Mesoporous Silicon Microspheres via In Situ Magnesiothermic Reduction: A High‑Performance Anode for Sodium‑Ion Batteries
Abstract
Silicon is a compelling anode material for sodium‑ion batteries (NIBs) because of its high theoretical capacity (954 mAh g⁻¹). Here we show that mesoporous silicon microspheres (MSMs) with uniformly distributed pores (1–10 nm) can be fabricated through an in‑situ magnesiothermic reduction of SiO₂. The MSMs calcined at 650 °C deliver a reversible capacity of 390 mAh g⁻¹ at 100 mAg⁻¹ after 100 cycles and retain 160 mAh g⁻¹ at 1000 mAg⁻¹, with a negligible capacity fade (0.08 mAh g⁻¹ cycle⁻¹). The outstanding performance is attributed to the ultrafine particle size and mesoporous architecture, which buffer volume changes and enhance Na⁺ transport.
Background
Lithium‑ion batteries (LIBs) dominate portable electronics and electric vehicles due to their high energy density, but their reliance on scarce lithium limits large‑scale deployment. Sodium‑ion batteries (NIBs) offer a cost‑effective alternative, thanks to sodium’s abundance and low price. However, Na⁺ (0.98 Å) is considerably larger than Li⁺ (0.69 Å), rendering traditional graphite anodes unsuitable for Na⁺ storage. The Na–Si phase diagram shows that the most Na‑rich binary compound is NaSi, providing a theoretical capacity of 954 mAh g⁻¹, which makes silicon an attractive anode candidate for NIBs. Numerous studies have explored silicon in various forms for NIBs. Mulder reported a capacity of ~300 mAh g⁻¹ after 100 cycles using Si nanoparticles, while Mukhopadhyay achieved 390 mAh g⁻¹ after 200 cycles with crystalline core/amorphous shell Si nanowires. Amorphous, nanoscale silicon consistently delivers superior Na⁺ insertion/extraction kinetics due to its higher Na‑ion diffusivity and better accommodation of volume expansion. Despite these advances, scalable synthesis remains a hurdle. Conventional methods are often complex or expensive. In contrast, in‑situ magnesiothermic reduction of SiO₂ offers a simple, scalable route to mesoporous silicon microspheres, which can simultaneously provide high capacity and structural stability.
Methods / Experimental
SiO₂ microspheres were prepared by a modified Stöber process: 20 mL tetraethyl orthosilicate was added to 100 mL deionized water, followed by 20 mL NH₃·H₂O and 80 mL 2‑propanol. The mixture was stirred at room temperature for 2 h, then the colloidal spheres were collected by centrifugation, washed, and dried at 100 °C. For the magnesiothermic reduction, 560 mg of SiO₂ microspheres and 600 mg of magnesium powder were placed in separate stainless‑steel containers and sealed in a stainless‑steel oven. The mixture was heated to 650 °C for 2 h under argon, initiating the reaction 2 Mg + SiO₂ → Si + 2 MgO. The resulting brown‑yellow powder was treated with 1 M HCl (200 mL) for 12 h to dissolve MgO, filtered, and dried under vacuum at 80 °C. Electrochemical testing employed 2032‑type coin cells. The anode slurry (10 wt % PVDF, 20 wt % acetylene black, 70 wt % active material) was cast onto copper foil and dried to 2 mg cm⁻². The cells were assembled in an Ar glovebox using Celgard 2250 separators, a 1 M NaClO₄/EC:DEC (1:1) electrolyte, Na foil counter electrode, and MSMs working electrode. Galvanostatic charge–discharge measurements were performed from 0.01 to 2.5 V using a LAND test system.
Results and Discussion
The XRD patterns (Fig. 1) of the MgO–Si nanocomposite, MSMs, and commercial Si microspheres show only the Si (111), (220), (311), (400), and (331) reflections (JCPDS 772107). No impurity peaks are present, confirming the complete removal of MgO after HCl treatment.
