Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
Abstract
In lithium‑ion battery (LIB) research, the binder is a critical yet often overlooked component that can decisively influence anode performance. We systematically compared five binders—polyvinylidene fluoride (PVDF) variants (HSV900, 301F, Solvay5130), a water‑soluble blend of styrene‑butadiene rubber and sodium carboxymethyl cellulose (SBR+CMC), and polyacrylonitrile (LA133)—in copper‑oxide (CuO) anodes. Electrochemical testing revealed that aqueous binders, particularly SBR+CMC, substantially improved bonding to the copper foil, cycle stability, and rate capability. SBR+CMC delivered an 87 % capacity retention after 50 cycles versus <30 % for PVDF. These results underscore the suitability of SBR+CMC for transition‑metal‑oxide anodes in LIBs, offering performance gains while eliminating hazardous organic solvents.
Background
Lithium‑ion batteries are ubiquitous in portable electronics, aerospace, and electric transportation, owing to their high specific energy, long cycle life, and compact form factor [1‑5]. Conventional graphite anodes, while inexpensive and stable, are limited by a theoretical capacity of 372 mAh g⁻¹, prompting the exploration of high‑capacity alternatives such as 3d transition‑metal oxides (CuO, Fe₂O₃, Co₃O₄, NiO) [6‑10]. However, these materials suffer from rapid capacity fade driven by mechanical pulverization and SEI instability [8‑10]. Binder chemistry has emerged as a key lever for mitigating these issues. In 2014, our group demonstrated that CuO electrodes with PVDF 301F delivered robust cycling; yet two years later, the same formulation collapsed to <100 mAh g⁻¹ after 50 cycles, highlighting binder sensitivity [11]. Recent studies confirm the importance of binders: SBR+CMC has shown superior cycling and rate performance in TiO₂, ZnFe₂O₄, and Si anodes [12‑16].
PVDF remains the industry standard due to its electrochemical and thermal stability, yet it requires toxic, volatile solvents such as NMP, DMAc, and DMF, which pose environmental and safety challenges [17‑21]. Consequently, the field is shifting toward aqueous binders—CMC, SBR, LA133, PAA, PVA, PEG, PAI—whose solubility in water eliminates hazardous solvents while providing good adhesion and flexibility [22‑31]. Among these, the SBR+CMC combination offers high flexibility, strong adhesion, and excellent cycle stability, making it a prime candidate for metal‑oxide anodes.
Synopsis of chemical structures of polymers introduced in this work
In this study, we systematically examined the performance of CuO anodes prepared with five binders—PVDF HSV900, PVDF 301F, PVDF Solvay5130, SBR+CMC, and LA133—using identical electrode formulations (except for binder). The electrochemical metrics (galvanostatic cycling, cyclic voltammetry, rate capability, EIS) collectively demonstrate the superiority of SBR+CMC.
Experimental
Preparation of Anode Electrode
CuO nanoparticles were synthesized via a chemical reduction route previously reported by our group [11]. For PVDF binders, a slurry of 60 wt % CuO, 10 wt % acetylene black, and 30 wt % PVDF (dissolved in NMP) was cast on copper foil and dried at 80 °C for 5 h. For SBR+CMC, a slurry of 80 wt % CuO, 10 wt % acetylene black, 5 wt % SBR, and 5 wt % CMC (1 % aqueous solution, viscosity >1900 mPa s) was cast and dried at 50 °C for 4 h. For LA133, the slurry composition was 80 wt % CuO, 10 wt % acetylene black, 10 wt % LA133 (water‑soluble) and dried similarly. The active‑material ratio was tuned for each binder to achieve optimal performance.
Cell Assembly and Electrochemical Studies
CR2025 coin cells were assembled in an argon glove box (<1 ppm H₂O/O₂). Metallic lithium served as both reference and counter electrode, with Celgard 2320 as separator. The electrolyte was 1 M LiPF₆ in EC:DMC (1:1 v/v). Galvanostatic charge–discharge tests were conducted on a LAND CT2001A at 0.2 C (0.01–3.00 V vs. Li⁺/Li). Cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) were performed on a CHI604D workstation (frequency 0.01–100 kHz, 5 mV AC). All measurements followed the protocols outlined in the Supporting Information.
