Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions
Abstract
We present a simple, scalable two‑step route to Ag3PO4/TiO2 heterostructures that deliver outstanding visible‑light photocatalytic performance. The composites were comprehensively characterized by X‑ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), high‑resolution TEM (HR‑TEM), energy‑dispersive X‑ray spectroscopy (EDX), X‑ray photoelectron spectroscopy (XPS), and UV‑vis diffuse reflectance spectroscopy (DRS). The highly crystalline structures exhibit a uniform distribution of TiO2 nanoparticles on Ag3PO4 surfaces, yielding a pronounced Z‑scheme charge‑transfer pathway. In Rhodamine B (RhB) degradation tests, TiO2400/Ag3PO4 achieved ~100 % removal within 25 min under visible illumination, with a reaction rate constant of 0.02286 min-1—twice that of Ag3PO4 alone and 6.6 times higher than pristine TiO2400. The catalyst maintained >95 % activity after four recycles. Radical‑trapping experiments identified holes (h+) and superoxide anions (O2·-) as the primary oxidative species, while hydroxyl radicals (·OH) contributed partially. A detailed Z‑scheme mechanism is proposed, explaining the synergistic electron–hole separation and the suppression of Ag3PO4 photochemical corrosion.
Background
Semiconductor photocatalysts have become pivotal for environmental remediation and solar‑energy conversion, owing to their low cost and chemical stability. Titanium dioxide (TiO2), a prototypical photocatalyst, offers excellent photocorrosion resistance and high oxidative potential, yet its wide band gap (3.0–3.2 eV) limits activity to the UV region. Silver orthophosphate (Ag3PO4) possesses a narrower band gap (~2.45 eV) and a quantum yield exceeding 90 % under visible light, but suffers from photocorrosion that generates metallic Ag, diminishing long‑term performance. Coupling TiO2 with Ag3PO4 can harness complementary band structures, enhance charge separation, and mitigate Ag3PO4 degradation. Z‑scheme heterojunctions, in particular, preserve the strong redox potentials of both components while enabling efficient interfacial electron transfer.
Methods
Hydrothermal Preparation of Nano‑TiO2
A mixed solution of P123 (0.4 g) in ethanol (7.6 mL) and deionized water (0.5 mL) was stirred until complete dissolution (solution A). Separately, butyl titanate (2.5 mL) and concentrated HCl (12 mol L-1, 1.4 mL) formed solution B, which was added dropwise to A. After 30 min stirring, ethylene glycol (32 mL) was introduced and the mixture was stirred another 30 min. The suspension was sealed in an autoclave at 140 °C for 24 h, then cooled, centrifuged, washed, and dried at 80 °C for 8 h. Calcination at 300, 400, or 500 °C produced TiO2300, TiO2400, and TiO2500, respectively.
Preparation of TiO2/Ag3PO4 Photocatalyst
TiO2 (0.1 g) was dispersed in 30 mL AgNO3 solution (0.612 g) by ultrasound for 30 min. Subsequently, 30 mL Na2HPO4.12H2O (0.43 g) was added, and the mixture stirred for 120 min at room temperature. After centrifugation, washing with deionized water and anhydrous ethanol, the solids were dried at 60 °C. The resulting composites were labeled TiO2300/Ag3PO4, TiO2400/Ag3PO4, and TiO2500/Ag3PO4. Pure Ag3PO4 was synthesized under identical conditions without TiO2.
Characterization
XRD patterns were recorded on a D/MaxRB diffractometer (Cu Kα, 35 kV, 0.02° s-1, 10–80°). SEM (JEOL JSM‑6510/2100) and TEM (JEOL JSM‑2100) provided morphological insights at 10 kV. EDX coupled with TEM confirmed elemental composition. XPS (ESCALAB 250, 300 W Cu Kα, 3 × 10-9 mbar) supplied valence‑state information, calibrated to the C 1s line at 284.6 eV. UV‑vis DRS spectra (400–800 nm) were obtained with a Lambda 35 spectrophotometer, and band gaps were derived from Kubelka–Munk plots.
Photocatalytic Activity Measurement
Photodegradation of RhB (10 mg L-1) was carried out in a 50 mL suspension containing 50 mg of catalyst. After 30 min in the dark (to reach adsorption equilibrium), the mixture was irradiated with a 1000‑W Xe lamp (visible light) while maintaining the temperature at 25 °C using a water bath. Absorbance at 553 nm was recorded every 5 min; the decolorization ratio was calculated as Ct/C0. The experiment was repeated four times to evaluate recyclability.
