High‑Efficiency ZnO@NiO Core–Shell Heterostructures for Ultraviolet Photocatalysis of Organic Dyes
Abstract
ZnO@NiO core–shell heterostructures, fabricated by electrochemical deposition onto carbon fiber cloth, exhibit superior photocatalytic activity and excellent reusability. Their performance was evaluated by monitoring the UV‑induced degradation of rhodamine B and methyl orange. Compared with pristine ZnO nanorods and NiO nanosheets, the ZnO@NiO composites achieved a 95 % degradation of methyl orange in 180 min, owing to the formation of an efficient p–n heterojunction that promotes charge‑carrier separation. Photocurrent measurements revealed an eight‑fold increase in the ZnO@NiO heterostructures relative to bare ZnO, confirming enhanced electron–hole separation. The composites retained 90 % of their activity after three consecutive cycles, underscoring their durability and practical viability.
Background
Semiconductor photocatalysis has emerged as a green technology for environmental remediation and renewable energy generation. Among the widely studied semiconductors—TiO₂, ZnO, Cu₂O, CdS, C₃N₄—ZnO stands out due to its high electron mobility, tunable morphology, low cost, and non‑toxic nature. However, ZnO’s wide bandgap (3.37 eV) restricts activity to UV light, and rapid electron–hole recombination limits its photocatalytic efficiency. Moreover, photocorrosion hampers long‑term stability. Therefore, strategies to improve charge separation and broaden the spectral response remain critical.
Various approaches—doping, noble‑metal loading, and heterostructure engineering—have been explored to address these challenges. Nickel oxide, a p‑type semiconductor with a 3.5 eV bandgap and high hole mobility, is an attractive partner for ZnO due to its compatible lattice and potential to form type‑II band alignment. The resulting ZnO@NiO heterojunction can suppress recombination and facilitate efficient charge transfer, as demonstrated in recent reports on NiO/ZnO nanofibers and nanoneedles. Nonetheless, conventional synthesis routes often involve complex steps, hinder catalyst recovery, and limit scalability.
In this study, we employ a straightforward electrochemical deposition method on carbon fiber cloth to construct ZnO@NiO core–shell heterostructures. The cloth substrate enables easy separation and recycling of the catalyst, while the core–shell architecture enhances charge‑carrier dynamics, ultimately boosting photocatalytic performance.
Methods
Materials Preparation
ZnO nanorods were grown on pre‑cleaned carbon fiber cloth via electrochemical deposition from a 5 mM Zn(NO₃)₂·6H₂O/5 mM HMTA electrolyte at –0.9 V (vs. Ag/AgCl) for 30 min at 90 °C. After rinsing with deionized water and drying at 60 °C for 24 h, the cloth exhibited densely packed, 200 nm‑diameter ZnO nanorods.
NiO nanosheets were deposited by reducing 0.01 mol Ni(NO₃)₂·6H₂O in 500 mL water at –1 V for 10 min, followed by annealing at 400 °C for 2 h in air to crystallize NiO.
To form ZnO@NiO heterostructures, a NiO layer was electrochemically deposited onto the ZnO nanorods for 5, 10, or 15 min, yielding ZN1, ZN2, and ZN3, respectively. The process is illustrated in Figure 1.

Electrochemical fabrication of hierarchical ZnO@NiO core–shell heterostructures.
Material Characterization
Morphology and crystal structure were examined by FESEM (NoVaTM Nano SEM 250), XRD (Bruker D8 Advance), and TEM (Tecnai G2 F20). Elemental composition of ZN2 was confirmed by XPS (Thermo ESCALAB 250XI) and EDS mapping. Photoluminescence (PL) spectra were recorded using a JY‑630 micro‑Raman spectrometer with 325‑nm excitation.
Photocatalytic Activity
Photodegradation of RhB and MO (5 mg L⁻¹) was monitored under 500 W Hg lamp UV irradiation. Catalyst (2 cm × 1.5 cm) was immersed in 20 mL dye solution, equilibrated for 60 min in the dark, then irradiated. Absorbance at 554 nm (RhB) and 464 nm (MO) was measured with a UV‑Vis spectrophotometer (TU‑1900/1901) at room temperature.