Results and Discussion
Galvanostatic Cycling Performance
PVDF Binder
Figure 2 presents the charge–discharge curves for CuO electrodes prepared with PVDF HSV900, 301F, and Solvay5130 at 0.2 C. While PVDF Solvay5130 offered the highest initial capacity (~870 mAh g⁻¹), capacity retention dropped below 30 % after 50 cycles, with the 301F binder yielding <100 mAh g⁻¹. The poor performance is attributed to the low adhesion strength of PVDF in NMP, leading to electrode delamination and excessive SEI growth [33‑35]. Even varying the PVDF molecular weight could not overcome these limitations.
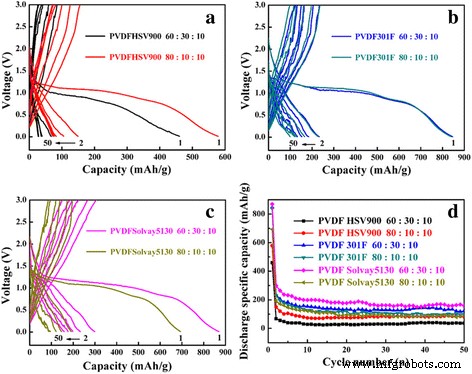
Charge‑discharge curves of CuO with different PVDF binders (a–c) and cycling performance (d). (a) PVDF HSV900, (b) PVDF 301F, (c) PVDF Solvay5130 at 0.2 C.
SBR+CMC Binder
Using the SBR+CMC blend dramatically improved both capacity and stability. At the optimal 80:10:10 (CuO:acetylene black:SBR+CMC) ratio, the electrode delivered 461.3 mAh g⁻¹ after 50 cycles with 86.9 % retention (Figure 3e). The enhanced adhesion is attributed to the elastomeric SBR providing flexible, spot‑type bonding, while CMC supplies carboxylate groups that form strong interactions with CuO and the copper foil [14, 32]. This binder configuration has also been successful in ZnFe₂O₄ and Si anodes, achieving capacities >1000 mAh g⁻¹ and exceptional cycle life [36].
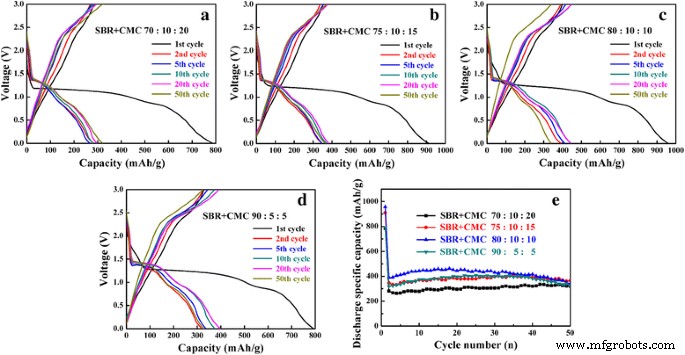
Charge‑discharge curves with SBR+CMC binder at different CuO ratios (a–d) and cycling performance (e). (a) 70:10:20, (b) 75:10:15, (c) 80:10:10, (d) 90:5:5.
LA133 Binder
LA133, a polyacrylonitrile derivative, also yielded superior results. The 80:10:10 ratio achieved a 99 % retention and 450.2 mAh g⁻¹ after 50 cycles (Figure 4g). LA133 can be applied to both anodes and cathodes, whereas SBR+CMC is best suited for anodes due to its oxidation sensitivity at high potentials. Nevertheless, both binders outperform PVDF in terms of adhesion and electrochemical stability.
