CNT@Li₂FeSiO₄@C Core‑Shell Heterostructure: A Robust Cathode for High‑Performance Lithium‑Ion Batteries
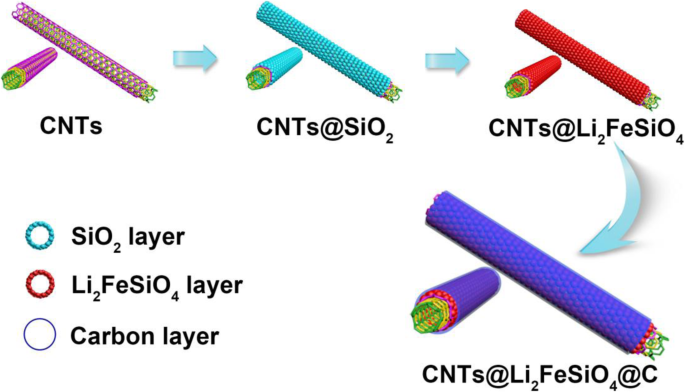
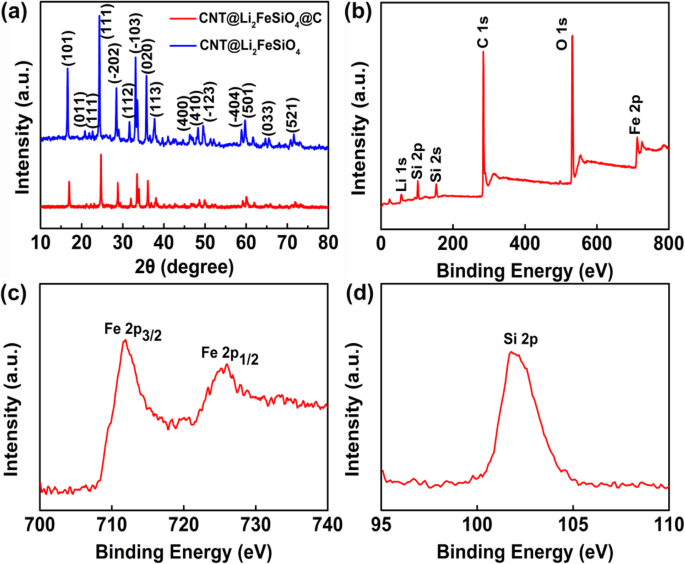
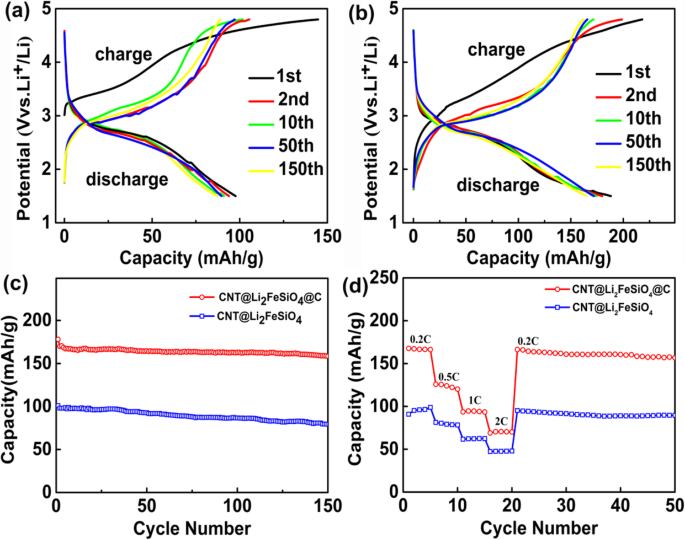
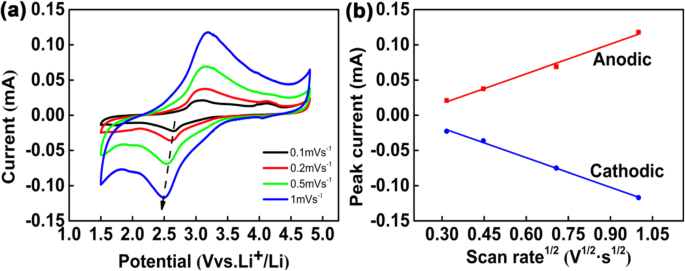
Designing nanostructured cathodes is essential for overcoming the intrinsic limitations of Li₂FeSiO₄. In this study, we present a novel CNT@Li₂FeSiO₄@C heterostructure that combines a conductive carbon nanotube core, a 19‑nm Li₂FeSiO₄ shell, and a 2‑nm amorphous carbon coating. This architecture accelerates lithium‑ion diffusion, enhances electronic conductivity, and buffers volumetric changes during cycling. As a result, the material delivers superior reversible capacity and exceptional cycle stability, outperforming conventional Li₂FeSiO₄ cathodes. Lithium‑ion batteries (LIBs) dominate portable electronics and electric vehicles due to their high energy density, long cycle life, and low self‑discharge. The pursuit of next‑generation LIBs with higher energy density, faster rate capability, and longer cycle life hinges on the development of advanced cathode materials. Li₂FeSiO₄ has emerged as a promising candidate because of its low cost, chemical stability, and environmental friendliness. Theoretically, it can host two lithium ions per formula unit, yielding a capacity of 332 mAh g⁻¹. However, practical performance is limited by its extremely low electronic conductivity (~6 × 10⁻¹⁴ S cm⁻¹) and sluggish lithium‑ion diffusivity (~10⁻¹⁴ cm² s⁻¹). Strategies such as nanosizing and carbon coating have been explored, but challenges remain: synthesis often introduces secondary phases (e.g., Fe₃O₄, Li₂SiO₃) that degrade capacity, and high‑temperature treatments can damage the nanostructure. Here, we report a core‑shell heterostructure CNT@Li₂FeSiO₄@C that avoids impurity phases and preserves structural integrity by fine‑tuning reagent ratios and reaction conditions. The embedded CNT network boosts electronic pathways, the ultra‑thin Li₂FeSiO₄ shell (20–25 nm) facilitates rapid Li⁺ diffusion, and the outer carbon layer further improves conductivity and protects the inner framework. Collectively, these features translate into markedly enhanced specific capacity, cycle life, and rate performance. The CNT@Li₂FeSiO₄@C was fabricated through a multi‑step, layer‑by‑layer approach. First, a coaxial CNT@SiO₂ scaffold was prepared by a sol‑gel coating: 8 mg CNTs were dispersed in 80 mL ethanol and 30 mL deionized water, sonicated for 30 min, then mixed with 2 mL aqueous ammonia (25–28 wt %) and 0.16 g CTAB under continuous sonication for 20 min. A TEOS/ethanol solution (0.45 mL TEOS in 40 mL ethanol) was then added dropwise with stirring for 40 min, followed by 10 h stirring. The product was collected by centrifugation and washed with water and ethanol. Next, the CNT@SiO₂ was impregnated with Li and Fe precursors: 0.334 g LiAc·2H₂O and 0.734 g Fe(NO₃)₃·9H₂O were dissolved in 30 mL ethanol, then the CNT@SiO₂ was sonicated for 40 min and stirred for 20 min. After drying at 80 °C for 