Results and Discussion
XRD Analysis
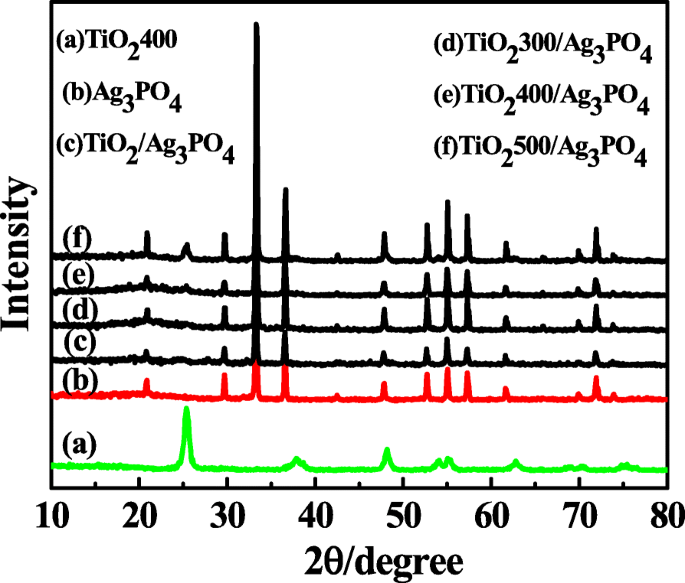
The XRD patterns (Fig. 1) confirm that TiO2400 crystallizes in the anatase phase (JCPDS 71‑1166). Ag3PO4 peaks match JCPDS 70‑0702, while the composites display superimposed reflections of both constituents, indicating successful heterojunction formation. The anatase (101) peak shifts to higher angles with increased calcination temperature, reflecting enhanced crystallinity.
Morphology and Composition
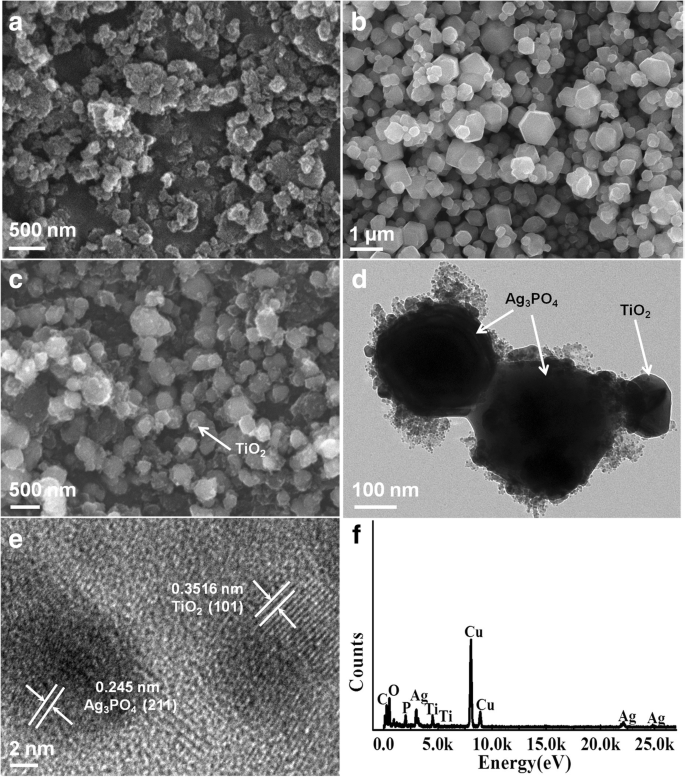
SEM images (Fig. 2) reveal TiO2400 as spherical nanoparticles (100–300 nm), Ag3PO4 as regular hexagonal crystals (0.1–1.5 µm), and TiO2400/Ag3PO4 as TiO2 nanoparticles uniformly anchored to Ag3PO4 surfaces. TEM and HRTEM (Fig. 2d–e) confirm lattice spacings of 0.3516 nm (TiO2 (101)) and 0.245 nm (Ag3PO4 (211)), indicative of intimate contact. EDX mapping (Fig. 2f) confirms the presence of Ti, O, Ag, and P, validating the composite structure.