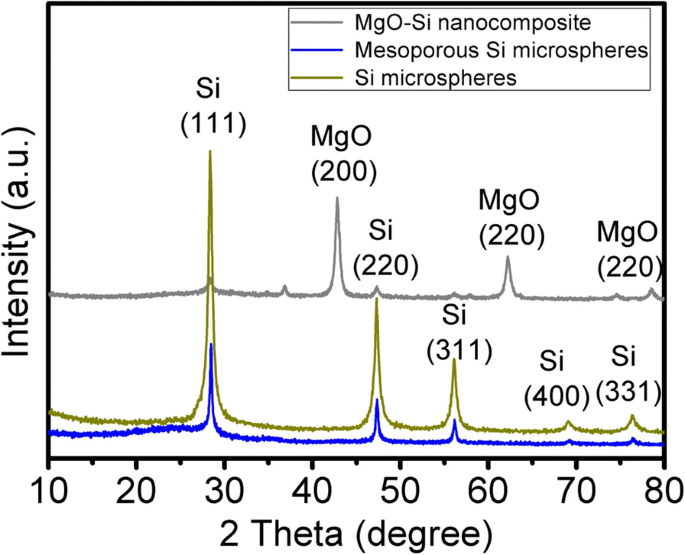
XRD patterns of the samples
SEM and TEM images (Fig. 2) reveal that the MSMs possess abundant mesopores ranging from 1 to 10 nm. The BET analysis shows a surface area of 200 m² g⁻¹ and a pore volume of 0.25 cm³ g⁻¹. These mesopores act as buffer zones, accommodating silicon volume changes during sodiation/desodiation and maintaining structural integrity.
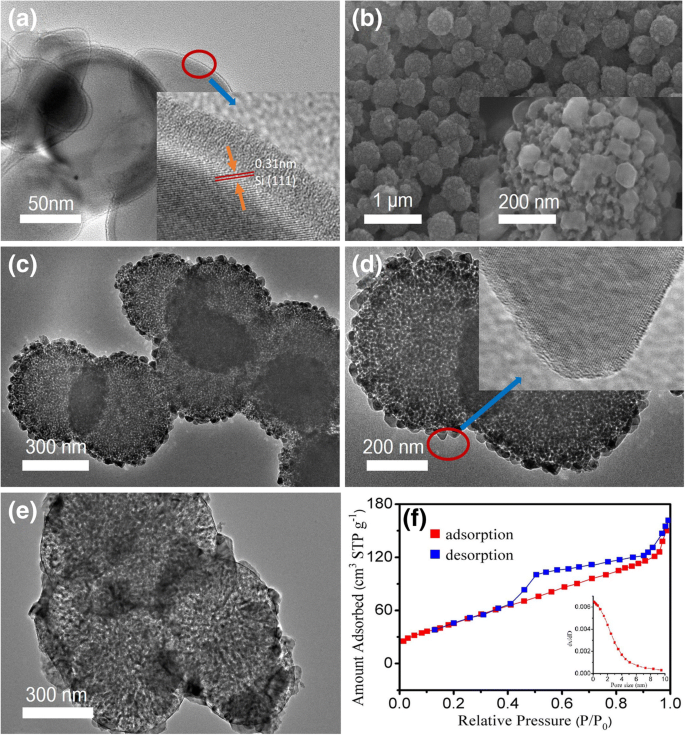
SEM (a) and inset TEM (a) images of silicon microspheres. SEM (b) and TEM (c‑d) images of MSMs. TEM (e) 100 mAg⁻¹ after 100 cycles. The adsorption–desorption curve (f) of MSMs, inset: particle size distribution.
Cyclic voltammetry (Fig. 3a) shows a cathodic peak at 0.04 V attributed to Na⁺ insertion into crystalline Si, with an anodic peak at 0.08 V during de‑sodiation. Amorphous Si exhibits broader Na⁺ absorption between 0 and 0.8 V. Increasing scan rate shifts peaks to lower potentials, indicating overpotential effects.
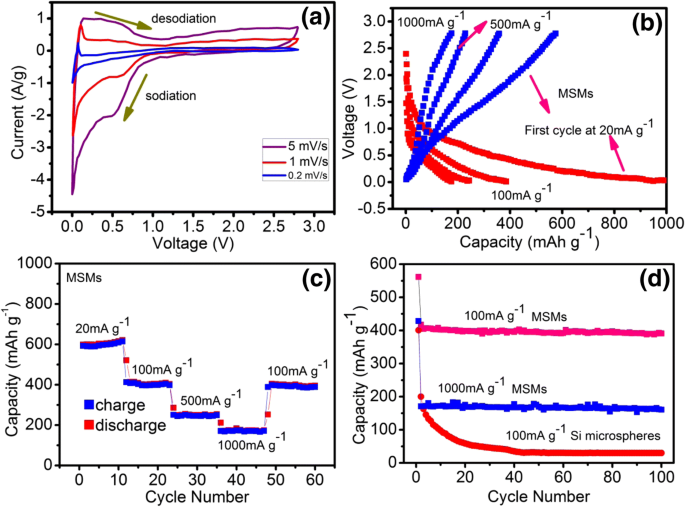
a Cyclic voltammetry of MSMs at different scan rates. b Representative charge–discharge curves at various current densities. c Capacity retention at different current densities. d Discharge capacity retention of MSMs versus Si microspheres at 1000 mAg⁻¹ and 100 mAg⁻¹.