12 h, the powder was calcined at 400 °C for 2 h and then at 650 °C for 10 h under Ar, yielding CNT@Li₂FeSiO₄. Finally, a thin carbon coating was applied using glucose as the carbon source: 0.2 g CNT@Li₂FeSiO₄ was dispersed in 40 mL ethanol, sonicated for 40 min, then 0.1 g glucose was added. The mixture was dried at 80 °C for 6 h and calcined at 400 °C for 4 h in Ar, producing the final CNT@Li₂FeSiO₄@C. X‑ray diffraction (XRD, D2 PHASER Bruker) characterized crystal phases (10°–80° 2θ, Cu‑Kα). X‑ray photoelectron spectroscopy (XPS, K‑ALPHA 0.5EV) confirmed elemental composition and valence states. Scanning electron microscopy (SEM, FESEM, S‑4800) and transmission electron microscopy (TEM, Tecnai G2 F 20) revealed morphology and core‑shell structure. Energy‑dispersive X‑ray spectroscopy (EDX) mapped elemental distribution. Thermogravimetric analysis (TGA, STD Q600 TA) quantified carbon content (10 °C min⁻¹, RT–800 °C). Electrodes were cast from a slurry of 10 wt % PVDF, 20 wt % acetylene black, and 70 wt % active material in NMP, coated onto Al foil, and dried at 80 °C under vacuum for >12 h. The active‑material loading was ~1.5 mg cm⁻². Coin cells (CR‑2032) were assembled in an Ar glove box with Li metal anode and 1 M LiPF₆ in EC/DEC. Galvanostatic charge–discharge tests were performed on a Neware system between 1.5 and 4.8 V. Cyclic voltammetry (CV) used a VMP3 workstation (France, Bio‑logic) at scan rates from 0.1 to 1.0 mV s⁻¹. The synthesis pathway of CNT@Li₂FeSiO₄@C is illustrated in Figure 1. Precise control of CNT and TEOS amounts ensured a uniform 19‑nm Li₂FeSiO₄ shell. Calcination at 650 °C in Ar produced crystalline CNT@Li₂FeSiO₄, followed by a mild 400 °C carbon coating to yield the core‑shell heterostructure. Schematic diagram of the synthesis of the core‑shell heterostructure CNT@Li₂FeSiO₄@C Figure 2a shows XRD patterns confirming the monoclinic Li₂FeSiO₄ structure (space group p2₁/n) with no detectable impurity peaks. XPS spectra (Fig. 2b–d) reveal Fe²⁺ states (Fe 2p₃/₂ at 710.3 eV, Fe 2p₁/₂ at 723.8 eV) and Si⁴⁺ (Si 2p at 101.8 eV), confirming the formation of orthosilicate lattices. a XRD of CNT@Li₂FeSiO₄ and CNT@Li₂FeSiO₄@C, b XPS full spectra, c Fe 2p, d Si 2p SEM images (Fig. 3a–d) confirm the one‑dimensional CNT‑based morphology without free Li₂FeSiO₄ particles. TGA indicates carbon contents of 16.93 % (CNT@Li₂FeSiO₄) and 22.69 % (CNT@Li₂FeSiO₄@C). TEM and HR‑TEM (Fig. 3e–k) clearly show the core‑shell architecture: a 40‑nm CNT core, a 16–22‑nm Li₂FeSiO₄ shell, and a ~2‑nm amorphous carbon coating. The lattice fringe (0.27 nm) matches the (−103) plane of orthorhombic Li₂FeSiO₄. EDX mappings (Fig. 3l–o) confirm uniform Fe, Si, and O distribution. a, b SEM images of CNT@Li₂FeSiO₄, c, d SEM images of CNT@Li₂FeSiO₄@C; e, f TEM images of CNT@Li₂FeSiO₄; h, i TEM images of CNT@Li₂FeSiO₄@C; j, k HRTEM of CNT@Li₂FeSiO₄ and CNT@Li₂FeSiO₄@C; l–o EDX elemental mappings of Fe, Si, and O Electrochemical performance (Fig. 4) demonstrates the superiority of CNT@Li₂FeSiO₄@C. At 0.2 C, the initial discharge capacity reaches 