Photocurrent Response Characterization
Photocurrent was measured in a 0.5 M Na₂SO₄ electrolyte (pH ≈ 7) using a standard three‑electrode cell and a 10 W UV lamp as the light source.
Results and Discussion
Figure 2 shows XRD patterns of ZnO nanorods, NiO nanosheets, and ZnO@NiO composites. The ZnO peaks at 2θ = 31.8°, 34.4°, 36.2°, and 47.5° correspond to the (100), (002), (101), and (103) planes of hexagonal wurtzite ZnO. NiO peaks at 37.0° and 42.9° match the (111) and (200) planes of cubic NiO. In the composites, NiO reflections intensify with increasing deposition time, confirming successful shell growth without altering ZnO crystallinity.
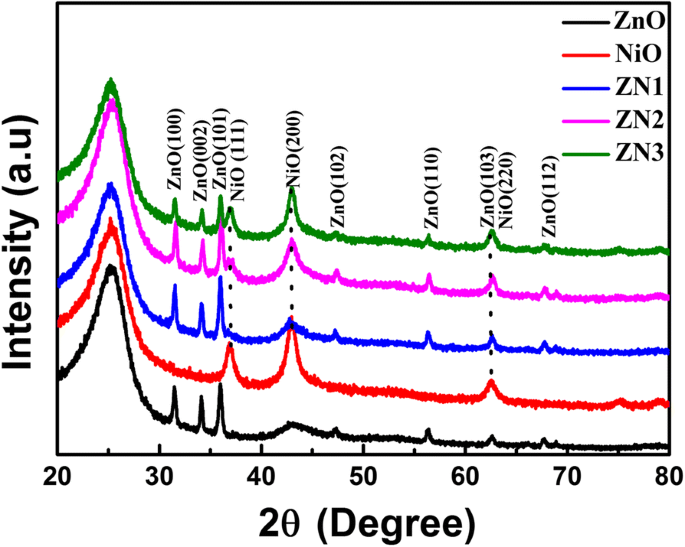
XRD patterns of ZnO, NiO, and ZnO@NiO composites.
FESEM images (Figure 3) reveal that NiO forms uniform sheet‑like layers on the cloth, while ZnO grows as smooth‑surfaced nanorods (~200 nm diameter). After NiO deposition, the composite diameter increases, and the surface becomes denser with 10‑min deposition, indicating optimal NiO coverage. Extended deposition to 15 min leads to interconnection of NiO sheets, consistent with XRD data.

SEM images of a carbon cloth, b NiO nanosheets, c ZnO nanorods, d ZN1, e ZN2, and f ZN3.
EDS mapping of ZN2 (Figure 4) confirms uniform distribution of Zn, Ni, and O, indicating a homogeneous core–shell structure. TEM analysis (Figure 5) shows a clear interface between ZnO nanorods (core) and NiO nanosheets (shell). HR‑TEM reveals lattice spacings of 0.26 nm (ZnO (002)) and 0.241 nm (NiO (111)), confirming crystalline integrity and intimate contact conducive to charge transfer.

a TEM image of ZnO@NiO heterostructure (ZN2). b High‑resolution TEM of ZN2.
XPS survey spectra (Figure 6) confirm the presence of Zn, Ni, O, and C. The Zn 2p₃/₂ peak at 1022.3 eV and Zn 2p₁/₂ at 1045.2 eV indicate Zn²⁺. Ni 2p₃/₂ peaks at 854.0, 856.1, and 861.1 eV correspond to Ni–O bonding, while Ni 2p₁/₂ peaks at 873.1 and 879.6 eV confirm Ni²⁺ states. The O 1s spectrum shows lattice oxygen (529.5 eV) and surface hydroxyl/adsorbed O₂ (532.2 eV).
