Penta‑Siligraphene: A Thermally Stable, High‑Capacity Anode for Li‑Ion Batteries – A First‑Principles Study
Abstract
Using first‑principles calculations, we predict that penta‑siligraphene (P‑Si2C4) is a promising anode material for Li‑ion batteries. The hybrid Si/C structure is more thermally stable than penta‑graphene and possesses a buckled pentagonal lattice that hosts an empty C‑2pz band. This electronic feature readily accepts electrons donated by Li, making Li adsorption highly exothermic. Consequently, each formula unit can accommodate four Li atoms, yielding a theoretical gravimetric capacity of 1028.7 mAh g−1. The Li‑adsorbed material becomes metallic and exhibits Li‑migration barriers below 0.2 eV, ensuring rapid charge‑discharge performance. These findings outline a new design route for two‑dimensional Si/C anodes with high capacity and stability.
Background
Commercial Li‑ion batteries (LIBs) suffer from insufficient energy density, a critical bottleneck for electric vehicle (EV) adoption [1, 2]. Graphite, the dominant anode, offers only 372 mAh g−1, while silicon can deliver 4200 mAh g−1 but expands by up to 420 % during lithiation [5–6]. Integrating Si and C into a single, stable matrix promises the best of both worlds. Siligraphene—graphene with partial Si substitution—has already been synthesized [11–12] and is predicted to deliver 1520 mAh g−1 for g‑SiC5 [13]. However, the sp2→sp3 transition during Li adsorption compromises cycling stability. Designing a pre‑existing sp3‑like network could mitigate this issue.
Carbon’s diverse allotropes illustrate how hybridization affects Li binding. Pristine graphene’s delocalized π‑system resists Li adsorption because charge transfer breaks the π‑bond, yielding adsorption energies above Li metal’s cohesive energy [15]. Hard carbon, containing both sp2 and sp3 sites, stores Li at higher capacities [16–18]. Penta‑graphene, an sp2–sp3 2D carbon, shows strong Li binding in theory [21] and has a higher cohesive energy than graphene, limiting large‑scale synthesis [22]. Replacing the sp3 C atoms in penta‑graphene with Si—which prefers sp3 hybridization—yields penta‑siligraphene (P‑Si2C4). Experiments have produced pentagonal Si ribbons, indicating feasibility [26]. Previous studies on P‑SiC2 revealed tunable transport and unique thermal behavior [27–30], and Si’s inherent affinity for Li suggests potential as a Li‑storage platform [31–32].
In this work, we evaluate P‑Si2C4 as an anode via density functional theory (DFT). We examine its thermodynamic and dynamic stability, Li adsorption energetics, electronic evolution upon lithiation, and Li‑migration pathways, ultimately demonstrating its suitability for high‑performance LIBs.
Computational Methods
All calculations employed VASP [33] with PAW potentials [34–35] and PBE GGA exchange‑correlation [36]. A plane‑wave cutoff of 450 eV and k‑point meshes denser than 0.05 Å−1 were used. Geometry optimizations converged to forces below 0.02 eV Å−1. Electronic band structures were obtained with the HSE06 hybrid functional [37], and density of states used Gaussian smearing (0.05 eV). Bader analysis [39] quantified charge transfer. Li migration paths were located with the climbing‑image nudged elastic band (CINEB) method [40]. Adsorption energies were calculated via Ead = (Ehost+Li – Ehost – n ELi)/n. Van der Waals corrections employed the DFT‑D3(Becke‑Jonson) scheme [41]. The average Li intercalation potential relative to Li+/Li is Vave = –(Ead – Ecoh), expressed in volts.
Results and Discussions
Structure and Stability of Penta‑siligraphene
Penta‑graphene (P‑C6) adopts the P‑421m space group (No. 113) with lattice constants a = b = 3.636 Å [20, 21]. Two carbon types exist: sp3‑like C1 and sp2‑like C2. Replacing all C1 sites with Si yields P‑Si2C4 (P‑Si2C4), whose optimized lattice constants expand to a = b = 4.405 Å, matching prior reports [27–30].