178 mAh g⁻¹, with 89.3 % retention after 150 cycles. In contrast, CNT@Li₂FeSiO₄ delivers 100.8 mAh g⁻¹ initially and 78.2 % after 150 cycles. Rate capability tests (Fig. 4d) show capacities of 167.7, 125.8, 94.6, and 70.6 mAh g⁻¹ at 0.2, 0.5, 1, and 2 C, respectively, outperforming literature reports. a Charge/discharge curves at 0.2 C for CNT@Li₂FeSiO₄, b for CNT@Li₂FeSiO₄@C, c cycling performance, d rate performance CV analysis (Fig. 5) reveals a single Fe²⁺/Fe³⁺ redox pair at 3.1 V (anodic) and 2.7 V (cathodic), indicating one Li⁺ per formula unit during cycling. The peak current scales linearly with the square root of scan rate, yielding Li⁺ diffusion coefficients of 7.32 × 10⁻¹¹ cm² s⁻¹ (anodic) and 6.4 × 10⁻¹³ cm² s⁻¹ (cathodic), superior to previous materials. EIS (Figure S4) shows a charge‑transfer resistance of 76.5 Ω for CNT@Li₂FeSiO₄@C versus 193.4 Ω for CNT@Li₂FeSiO₄, confirming enhanced electron transport. Kinetic analysis: a CV profiles at various scan rates, b peak current vs. √scan rate Collectively, the CNT@Li₂FeSiO₄@C architecture synergistically enhances conductivity, Li⁺ diffusion, and structural stability, resulting in high capacity, robust cycling, and superior rate performance. We have successfully fabricated a CNT@Li₂FeSiO₄@C core‑shell heterostructure via a controlled, layer‑by‑layer strategy. The CNT core provides continuous electronic pathways; the ultrathin Li₂FeSiO₄ shell ensures rapid Li⁺ transport; and the outer carbon coating protects the framework while further boosting conductivity. This integrated design delivers a cathode with high specific capacity, excellent cycle life, and remarkable rate capability, positioning CNT@Li₂FeSiO₄@C as a promising candidate for next‑generation lithium‑ion batteries. All data and materials are fully available without restriction. Carbon nano‑tube Cetyltrimethylammonium bromide Cyclic voltammetry Energy‑dispersive X‑ray spectroscopy Electrochemical impedance spectroscopy High‑resolution transmission electron microscopy Lithium‑ion batteries N-methyl‑2‑pyrrolidone Polyvinylidene fluoride Solid electrolyte interface Scanning electron microscopy Transmission electron microscopy Tetraethoxysilane Thermogravimetric analyzer X‑ray photoelectron spectroscopy X‑ray diffractionAbstract
Introduction
Methods
Materials and Synthesis
Materials Characterization
Electrochemical Measurements
Results and Discussion

Conclusions
Availability of Data and Materials
Abbreviations
Nanomaterials
- Arkema Unveils 100% Renewable PVDF Grades for Lithium‑Ion Batteries
- LiNi0.5Mn1.5O4 Cathodes with Cr3+ and F− Composite Doping: Synthesis, Stability, and Electrochemical Performance
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- High‑Performance Nanocrystalline Fe₂O₃ Thin‑Film Anodes via Pulsed Laser Deposition for Lithium‑Ion Batteries
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Penta‑Siligraphene: A Thermally Stable, High‑Capacity Anode for Li‑Ion Batteries – A First‑Principles Study
- Molecular‑Sieve‑Enhanced Separator Boosts Lithium‑Ion Battery Performance & Safety



