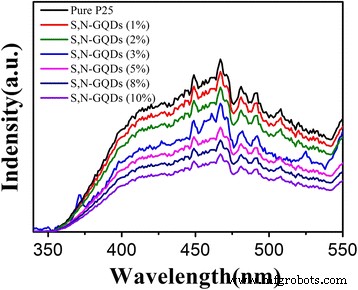
PL spectra of pure P25 and S,N‑GQD/P25 composites with different amounts of S,N‑GQDs. Excitation wavelength: 280 nm
Figure 11 summarizes the proposed mechanism: under UV light, electrons from TiO₂ are trapped by S,N‑GQDs, reducing recombination; under visible light, the GQDs act as photosensitizers, donating electrons to TiO₂’s conduction band. The combined effect results in efficient hydrogen evolution, as described by the equations (1)–(6).
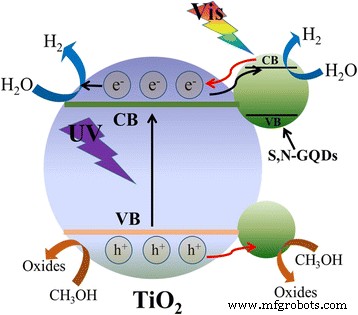
Proposed mechanism for the photocatalytic H₂ evolution of S,N‑GQD/P25 composites under UV‑vis light irradiation
$$\mathrm{photocatalyst}+hv\rightarrow\mathrm{e}^{-}+\mathrm{h}^{+}$$ $$\mathrm{h}^{+}+\mathrm{H_2O}\rightarrow\cdot\mathrm{OH}+\mathrm{H}^{+}$$ $$\mathrm{CH_3OH}+\cdot\mathrm{OH}\rightarrow\cdot\mathrm{CH_2OH}+\mathrm{H_2O}$$ $$\cdot\mathrm{CH_2OH}\rightarrow\mathrm{HCHO}+\mathrm{H}^{+}+\mathrm{e}^{-}$$ $$2\mathrm{H_2O}+2\mathrm{e}^{-}\rightarrow\mathrm{H_2}+2\mathrm{OH}^{-}$$ $$\mathrm{Overall:\ CH_3OH}\rightarrow\mathrm{HCHO}+\mathrm{H_2}$$
Conclusions
We have successfully fabricated S,N‑co‑doped GQD/TiO₂ composites that display a broadened visible‑light absorption and dramatically enhanced photocatalytic hydrogen production. The 3 wt% composite delivers a 3.6‑fold higher rate than pristine P25 under UV‑visible irradiation and remains active under visible light alone. The composites also show robust cycling stability, underscoring their potential as metal‑free photocatalysts for sustainable hydrogen generation.
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Synergistic Graphene-Modified TiO₂ Composites Deliver Superior Photocatalytic Activity
- Pd‑Loaded Zr‑MOF for Visible‑Light Photocatalytic Hydrogen Generation
- Highly Efficient Hydrogen Production via Hierarchical ZnO@TiO₂ Hollow Spheres
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Efficient Visible‑Light Hydrogen Production with Eosin Y‑Sensitized g‑C3N4/GO Hybrid Loaded with PtNi Alloy Cocatalyst
- Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- Efficient Charge Transfer in Au/CdSe Janus Nanoparticles Boosts Photocatalytic Hydrogen Production
- CKAP4-Linked Silicon Quantum Dot Micelles: A Novel Targeted Imaging Agent for Lung Cancer