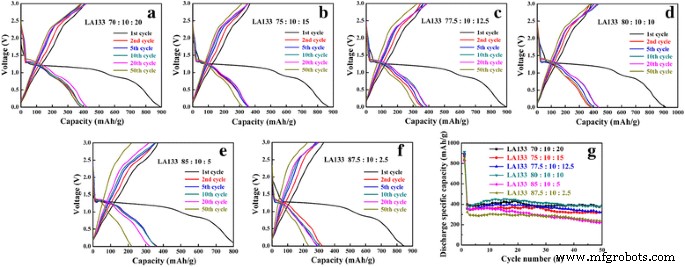
Charge‑discharge curves with LA133 binder at different CuO ratios (a–f) and cycling performance (g). (a) 70:10:20, (b) 75:10:15, (c) 77.5:10:12.5, (d) 80:10:10, (e) 85:10:5, (f) 87.5:10:2.5.
Conclusions of Binders
Figure 5 summarizes the comparative performance: both SBR+CMC and LA133 deliver markedly higher capacities and retention than any PVDF variant. PVDF’s weak adhesion leads to rapid delamination and high SEI resistance, while the aqueous binders maintain robust contact and lower charge‑transfer resistance. These findings are consistent with recent reports on MnCo₂O₄ and other oxide anodes, where CMC+SBR achieved >1600 mAh g⁻¹ over 700 cycles [37].
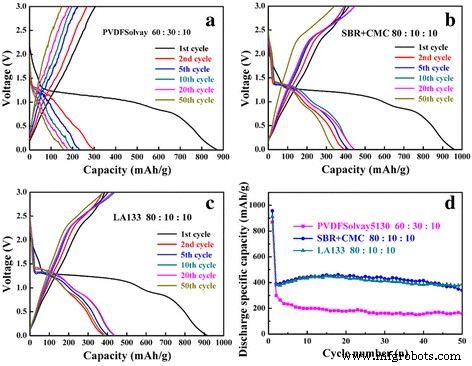
Charge‑discharge curves of CuO with different binders (a–c) and cycling performance (d). (a) PVDF, (b) SBR+CMC, (c) LA133.
Morphological and Structural Characterization
Optical imaging (Figure 6) shows that PVDF‑based electrodes suffered extensive delamination after 50 cycles, whereas SBR+CMC and LA133 maintained intact films. SEM cross‑sections (Figure 7) confirm that the gap between electrode and copper foil is smaller for SBR+CMC (1.4 µm) compared to LA133 (1.8 µm), reflecting superior adhesion and reduced mechanical strain.

Optical image of CuO electrodes before (left) and after (right) charge‑discharge cycles using different binders.

SEM and cross‑section SEM of CuO electrodes using different binders. (a, c) SBR+CMC before cycle; (b, d) after cycle; (e, g) LA133 before; (f, h) after.
Rate Performance
Rate tests (Figure 8) revealed that SBR+CMC achieved the highest specific capacities across all rates (0.2→5.0 C) and fully recovered to 87 % of the 0.2 C capacity when the current was reduced again, outperforming LA133 (71.7 %) and PVDF (61.3 %). This underscores the lower polarization and superior ionic/electronic pathways afforded by the SBR+CMC network.
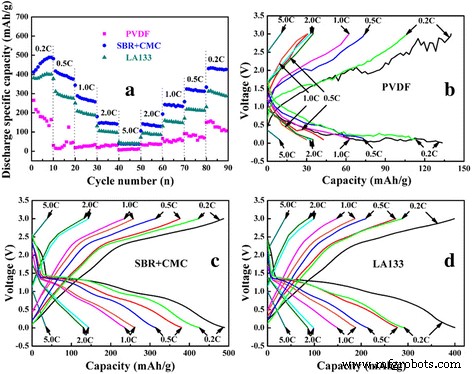
Rate performance (a) and charge‑discharge curves (b–d) for PVDF, SBR+CMC, and LA133 binders.
Cyclic Voltammetry
CV curves (Figure 9a–c) show that SBR+CMC yields the sharpest redox peaks at 0.85 V (CuO→Cu₂O) and 1.28 V (Cu₂O→Cu), indicating low kinetic barriers. The linear dependence of peak current on the square root of scan rate (Figure 9d–f) confirms diffusion‑controlled processes, with the diffusion coefficient for SBR+CMC‑based electrodes being the highest (Table 1). Lower polarization and higher Li⁺ diffusivity explain the superior cycling performance.
