Solution‑Processed Nanocrystalline CuAlO₂ Thin‑Film Transistors: Optimized p‑Type Performance for Low‑Cost CMOS Applications
Abstract
Although n‑type metal‑oxide TFTs have dominated the field, high‑performance p‑type devices remain scarce. In this study we report a scalable, solution‑based route to fabricate nanocrystalline CuAlO₂ thin films by spin‑coating, followed by post‑annealing in nitrogen at temperatures up to 1000 °C. Systematic analysis of the films’ microstructure, composition, morphology, and optical properties demonstrates a complete phase conversion from mixed CuO/CuAl₂O₄ to single‑phase CuAlO₂ above 900 °C, accompanied by increased transmittance, band gap, grain size, and surface roughness. Bottom‑gate, top‑contact p‑type TFTs fabricated on SiO₂/Si substrates using these films exhibit a threshold voltage of −1.3 V, a field‑effect mobility of ≈0.1 cm² V⁻¹ s⁻¹, and an on/off ratio of ≈10³. These results mark a significant advance toward cost‑effective complementary oxide electronics.
Background
Metal‑oxide thin‑film transistors (TFTs) are integral to next‑generation displays and flexible electronics, thanks to their superior electrical performance and optical transparency.1–3 However, most progress has focused on n‑type oxides, while p‑type counterparts lag due to intrinsic challenges such as localized O 2p orbitals, high electronegativity, and unintentional donor defects.4–6 Consequently, the development of reliable p‑type oxide semiconductors is essential for all‑oxide p‑n junctions and CMOS logic circuits.
Delafossite oxides, notably CuMO₂ (M = Al, Ga, In) and SrCu₂O₂, have emerged as promising p‑type candidates. CuAlO₂, in particular, offers a wide band gap (~3.5 eV) and a highly dispersive valence band dominated by hybridized O 2p and Cu 3d¹⁰ orbitals, which promotes optical transparency and hole transport.7–9 Despite early demonstrations in 1997,8–9 the scarcity of high‑quality, solution‑processed CuAlO₂ TFTs has limited the field. Magnetron sputtering has produced p‑type devices, yet it demands stringent vacuum conditions and complex processing.10–12 In contrast, solution processing delivers simplicity, low cost, compositional tunability, and atmospheric fabrication. Here, we present a thorough investigation of CuAlO₂ thin‑film formation by spin‑coating, the influence of annealing temperature on film properties, and the performance of bottom‑gate TFTs incorporating these films.
Methods/Experimental
Precursor Preparation and Thin‑Film Fabrication
CuAlO₂ precursors were synthesized by mixing equimolar copper nitrate trihydrate (Cu(NO₃)₂·3H₂O) and aluminum nitrate nonahydrate (Al(NO₃)₃·9H₂O) in ethylene glycol methyl ether at 0.2 M, with acetylacetone added to stabilize the deep‑green solution. The mixture was stirred in an 80 °C water bath until homogenous. SiO₂/Si substrates were cleaned sequentially in acetone, ethanol, and deionized water (5 min each). Films were spin‑coated at 500 rpm for 9 s followed by 5000 rpm for 30 s, then annealed at 350 °C for 20 min. Repeating this cycle four times yielded ~40 nm films, which were subsequently annealed at 700–1000 °C for 2 h in nitrogen and cooled under the same atmosphere.
Fabrication of CuAlO₂ TFTs
Bottom‑gate TFTs were built on 300 nm SiO₂/Si substrates. After CuAlO₂ deposition, 50 nm Au source/drain electrodes were thermally evaporated (0.08 nm s⁻¹) through a shadow mask, defining a channel width of 1000 µm and length of 100 µm. An indium wire served as the back‑gate electrode.
Film and TFT Characterization
X‑ray diffraction (XRD, Cu Kα, λ = 0.154 nm) assessed crystalline phases. Raman spectra were recorded with a 633 nm solid‑state laser (Renishaw‑1000). Surface morphology was examined by SEM (JSM‑5600LV) and AFM (Veeco Dimension Icon). X‑ray photoelectron spectroscopy (XPS, Thermo Scientific Escalab 250 Xi) was performed after 3 nm surface etching. Optical transmittance was measured by UV‑Vis (Varian Cary 5000). Electrical characteristics were obtained with a Keithley 2612B semiconductor parameter analyzer.
Results and Discussion
Figure 1a shows the XRD patterns of films annealed at 700–1000 °C. At 700 °C, only weak CuO peaks appear (35.8° and 38.9°). At 800 °C, new peaks emerge at 31.7° (CuAlO₂) and 37.1° (CuAl₂O₄). Above 900 °C, CuO intensity diminishes and CuAl₂O₄ disappears; the pattern is dominated by CuAlO₂ peaks at 36.8°, 42.5°, 48.5°, 57.5°, and 31.7°. At 1000 °C, single‑phase CuAlO₂ is achieved, reflecting enhanced crystallinity due to higher thermal energy.