XPS Analysis
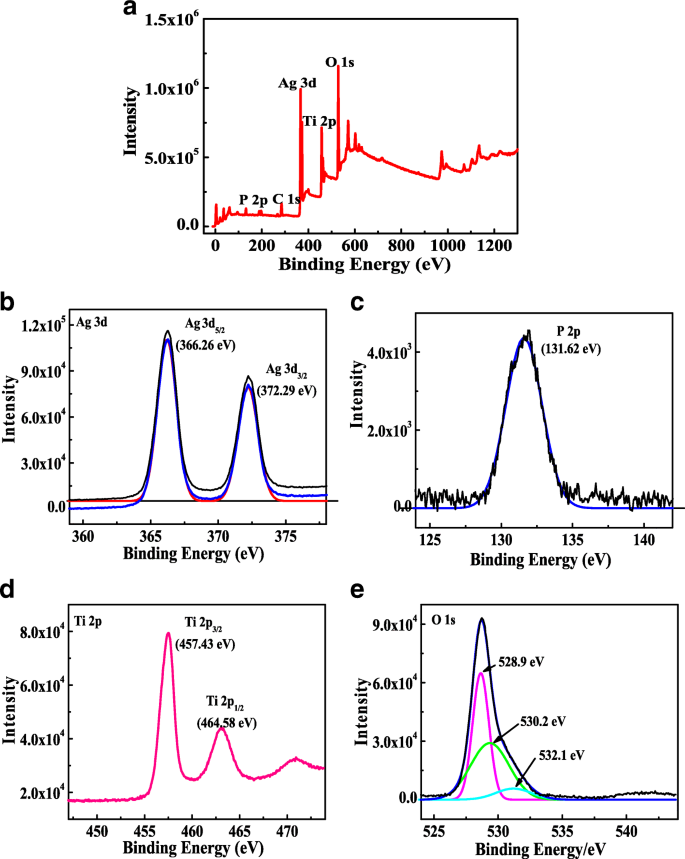
Survey spectra (Fig. 3a) show Ti, O, Ag, P, and C. Ag 3d peaks at 366.26 and 372.29 eV confirm Ag+ oxidation state. P 2p at 131.62 eV indicates PO43-. Ti 2p peaks at 457.43 and 464.58 eV correspond to Ti4+. O 1s deconvolution reveals lattice oxygen (528.9 eV), TiO2 oxygen (530.2 eV), and surface hydroxyls (532.1 eV). These results corroborate the successful coupling of TiO2 and Ag3PO4.
Optical Properties
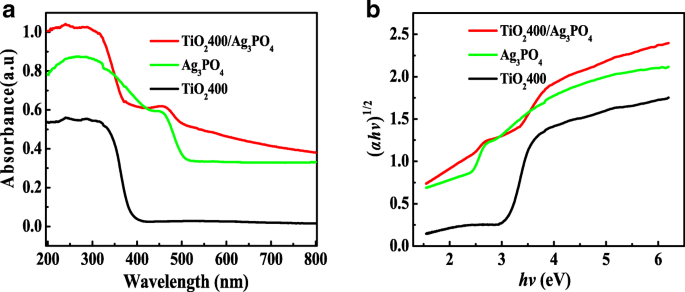
DRS spectra (Fig. 4a) show TiO2400 absorbing at ~400 nm and Ag3PO4 at ~500 nm. The composite extends absorption up to 700 nm, reflecting strong interfacial interaction. Kubelka–Munk plots (Fig. 4b) yield band gaps of 3.10 eV (TiO2400), 2.45 eV (Ag3PO4), and 2.75 eV (TiO2400/Ag3PO4), confirming the suitability for visible‑light activation.
Photocatalytic Performance
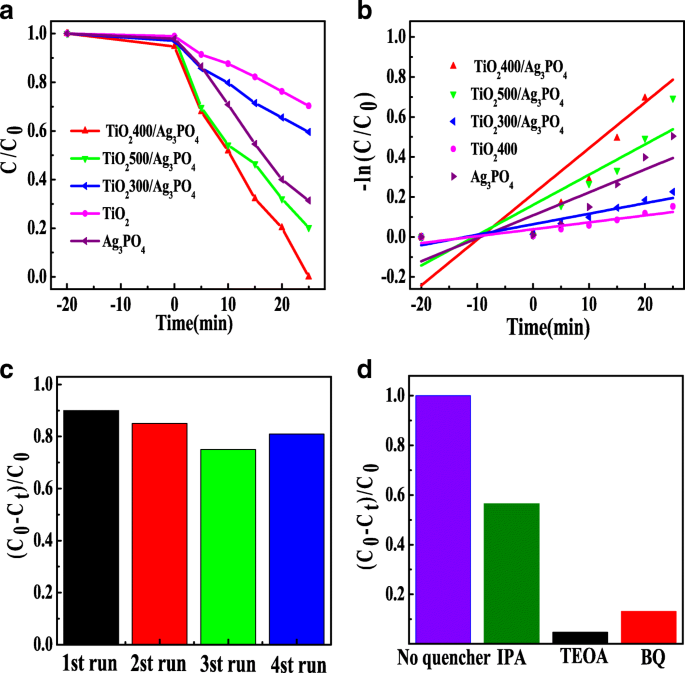
Under visible light, TiO2400 alone degrades 30 % of RhB in 25 min, whereas Ag3PO4 achieves 69 %. The TiO2400/Ag3PO4 composite reaches complete decolorization (≈100 %) within the same period (Fig. 5a). Kinetic analysis follows pseudo‑first‑order behavior; the rate constants are 0.00345, 0.01148, 0.00525, 0.02286, and 0.01513 min-1 for TiO2400, Ag3PO4, TiO2300/Ag3PO4, TiO2400/Ag3PO4, and TiO2500/Ag3PO4, respectively (Table 1). The TiO2400/Ag3PO4 catalyst shows excellent recyclability, retaining >95 % activity after four cycles (Fig. 5c). Trapping experiments with IPA, BQ, and TEOA reveal that holes and superoxide anions dominate the oxidative process; hydroxyl radicals contribute modestly (Fig. 5d).