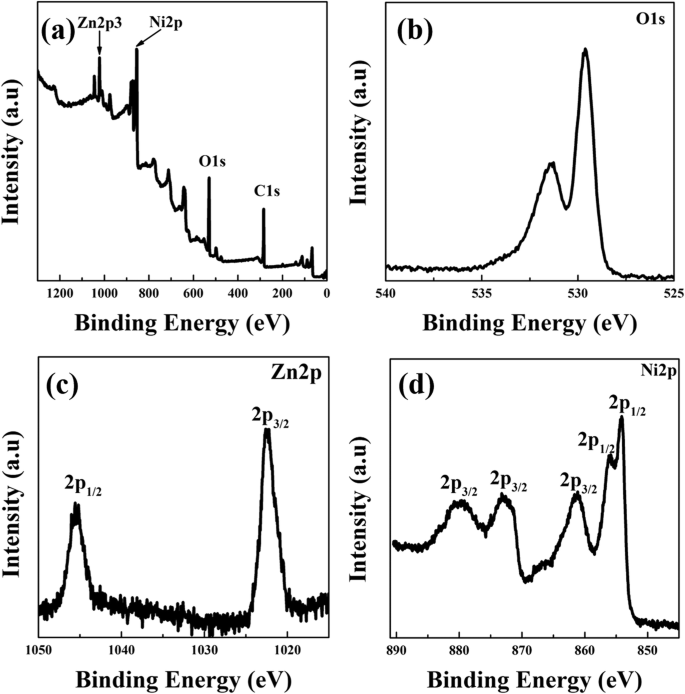
XPS spectra of ZN2. a Survey spectra. b O 1s. c Zn 2p. d Ni 2p spectra.
Photocatalytic performance (Figure 7) demonstrates that ZN2 outperforms both ZnO and NiO: after 180 min, 95 % of RhB and 95 % of MO were degraded under UV irradiation, whereas ZnO and NiO achieved only ~38 % and ~33 %, respectively. The pseudo‑first‑order rate constants (k) were 0.01656 min⁻¹ (ZN2), 0.00257 min⁻¹ (ZnO), and 0.00208 min⁻¹ (NiO). The optimal NiO thickness at 10 min deposition balances surface area and interfacial contact, as corroborated by BET surface area trends (Additional file 1).
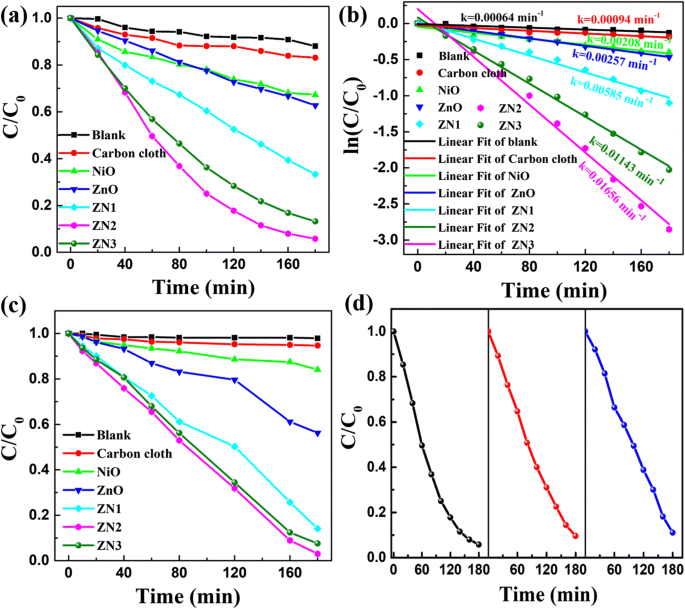
a Relative concentration (C/C₀) of RhB over ZN1–ZN3, ZnO, and NiO. b –ln(C/C₀) vs. time. c Relative concentration of MO over ZnO, NiO, and ZnO@NiO. d Repeated RhB degradation by ZN2.
Photocurrent measurements (Figure 8a) reveal that ZnO@NiO composites generate significantly higher photocurrent densities, peaking for ZN2. The rapid on/off response indicates efficient charge separation, attributable to the type‑II band alignment that drives electrons from NiO CB to ZnO CB and holes from ZnO VB to NiO VB. PL spectra (Figure 8b) show a suppressed emission for ZnO@NiO compared to ZnO, confirming reduced recombination.