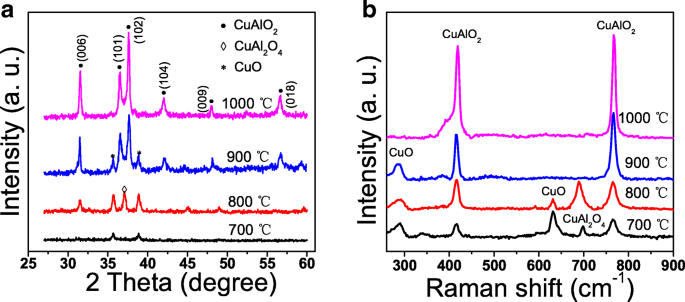
a XRD patterns. b Raman spectra of the CuAlO₂ thin films annealed at different temperatures
Figure 1b displays Raman spectra. The delafossite structure supports two active modes: A₁g (416 cm⁻¹) and E_g (771 cm⁻¹). Both modes are present in all films, confirming CuAlO₂ formation even at 700 °C, where XRD is inconclusive. Additional peaks at 798, 297, and 632 cm⁻¹ correspond to CuAl₂O₄ (F₂g) and CuO (A_g, B_g) vibrations. Their intensities decline with higher annealing temperatures, aligning with XRD results.
XPS analysis (Fig. 2a) reveals Cu 2p₃/₂ peaks at ~932.8 eV (Cu⁺) and ~934.2 eV (Cu²⁺). The Cu⁺ fraction rises from 62.5 % at 700 °C to 78.9 % at 1000 °C, indicating reduced Cu²⁺ and improved phase purity. CuO satellite peaks disappear after high‑temperature annealing, corroborating the phase transition. O 1s spectra (Fig. 2b) show a dominant peak at ~529.8 eV, evidencing well‑bonded oxygen with minimal defect states.
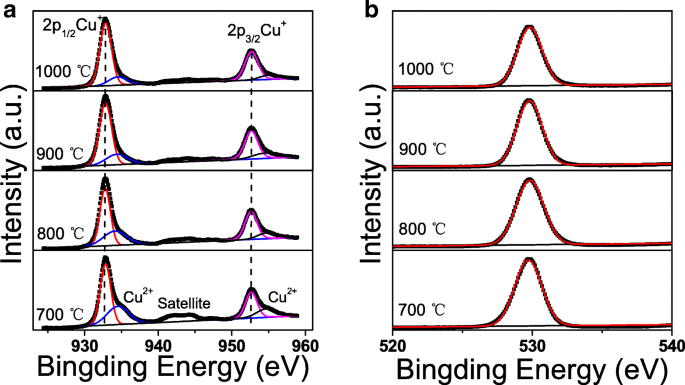
a Cu 2p. b O 1s XPS spectra of the CuAlO₂ thin films annealed at different temperatures
SEM images (Fig. 3a) reveal continuous, crack‑free films with uniform grain growth as annealing temperature increases. AFM (Fig. 3b) shows root‑mean‑square roughness rising from 0.92 nm (700 °C) to 2.96 nm (1000 °C). While larger grains reduce grain‑boundary scattering, increased roughness can introduce interface traps; thus, a balance between these factors influences device performance.

a SEM. b AFM of the CuAlO₂ thin films annealed at different temperatures
Optical transmission (Fig. 4) shows steep absorption edges and strong UV absorption, indicative of high crystallinity. Visible transmittance increases from ~60 % to ~80 % with annealing temperature. Using Tauc plots, the band gap widens from 3.25 eV (700 °C) to 3.80 eV (1000 °C), reflecting improved crystallinity and reduced defect states.
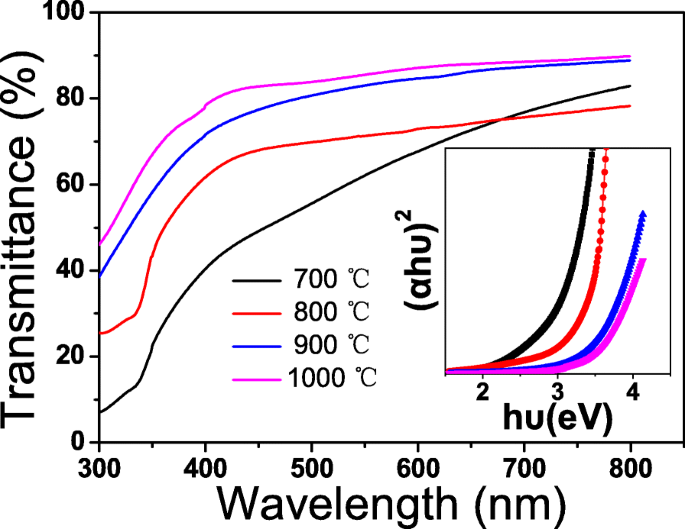
The optical transmission spectra for CuAlO₂ films annealed at different temperatures. The inset shows a plot of (αhν)² vs. hν of the films
Device measurements (Fig. 5) demonstrate clear p‑type behavior. Output curves (Fig. 5b) show increased on‑state current with higher annealing temperatures, attributable to reduced CuAl₂O₄ and enhanced CuAlO₂ crystallinity. Transfer characteristics (Fig. 5c–f) yield on/off ratios around 10³ across all temperatures. The threshold voltage shifts positively with temperature, while field‑effect mobility rises from 0.006 to 0.098 cm² V⁻¹ s⁻¹ between 700 °C and 1000 °C, consistent with improved grain connectivity and reduced interface traps. Subthreshold slopes also decrease, indicating fewer interface states.
