Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
Abstract
Hemp stems, an abundant and inexpensive biomass waste, offer a promising route to high‑performance anode materials. In this study, we prepared activated carbon (AC) from hemp stems via low‑temperature carbonization followed by ZnCl2 activation at 500–800 °C. The natural fibrous architecture of hemp generates a highly porous carbon with predominantly micropores, complemented by meso‑ and macropores. As a lithium‑ion battery anode, AC delivers a reversible capacity of 495 mAh g–1 after 100 cycles at 0.2 C, surpassing graphite and outperforming unactivated carbon (UAC) due to its optimized pore distribution. This work demonstrates a cost‑effective, scalable approach to producing high‑capacity anodes from renewable waste.
Introduction
Biomass waste, such as hemp stems, remains largely underutilized despite its potential as a functional material. Activated carbons derived from plant residues have been explored for adsorption, hydrogen storage, and supercapacitor electrodes [1–9]. Importantly, carbonaceous anodes for lithium‑ion batteries have emerged as a key application of biomass‑derived carbons [10–17]. For instance, hollow graphene spheres fabricated via oil‑bag emulsion display excellent lithium storage, while mesoporous activated carbon from corn stalk delivers a reversible capacity of 504 mAh g–1 after 100 cycles at 0.2 C [11]. Recent advances include bamboo‑derived carbon fibers achieving 627.1 mAh g–1 over 100 cycles [18]. These successes highlight biomass waste as a fertile source of high‑energy materials. Hemp (Cannabis sativa) is a green, high‑yield crop with expanding cultivation worldwide. Beyond fibers and bio‑fuel, hemp residues have found uses in concrete aggregates [24,25], biocomposites [26], and even ethanol production [27]. Yet, the stems—often discarded as waste—remain largely untapped. Their inherent porosity and fibrous network make them attractive candidates for porous carbon synthesis. Previous work has demonstrated that hemp stems yield activated carbons with large surface areas and excellent adsorption capacities [28–35]. However, their application as lithium‑ion battery anodes has not been reported. This study investigates the electrochemical performance of hemp‑stem‑derived AC, revealing that the natural pore structure and the activation process synergistically enhance lithium storage capability. The resulting material offers a low‑cost, high‑value route to next‑generation battery anodes.
Methods
Preparation of Hemp‑Stem‑Derived Activated Carbon
Raw hemp stems were collected from Heilongjiang Province, cleaned, dried at 60 °C, and ground into powder. The powder was carbonized at 300 °C for 3 h under argon (5 °C min–1) to remove volatiles. Subsequently, it was mixed with ZnCl2 (mass ratio 1:5) and activated in a tube furnace at 500–800 °C for 3 h, then cooled to room temperature. After activation, the product was ground, washed in 2 M HCl for 24 h to remove inorganic residues, and repeatedly rinsed with deionized water until neutral pH. Samples were labeled AC‑λ, where λ denotes the activation temperature. A reference sample, unactivated carbon (UAC), was prepared by carbonizing at 600 °C without ZnCl2.
Materials Characterization
Powder X‑ray diffraction (XRD) was performed on a Siemens D5000 diffractometer using Cu Kα radiation. Raman spectra were recorded on a Renishaw invia spectrometer. Morphology was examined by field‑emission scanning electron microscopy (JEOL JSM‑6700F) and transmission electron microscopy (TEM, JEM‑2100F). Specific surface area and pore size distribution were measured via nitrogen adsorption–desorption (Micromeritics ASAP2420) following BET and BJH analyses.
Electrochemical Measurements
Electrode slurry was prepared by mixing the active material, acetylene black, and PVDF in an 8:1:1 mass ratio with N‑methyl‑2‑pyrrolidone (NMP). After stirring, the slurry was coated onto copper foil and dried at 120 °C under vacuum for 12 h. Coin cells (CR2032) were assembled in an argon glove box (<0.1 ppm H2O and O2) using a lithium metal counter electrode, polypropylene separator, and an electrolyte of EC:DMC:EMC (1:1:1 v/v) containing 1 M LiPF6. Cycling tests were conducted with a LAND battery system from 0.02 to 3 V. Cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) were performed on an electrochemical workstation.
Results and Discussion
Figure 1 illustrates the transformation from raw hemp stems to the final AC. SEM images (Fig. 1c–d) reveal that UAC consists of a largely amorphous matrix with limited porosity, whereas AC‑600 displays a layered, slit‑like architecture that increases surface area and active sites. TEM and HRTEM (Fig. 2c–d) confirm that AC‑600 possesses a higher density of micropores compared to UAC, aligning with its superior electrochemical performance.