Z‑Scheme Mechanism
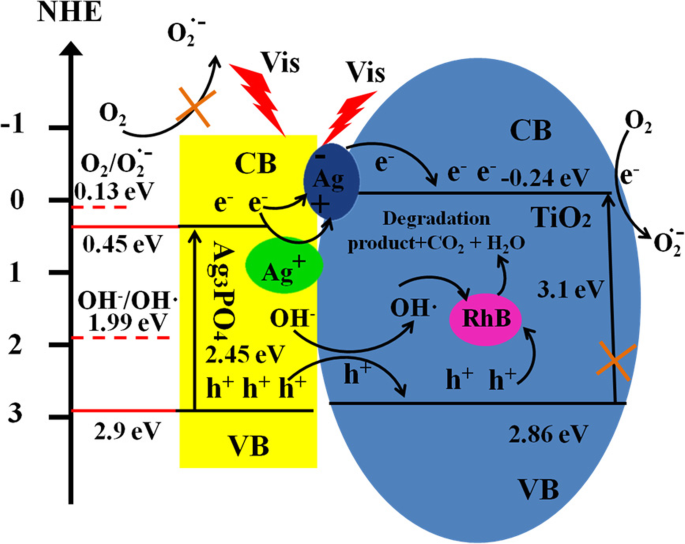
Based on band‑edge alignment (ECB ≈ 0.45 eV, EVB ≈ 2.90 eV for Ag3PO4; ECB ≈ –0.24 eV, EVB ≈ 2.86 eV for anatase TiO2), a Z‑scheme charge transfer is favored: photogenerated electrons in TiO2 CB recombine with holes in Ag3PO4 VB, leaving highly oxidative holes in TiO2 VB and strongly reducing electrons in Ag3PO4 CB. This configuration preserves the strong oxidizing power of TiO2 holes and the reducing ability of Ag3PO4 electrons, enabling efficient RhB oxidation and O2 reduction to superoxide. The simultaneous formation of small Ag nanoparticles under illumination further facilitates electron migration and protects Ag3PO4 from photocorrosion. A schematic of this pathway is shown in Scheme 1.
Conclusions
The TiO2400/Ag3PO4 heterojunction, fabricated via a facile two‑step method, delivers superior visible‑light photocatalytic activity, achieving complete RhB degradation in 25 min with a rate constant of 0.02286 min-1. The composite maintains high stability over repeated cycles, owing to efficient charge separation and suppression of Ag3PO4 corrosion. Hole and superoxide anion species dominate the degradation process, while a Z‑scheme mechanism rationalizes the observed synergy. This study demonstrates a practical route to high‑performance, durable photocatalysts for environmental remediation.
Availability of Data and Materials
The authors confirm that all materials and data are freely available to readers without restrictive transfer agreements. Full datasets are provided within the article.
Abbreviations
- BQ
- p‑benzoquinone
- DRS
- UV‑vis diffuse reflectance spectroscopy
- EDX
- energy‑dispersive X‑ray spectroscopy
- HR‑TEM
- high‑resolution transmission electron microscopy
- IPA
- isopropanol
- RhB
- rhodamine B
- SEM
- scanning electron microscopy
- TEM
- transmission electron microscopy
- TEOA
- triethanolamine
- XPS
- X‑ray photoelectron spectroscopy
- XRD
- X‑ray diffraction
Nanomaterials
- S,N‑Co‑Doped Graphene Quantum Dot/TiO₂ Composites for High‑Efficiency Visible‑Light Photocatalytic Hydrogen Generation
- Enhanced Photocatalytic Degradation of Rhodamine B Using an ATP/TiO₂/Ag₃PO₄ Ternary Nanocomposite Under Simulated Solar Light
- High‑Efficiency ZnO@NiO Core–Shell Heterostructures for Ultraviolet Photocatalysis of Organic Dyes
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Enhanced Visible-Light Photocatalysis by Anchoring Plasmonic Ag@AgCl Nanocrystals on ZnCo₂O₄ Microspheres
- Synthesis and Enhanced Photocatalytic Degradation of Rhodamine B by Ag3PO4/T‑ZnO Whisker Heterostructures
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping