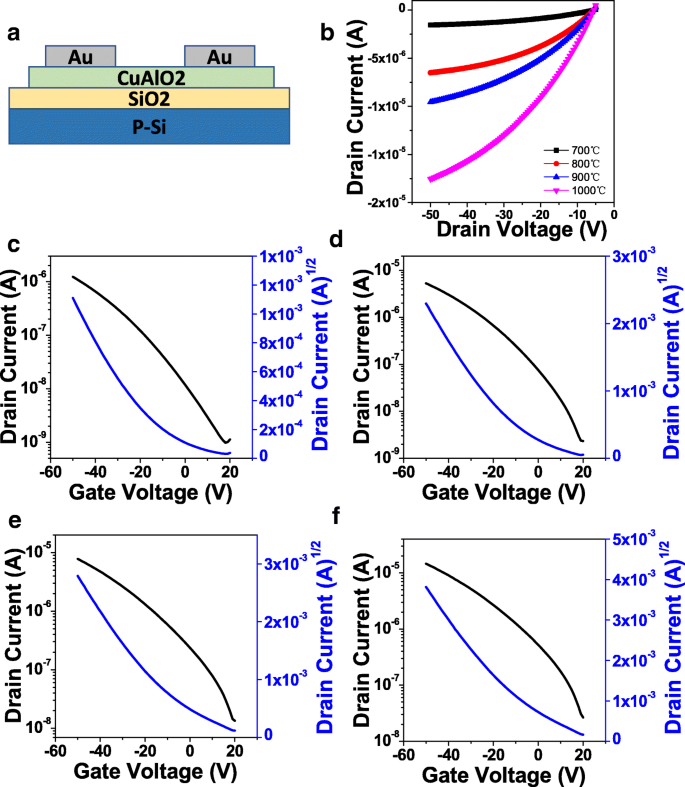
a Schematic diagram of the CuAlO₂ TFTs. b Summarized output curves. c–f Transfer curves of the CuAlO₂ TFTs annealed at 700 °C, 800 °C, 900 °C, and 1000 °C, respectively
Key electrical parameters are summarized in Table 1. The observed mobility values, while lower than some CuCrO₂ counterparts, are notable for a solution‑processed p‑type oxide. Further optimization—such as reducing annealing temperature via UV/ozone photochemical treatments or combustion synthesis—may enhance performance while improving process practicality.
Conclusions
We have demonstrated a scalable solution‑processing route to produce nanocrystalline CuAlO₂ thin films. Annealing above 900 °C converts mixed CuO/CuAl₂O₄ phases into single‑phase CuAlO₂, simultaneously increasing transmittance, band gap, grain size, and surface roughness. Bottom‑gate TFTs fabricated with these films exhibit a threshold voltage of −1.3 V, a mobility of ~0.1 cm² V⁻¹ s⁻¹, and an on/off ratio of ~10³. Compared to vacuum‑based sputtering, our method offers a low‑cost, atmospheric‑processing alternative, advancing the development of complementary oxide logic circuits.
Nanomaterials
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- TiO₂ Nanofluids: Part 1 – Preparation and Key Properties
- Low‑Temperature ALD of In₂O₃ Nanofilms for High‑Performance Thin‑Film Transistors
- Synthesis and Characterization of Monodisperse ZnO Nano‑Clips via a Simple Polyol Process
- Synthesis and Characterization of a Novel Thermoresponsive OEGDA‑MAA Nanogel Polymer
- Dialysis‑Derived Tadpole and Sphere Hemin Nanoparticles: A 308‑Fold Solubility Boost for Iron Bioavailability
- Simple Fabrication and Performance of Polyaniline/CeO₂‑Co‑Decorated TiO₂ Nanotube Arrays for Efficient Photoelectrocatalytic Degradation of TBBPA
- How Ambient Gases Influence Electrical Performance in Solution-Processed C8-BTBT Thin-Film Transistors
- Electrospun PAN/PS Micro‑Nanofibers Loaded with Ammonium Metatungstate and Cobalt(III) Acetylacetonate: Porous Architecture and Enhanced Through‑Pore Distribution
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries