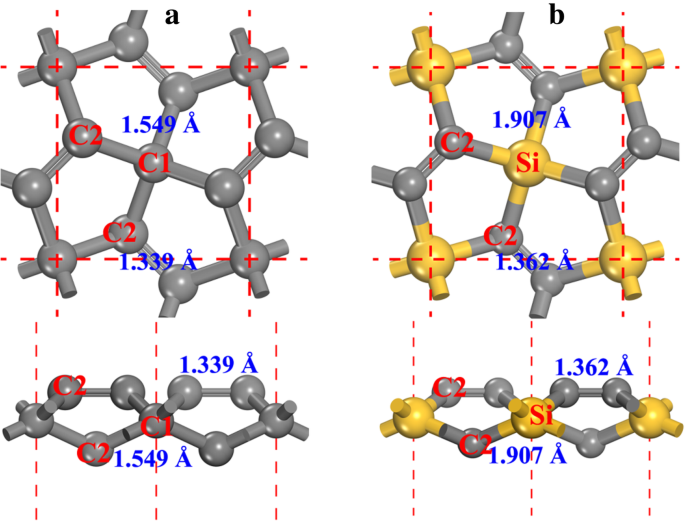
a Ball‑and‑stick models of penta‑graphene (left) and penta‑siligraphene (right). Gray spheres represent C; yellow spheres represent Si.
Table 1 lists cohesive energies of relevant allotropes. P‑C6 (−8.24 eV atom−1) is less stable than graphene (−9.14 eV atom−1), implying challenging large‑scale synthesis. In contrast, P‑Si2C4 (−7.26 eV atom−1) is only 0.2 eV higher than the most stable g‑Si2C4 (−7.46 eV atom−1), indicating feasible fabrication.
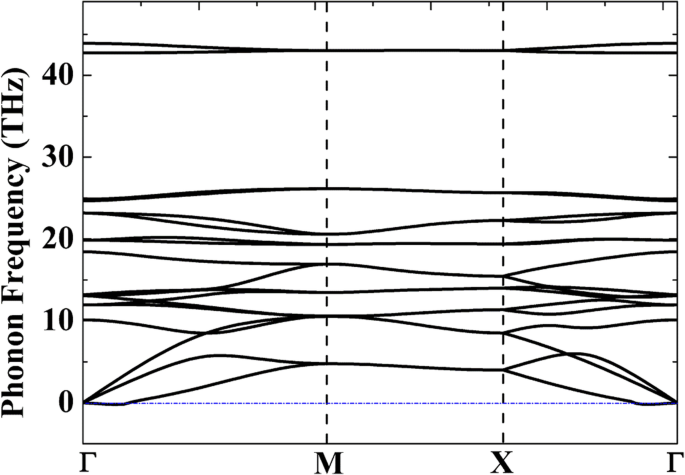
Phonon dispersion of P‑Si2C4 confirms dynamic stability; minor imaginary modes (<0.14 THz) near Γ are negligible [42].
Ab initio molecular dynamics (AIMD) simulations (3×3 and 4×4 supercells) show the structure remains intact up to 2000 K over 20 ps, but degrades at 2500 K, consistent with its cohesive energy trend.
Li Adsorption on Penta‑siligraphene
Four stable Li adsorption sites were identified: top of Si (T), hollow of the Si2C3 pentagon (H), and two bridge sites (B1 below the layer, B2 above). Table 2 lists adsorption energies and heights. The most favorable site is B1 (−1.922 eV). B1 and H have comparable energies (−1.905 eV). When all B1 sites are filled (Li2Si2C4), Li occupies H sites, and at full loading (Li4Si2C4) all H sites are filled. van der Waals corrections contribute an additional 0.12–0.17 eV, reinforcing Li binding.
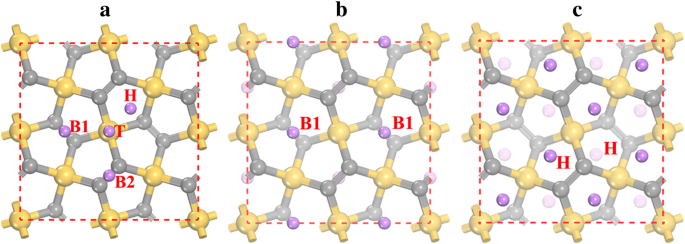
Li adsorption sites (a) and optimized configurations for Li2Si2C4 (b) and Li4Si2C4 (c). Colors: Si yellow, C gray, Li purple.
Adsorption energies below the bcc Li cohesive energy (−1.86 eV atom−1) confirm electrochemical activity. For Li/C ratios below 2/3 (i.e., four Li per unit cell), the average potential is 0.1–0.2 V, favorable for high‑voltage full cells. Lattice contraction upon lithiation is minimal (≤0.94 %), indicating excellent volumetric stability (Fig. 4).
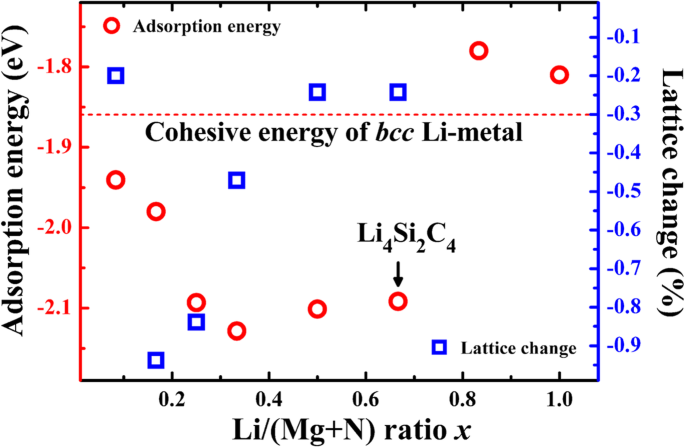
Li adsorption energies (red circles) and lattice constant change (blue squares) versus Li/C ratio. The cohesive energy of bcc Li is shown for reference.
Electronic Structure Analysis of the Penta‑siligraphene upon Li Adsorption
Pristine P‑Si2C4 is a semiconductor with an indirect gap of 2.35 eV, smaller than penta‑graphene’s 3.46 eV [Additional file 1: Figure S4]. The reduced gap arises from increased dispersion of the highest occupied band (bands 10–12), especially at M and Γ. The empty C‑2pz band (band 13) provides an electron‑accepting channel for Li, stabilizing adsorption.
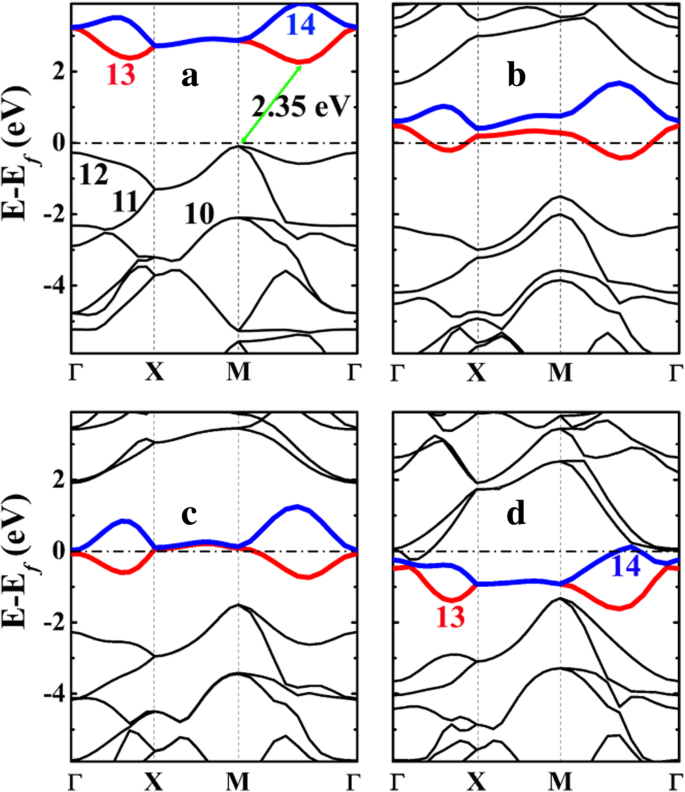
Band structures of (a) pristine P‑Si2C4 and (b–d) lithiated states P‑LiSi2C4, P‑Li2Si2C4, and P‑Li4Si2C4. Metallic behavior emerges upon lithiation.
Bader charge analysis shows Si carries +2.35 e, while C1 in penta‑graphene carries only +0.08 e. The stronger Coulomb attraction in P‑Si2C4 broadens the occupied band, enhancing Li acceptance. Buckling (≈0.876 Å in Li‑rich state) approaches ideal tetrahedral angles, indicating stronger sp3 bonding upon Li insertion. The empty C‑pz states are filled by Li‑donated electrons, leading to metallic conductivity (Fig. 6).
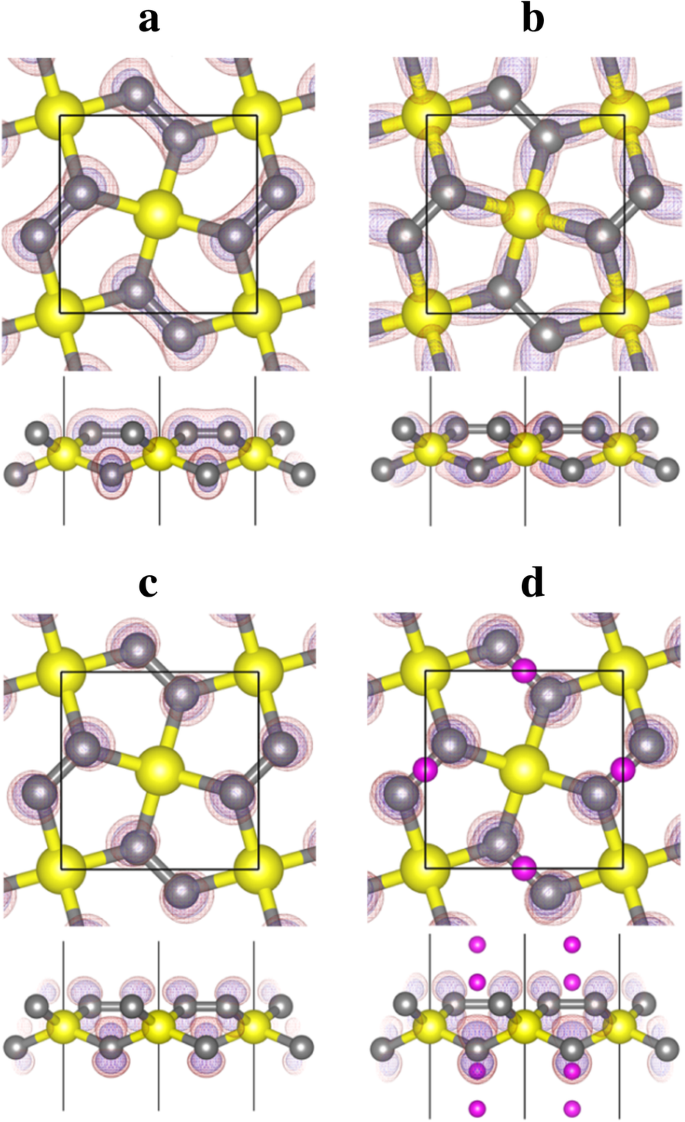
Band‑decomposed charge densities: (a) band 10, (b) band 12 (C‑Si σ), (c) band 13 (C‑pz), and (d) band 13 in Li‑4. The C‑pz states provide the Li‑accepting sites.
Li‑Ion Migration Dynamics on P‑Si2C4
Fast Li diffusion is essential for high‑rate capability. In the dilute‑Li regime (one Li per supercell), the lowest‑energy pathway (Path‑1) connects adjacent B1 sites, with an NEB barrier of 0.117 eV—lower than P‑C6 (0.17 eV) and g‑Si2C4 (0.548 eV).