a Hemp stem powder. b Carbide of hemp stem. c SEM image of UAC. d SEM image of AC

a TEM pattern of UAC. b TEM pattern of AC. c HRTEM pattern of UAC. d HRTEM pattern of AC
XRD patterns (Fig. 3a) show a broad peak at 22°, characteristic of the (002) reflection in disordered graphite, and a weak 44° peak attributed to sp2 honeycomb domains, confirming the amorphous nature of both samples. Raman spectra (Fig. 3b) reveal ID/IG ratios of 1.15 (UAC) and 1.17 (AC), indicating high defect density and numerous edge sites that facilitate lithium insertion.
a X‑ray diffraction patterns. b Raman spectra of UAC and AC
Nitrogen sorption analysis (Fig. 4) confirms a type I isotherm with a pronounced H4 hysteresis loop, indicative of abundant micropores and slit‑like mesopores. AC exhibits a BET surface area of 589.54 m2 g–1, a pore volume of 0.332 cm3 g–1, and an average pore diameter of 2.25 nm. This hierarchical porosity enhances ion transport and charge storage.
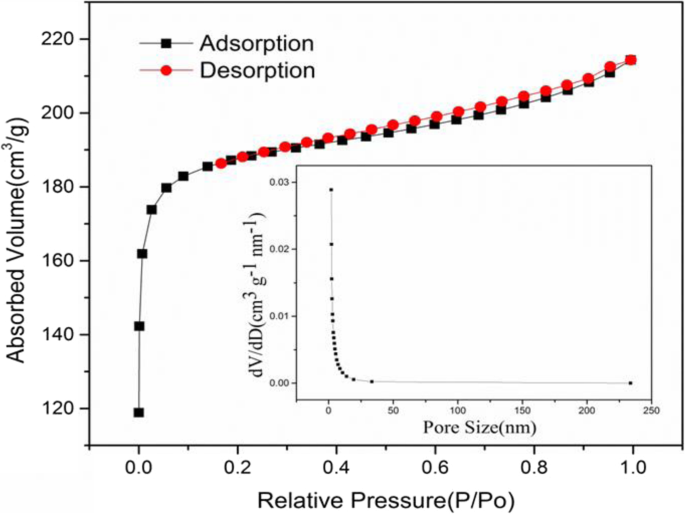
Isothermal adsorption‑desorption curve of AC (illustration is pore size distribution)
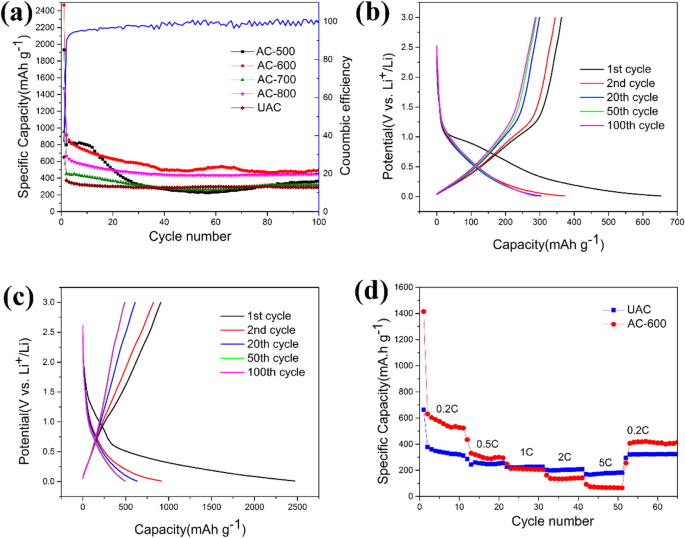
Electrochemical performance is summarized in Figure 5. At 0.2 C, AC‑600 delivers a reversible capacity of 495.4 mAh g–1 after 100 cycles, surpassing graphite’s theoretical 372 mAh g–1 and outperforming UAC (≈313 mAh g–1). The first discharge capacity is 2469.7 mAh g–1 due to initial SEI formation; Coulombic efficiency stabilizes near 100 % after the first cycle. Rate capability tests (Fig. 5d) show AC‑600 retains 65.2 mAh g–1 at 5 C, and recovers to 416.3 mAh g–1 when returning to 0.2 C, highlighting its robust kinetics.

a Cycle performance curves of different materials. b, c Charge‑discharge voltage curves of UAC and AC‑600. d Rate performance of UAC and AC‑600
Post‑cycling TEM (Fig. 6) reveals partial surface disruption in AC‑600, exposing internal pores and suggesting that over‑activation may compromise surface integrity, leading to minor capacity fade. EIS spectra (Fig. 7a–b) show a reduced charge‑transfer resistance for AC‑600 relative to UAC, reflecting facilitated lithium diffusion through its hierarchical pores. CV curves (Fig. 7c–d) indicate irreversible SEI formation in the first cycle (peaks at 0.7 V and 1.35 V) followed by stable intercalation/deintercalation at ~0.25 V. Subsequent cycles exhibit overlapping scans, confirming electrode stability.

TEM pattern of spent electrode materials after cycling
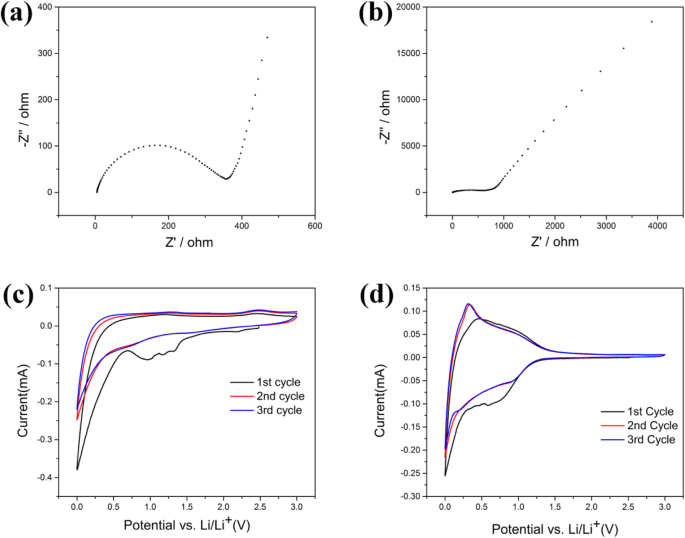
a Impedance spectrums of AC‑600. b Impedance spectrums of UAC. c Cyclic voltammogram profiles of AC‑600. d Cyclic voltammogram profiles of UAC
Conclusions
We have demonstrated that hemp‑stem‑derived activated carbon, produced via low‑temperature carbonization and ZnCl2 activation, constitutes a viable anode for lithium‑ion batteries. The resulting AC possesses a high BET surface area (589.54 m2 g–1) and a hierarchical pore network dominated by micropores, which underpin a reversible capacity of 495 mAh g–1 after 100 cycles at 0.2 C. Compared to unactivated carbon, the activation step markedly improves capacity, rate performance, and cycling stability. Despite minor surface damage from aggressive activation, the method offers a scalable, low‑cost route to convert hemp waste into high‑value battery materials, advancing both renewable resource utilization and energy storage technology.
Availability of Data and Materials
The conclusions drawn in this manuscript are based on the data presented herein.
Abbreviations
- AC:
Activated carbon
- CE:
Coulomb efficiency
- CV:
Cyclic voltammetry
- DMC:
Dimethyl carbonate
- EC:
Ethylene carbonate
- EMC:
Ethyl methyl carbonate
- SEI:
Solid electrolyte interface
- UAC:
Unactivated carbon
Nanomaterials
- Enhanced Lithium‑Ion Battery Anodes: Polypyrrole‑Coated MnO₂ Core–Shell Micromaterials Deliver Superior Cyclic Stability
- High‑Performance Nanocrystalline Fe₂O₃ Thin‑Film Anodes via Pulsed Laser Deposition for Lithium‑Ion Batteries
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- Pomegranate‑Shaped Fe₂O₃/C Core‑Shell Nanocomposites: A One‑Step Hydrothermal Route for High‑Performance Li‑Ion Battery Anodes
- Dialysis‑Derived Tadpole and Sphere Hemin Nanoparticles: A 308‑Fold Solubility Boost for Iron Bioavailability
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- ZnSe/N‑Doped Carbon Composites Derived from ZIF‑8 Deliver High Capacity and Long‑Term Cycling for Lithium‑Ion Batteries
- High‑Performance CoSe/NC Anodes from ZIF‑67 Precursors for Lithium‑Ion Batteries
- Enhancing Lithium‑Ion Battery Anodes: Deep Cryogenic Activation of Hemp‑Stem Derived Carbon
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries