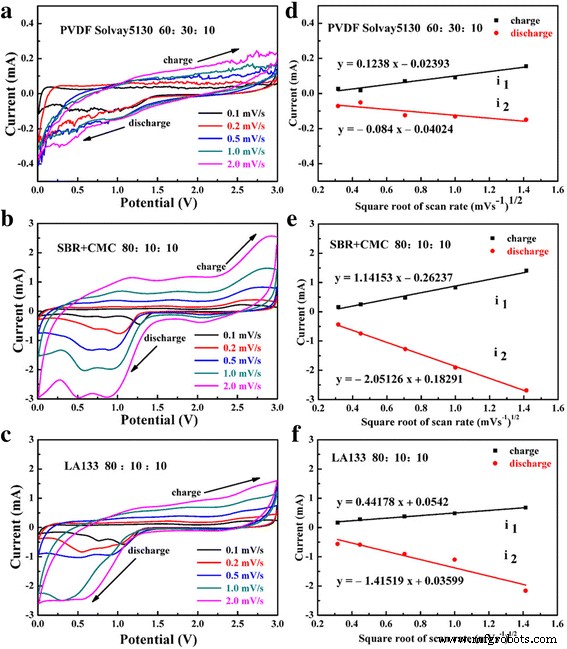
Cyclic voltammograms (left) and peak current vs. √scan rate (right) for PVDF (a, d), SBR+CMC (b, e), and LA133 (c, f).
Electrochemical Impedance Spectroscopy
EIS (Figure 10) shows that SBR+CMC and LA133 yield semicircles of ~50 Ω cm², far lower than PVDF’s >200 Ω cm², indicating faster charge‑transfer and reduced SEI resistance. Table 2 confirms that SBR+CMC electrodes have the lowest bulk resistance (≈200 Ω) compared to PVDF (≈500 kΩ). These impedance metrics correlate with the observed high-rate performance.
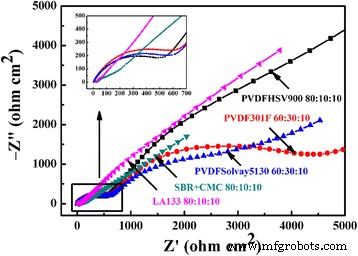
Electrochemical impedance spectra of CuO electrodes using different binders after 50 cycles.
Conclusions
Our systematic study demonstrates that aqueous binders—SBR+CMC and LA133—significantly outperform PVDF for CuO anodes. SBR+CMC, in particular, offers the best combination of electrical conductivity, charge‑transfer kinetics, adhesion, and cycle stability, enabling a 99 % capacity retention over 50 cycles at 0.2 C. Transitioning to water‑soluble binders eliminates toxic solvents, reduces manufacturing costs, and improves environmental sustainability without sacrificing performance.
Nanomaterials
- Enhancing Lithium‑Ion Battery Cathodes: LiNi0.8Co0.15Al0.05O2/Carbon Nanotube Composite with Superior Electrochemical Performance
- Enhanced Lithium‑Ion Battery Anodes: Polypyrrole‑Coated MnO₂ Core–Shell Micromaterials Deliver Superior Cyclic Stability
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- High‑Performance Nanocrystalline Fe₂O₃ Thin‑Film Anodes via Pulsed Laser Deposition for Lithium‑Ion Batteries
- Polypyrrole/ZnO Interlayer Boosts Lithium/Sulfur Battery Performance with Enhanced Polysulfide Suppression
- Mesoporous Silicon Microspheres via In Situ Magnesiothermic Reduction: A High‑Performance Anode for Sodium‑Ion Batteries
- High‑Performance CoSe/NC Anodes from ZIF‑67 Precursors for Lithium‑Ion Batteries
- Enhancing Lithium‑Ion Battery Anodes: Deep Cryogenic Activation of Hemp‑Stem Derived Carbon
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries
- Explore Professional Metal Finishing Options for Durability and Aesthetic Excellence