The MSMs electrode retains ~400 mAh g⁻¹ at 100 mAg⁻¹ and 160 mAh g⁻¹ at 1000 mAg⁻¹, with only 0.08 mAh g⁻¹ cycle⁻¹ decay over 100 cycles. In contrast, commercial Si microspheres lose most capacity after 100 cycles. The superior cycling stability of MSMs arises from their mesoporous structure, which mitigates pulverization and preserves electrical contact.
The synthesis mechanism (Fig. 4) involves Mg vapor reacting with SiO₂ at 650 °C to form a MgO–Si nanocomposite. Subsequent HCl etching removes MgO, leaving behind 3‑D MSMs. The mesopores serve as internal reservoirs, absorbing silicon volume changes and facilitating Na⁺ transport, thereby enhancing electrochemical performance.
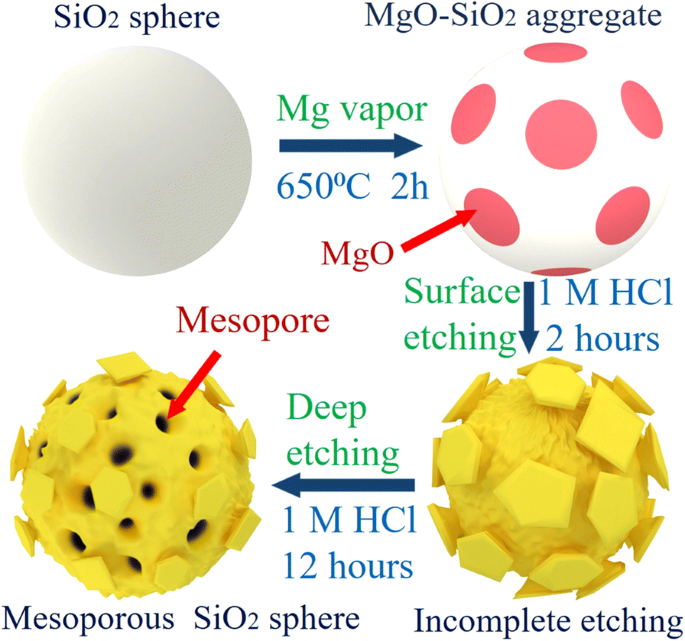
Schematic illustration of the MSMs synthesis process.
Conclusions
We have demonstrated that in‑situ magnesiothermic reduction of SiO₂ yields 3‑D mesoporous silicon microspheres with excellent Na‑ion storage performance at room temperature. The uniform mesoporous architecture confers high reversible capacity, robust rate capability, and superior cycling stability, positioning MSMs as a promising anode material for next‑generation sodium‑ion batteries.
Abbreviations
- 3D
Three dimensions
- BJH
Barrett–Joyner–Halenda
- CV
Cyclic voltammetry
- HCl
Hydrochloric acid
- LIBs
Lithium‑ion batteries
- MgO
Magnesium oxide
- MSMs
Mesoporous silicon microspheres
- NaSi
Sodium silicide
- NIBs
Sodium‑ion batteries
- SEM
Scanning electron microscopy
- Si
Silicon
- TEM
Transmission electron microscopy
- XRD
X‑ray diffraction
Nanomaterials
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Highly Efficient Magnetic Carbon Microspheres for Reusable Sulfonamide Removal from Water
- Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
- High‑Performance Na4Mn9O18/Carbon Nanotube Cathodes for Aqueous Sodium‑Ion Batteries
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- 3D Honeycomb‑Like SnS₂ Quantum Dot / rGO Composites: A High‑Performance Anode for Lithium and Sodium‑Ion Batteries
- Penta‑Siligraphene: A Thermally Stable, High‑Capacity Anode for Li‑Ion Batteries – A First‑Principles Study
- Co9S8 Nanostructures with N,S-Doped Carbon Coating: High-Performance Sodium-Ion Battery Anodes
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries