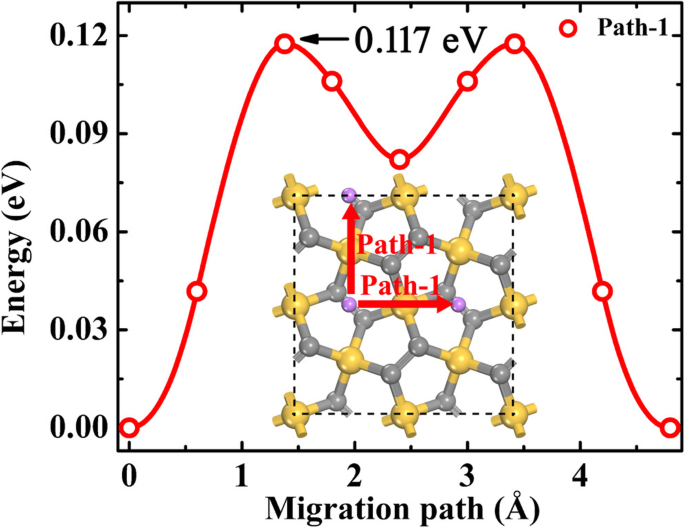
Dilute‑Li migration path and energy profile. Gray, gold, and purple spheres denote C, Si, and Li, respectively.
For dilute‑Li vacancies (removing one Li from Li4Si2C4), three migration paths were examined. Path‑3 (H ↔ H via B1) exhibits the lowest barrier of 0.052 eV, while Paths 1 and 2 show 0.155–0.165 eV. All barriers remain below those of P‑C6 (0.25 eV) and g‑Si2C4 (0.233 eV), indicating superior rate capability (Fig. 8).
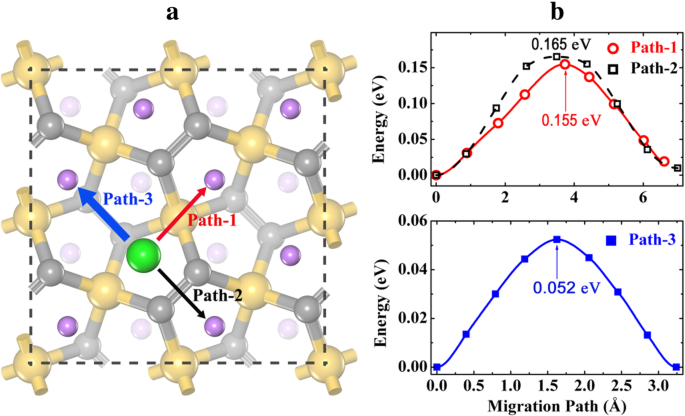
Li vacancy migration pathways (a) and energy profiles (b) on fully lithiated P‑Li4Si2C4. Fast pathways form a two‑dimensional diffusion network.
Conclusion
First‑principles calculations reveal that 2D penta‑siligraphene (P‑Si2C4) is a thermally stable, high‑capacity anode for Li‑ion batteries. Phonon and AIMD analyses confirm stability up to 2000 K. The buckled pentagonal lattice hosts empty C‑2pz states that accept Li electrons, enabling a 1028.7 mAh g−1 capacity with minimal volume change. Lithiated P‑Si2C4 becomes metallic, ensuring good electronic conductivity. Li‑migration barriers below 0.2 eV guarantee rapid charge‑discharge kinetics, outperforming penta‑graphene and siligraphene. These attributes make P‑Si2C4 a compelling candidate for next‑generation LIB anodes, offering a practical pathway to high‑performance Si/C composites.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Nanomaterials
- Sol‑Gel Fabricated SiO₂@C/MWNT Nanocomposites Deliver Superior Li‑Ion Battery Anodes
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- Mesoporous Silicon Microspheres via In Situ Magnesiothermic Reduction: A High‑Performance Anode for Sodium‑Ion Batteries
- Carbonized Leaf Cathodes with Melt‑Diffusion Selenium for High‑Performance Sodium‑Selenium Batteries
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Molecular‑Sieve‑Enhanced Separator Boosts Lithium‑Ion Battery Performance & Safety
- Advancing Lithium‑Ion Batteries: Binder‑Free Electrode Technology for Higher Energy Density
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries