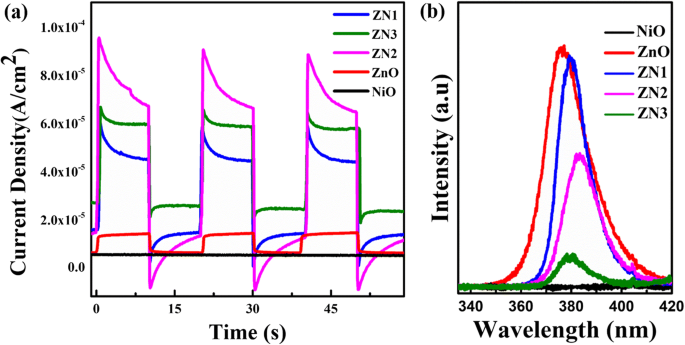
a Photocurrent response of NiO, ZnO, and ZnO@NiO under UV lamp (10 W). b PL spectra of ZnO, NiO, and ZnO@NiO.
Mechanistically, the p–n heterojunction generates an internal electric field that separates photogenerated carriers. Electrons transfer from NiO CB to ZnO CB, reacting with dissolved O₂ to form superoxide radicals (•O₂⁻), while holes migrate from ZnO VB to NiO VB and oxidize OH⁻ to hydroxyl radicals (•OH). These reactive species drive the efficient breakdown of organic dyes. The schematic in Figure 9 illustrates the band alignment.
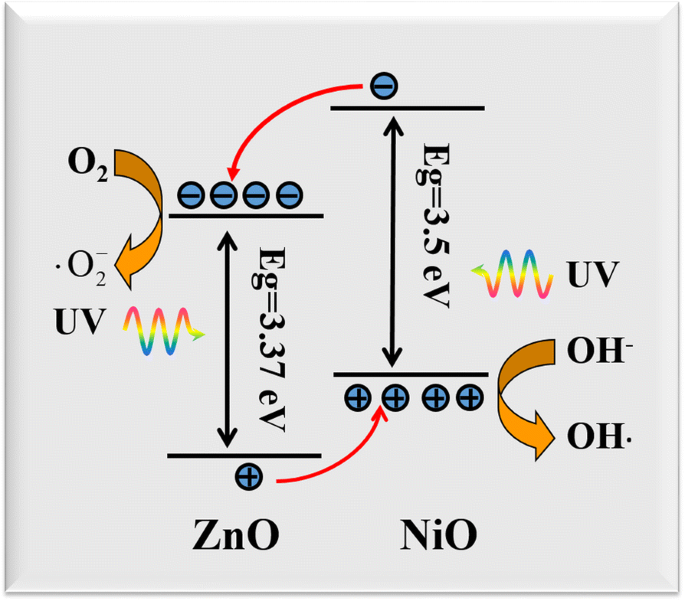
Energy band alignment between ZnO (n‑type) and NiO (p‑type).
Conclusions
We have successfully fabricated ZnO@NiO core–shell heterostructures via a scalable electrochemical deposition on carbon fiber cloth. The resulting composites exhibit markedly enhanced UV photocatalytic degradation of RhB and MO, achieving 95 % removal in 180 min and retaining 90 % activity after three cycles. The superior performance originates from the p–n heterojunction that facilitates rapid charge‑carrier separation, as confirmed by photocurrent and PL studies. Moreover, the cloth substrate enables straightforward catalyst recovery, underscoring the practical relevance of these materials for wastewater treatment and other environmental applications.
Abbreviations
- CB:
Conduction band
- EDS:
Energy dispersive X‑ray spectroscopy
- MO:
Methyl orange
- PL:
Photoluminescence
- RhB:
Rhodamine B
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
- VB:
Valence band
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- Benchmarking Lubrication Performance to Boost Reliability and Cut Costs
- Three Key Drivers for Superior Plant Performance
- Enhanced Photocatalytic Performance of ZnO/In₂O₃ Hybrid Nanostructures via Hydrothermal Synthesis
- Highly Efficient Hydrogen Production via Hierarchical ZnO@TiO₂ Hollow Spheres
- Enhanced Visible‑Light Photocatalysis of Methylene Blue and Phenol with N‑Doped ZnO/g‑C₃N₄ Nanocomposites
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions
- R‑Scheme Ti3C2 MXene/MoS2 2D/2D Heterojunctions: A Breakthrough in Photocatalytic Efficiency
- Synthesis and Enhanced Photocatalytic Degradation of Rhodamine B by Ag3PO4/T‑ZnO Whisker Heterostructures
- Sustainable Activated Carbon from Kusha Grass (Desmostachya bipinnata) Enhances Supercapacitor Performance



