3D Interconnected V6O13 Nanosheets on Carbonized Textile: A High‑Performance Flexible Cathode for Lithium‑Ion Batteries
Abstract
Three‑dimensional, free‑standing nanostructures have emerged as highly promising electrodes for energy storage, delivering superior electrochemical performance and enabling wearable power solutions. In this study, we grew interconnected V6O13 nanosheets directly onto a flexible carbonized textile (c‑textile) using a seed‑assisted hydrothermal method, forming a 3‑D free‑standing electrode for lithium‑ion batteries (LIBs). The pristine electrode delivers a specific capacity of 170 mA h g−1 at 300 mA g−1. A subsequent coating with carbon nanotubes (CNTs) further enhances the capacity by 12–40 % across various current rates, while maintaining a reversible capacity of 130 mA h g−1 (74 % of the initial value) after 300 cycles at 300 mA g−1. These metrics surpass most mixed‑valence vanadium oxides, owing to the synergistic 3‑D nanostructure that promotes Li+ diffusion and the hierarchical conductive network formed by CNTs and the carbon fiber scaffold.
Background
Vanadium oxides—V6O13, V3O7, V2O5—are attractive cathode materials for LIBs due to their low cost, high theoretical capacity, and the abundant supply of vanadium [1–6]. V6O13 is especially compelling, with a theoretical capacity of 417 mA h g−1 and an energy density of 890 Wh kg−1 when fully lithiated to Li8V6O13 [2, 8]. However, its practical performance is hampered by a drop in electronic conductivity upon lithiation and modest Li+ diffusion coefficients (10−8–10−9 cm2 s−1) [7, 9]. Three‑dimensional nanostructuring mitigates these issues by enhancing ion and electron transport while preventing particle aggregation [15–20]. Prior work has demonstrated 3‑D V6O13 nanostructures on rigid substrates, yet direct growth of mixed‑valence vanadium oxide on flexible supports remains challenging, and flexible cathodes for wearable devices have not been reported.
In this work, we present a straightforward hydrothermal route that successfully grows interconnected V6O13 nanosheets on carbonized bamboo textile, producing a 3‑D free‑standing electrode. The resulting cathode delivers 161 and 105 mA h g−1 at 300 and 1200 mA g−1, respectively. CNT coating further raises the capacities to 170 and 140 mA h g−1 and improves cycle life, retaining 74 % of the initial capacity after 400 cycles at 300 mA g−1. These enhancements are attributed to the synergistic effect of the 3‑D nanostructure and the hierarchical conductive network.
Methods
Synthesis of c‑textile
Commercial bamboo cloth was soaked in a 2.5 g NaF/60 mL H2O solution for 1 h, then dried at 120 °C for 5 h. The dried textile was carbonized at 800 °C under nitrogen for 30 min, yielding the flexible c‑textile.
Growth of 3D V6O13 nanostructure on c‑textile
The c‑textile was briefly oxidized in 80 wt % nitric acid (30 min) to introduce oxygen‑bearing functional groups. A 1 mg V2O5 suspension in 5 mL deionized water was sonicated for 15 min to create a seed solution. The oxidized textile was immersed in this suspension for 2 h, dried, and heated at 300 °C for 10 min to nucleate vanadium oxide seeds. Subsequently, 16 mg V2O5 was added to 224 µL of 30 wt % H2O2, stirred 10 min, and diluted with 40 mL water; the mixture was stirred 30 min before being transferred to a 25 mL stainless‑steel autoclave. The oxidized c‑textile was immersed, and the autoclave was maintained at 180 °C for 48 h. After washing with water and alcohol and drying at 60 °C for 8 h, a flexible 3‑D V6O13 electrode was obtained. CNTs were then coated by repeatedly dipping the electrode in a 0.5 mg mL−1 NMP suspension of multi‑walled CNTs and drying, producing the V6O13/CNT composite.
Characterization of Materials
Morphology was examined by scanning electron microscopy (SEM, Philips XL30 FEG) and transmission electron microscopy (TEM, JEOL JEM‑2010). X‑ray photoelectron spectroscopy (XPS, K‑Alpha) was performed with a monochromatic Al Kα source.
Battery Fabrication and Electrochemical Measurements
CR2016‑type coin cells were assembled in an argon glove box, using the V6O13 electrode (≈1 mg cm−2) as the cathode and lithium foil as the anode. The electrolyte was 1 M LiPF6 in a 1:1:1 volume mixture of EC/DEC/DMC, and a polypropylene separator was employed. Cells were cycled between 1.5 and 4.0 V (vs. Li/Li+) at 25 °C on a LAND battery test system. Electrochemical impedance spectroscopy (EIS) was performed using an Autolab PGSTAT302N workstation over 10 mHz–10 kHz.
Results and Discussion
The growth process of the 3‑D V6O13 nanosheets on c‑textile is illustrated in Additional file 1: Figure S1. Initially, the bamboo cloth (Fig. 1a) was carbonized to produce c‑textile (Fig. 1b). SEM images (Fig. 2a) show that the c‑textile consists of woven bundles of carbon fibers (~5 µm diameter), exhibiting excellent flexibility and a sheet resistivity of 5 Ω sq−1. After hydrothermal growth, the textile becomes coated with a thin yellow‑green film (Fig. 1d) while remaining flexible. The resistivity increases to 50 Ω sq−1, yet the structure remains intact. SEM (Fig. 2b,c) reveals interconnected nanosheets several microns long and a few hundred nanometers wide, forming the 3‑D architecture. High‑resolution TEM (Fig. 2f) displays lattice fringes with a spacing of 3.5 Å, matching the (110) plane of orthorhombic V6O13 (PDF No. 71‑2235). XPS confirms the mixed valence of vanadium, with V4+ (516.0 eV) and V5+ (517.3 eV) peaks, consistent with literature values [25–27]. These results confirm successful growth of 3‑D V6O13 on c‑textile.

Optical images of (a) commercial textile, (b) carbonized textile, (c) rolled c‑textile, and (d) c‑textile with grown V6O13 at the rolled state, inset: at the flat state

Microstructure of 3D free‑standing interconnected V6O13 nanosheets on c‑textile: (a) low‑resolution SEM of c‑textile without nanosheets; (b) with nanosheets; (c) high‑resolution SEM of nanosheets; (d) high‑resolution SEM of nanosheets; (e) low‑resolution TEM; (f) high‑resolution TEM
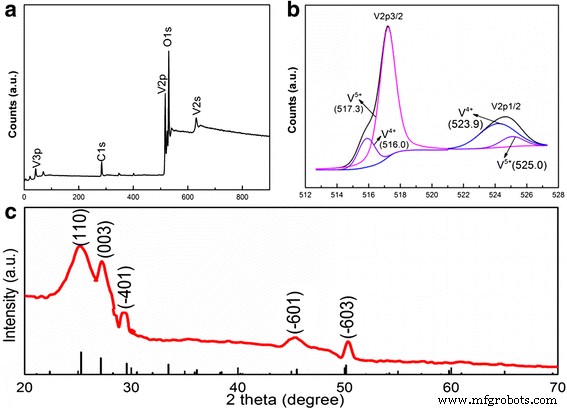
(a) Survey XPS spectrum; (b) V 2p and O 1s peaks with fitting; (c) XRD pattern of V6O13
Electrochemical performance was assessed in half‑cell configuration. CV curves (Fig. 4a) show distinct redox peaks at 2.8/2.5 V, with additional broad peaks at ~3.2 V and 2.3 V (anodic) and ~1.8 V (cathodic), indicating multi‑step phase transitions consistent with prior reports [2, 28]. Galvanostatic discharge/charge at 30 mA g−1 (Fig. 4b) exhibits sloped plateaus at 2.3 and 2.8 V. Specific capacities at 30, 150, 300, 600, and 1200 mA g−1 are 253, 176, 161, 133, and 105 mA h g−1, respectively, highlighting the advantage of the open 3‑D architecture for Li+ transport. SEM of the cycled electrode (Additional file 1: Figure S2) confirms that the nanosheet morphology is preserved, underscoring structural integrity.
To further enhance conductivity, the electrode was coated with CNTs. SEM (Fig. 5a,b) shows CNTs uniformly deposited on the nanosheets, forming inter‑sheet bridges (Fig. 5c). The sheet resistivity drops from 50 to 20 Ω sq−1. CV peaks remain at the same potentials but with increased current, indicating faster kinetics. Rate performance (Fig. 4c) improves: capacities of 261, 185, 170, 153, and 140 mA h g−1 at 30, 150, 300, 600, and 1200 mA g−1 represent a 12–40 % boost over the uncoated electrode. Li+ diffusion coefficients calculated from CV are 4.79 × 10−8 and 2.01 × 10−8 cm2 s−1 for the anodic and cathodic processes, respectively, higher than those of the bare electrode (2.42 × 10−8 and 1.70 × 10−8 cm2 s−1).
EIS Nyquist plots (Fig. 6a) show a reduced charge‑transfer resistance (Rct) of 37.24 Ω for the CNT‑coated electrode versus 55.58 Ω for the bare one, attributable to the CNT‑mediated conductive network. The equivalent circuit (inset) includes a constant phase element and Warburg impedance to account for Li+ diffusion.
Cycling stability (Fig. 4d) is markedly improved: the CNT‑coated electrode retains 74 % of its initial capacity after 300 cycles at 300 mA g−1, compared to 42 % for the bare electrode. The enhanced durability is ascribed to mechanical reinforcement by CNTs, continuous attachment of nanosheets to the conductive scaffold, and suppression of side reactions. Future improvements may involve reducing c‑textile resistance, doping V6O13 with sulfur, or applying conductive polymer coatings.
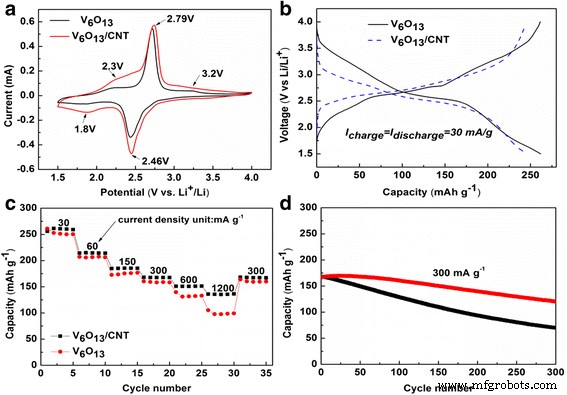
Electrochemical performance of 3D free‑standing V6O13 electrodes with/without CNT coating: (a) CV curves; (b) galvanostatic charge/discharge; (c) rate capability; (d) cycling performance.

Microstructure of CNT‑coated V6O13 electrode: (a) low‑resolution SEM; (b) high‑resolution SEM showing CNTs covering nanosheets; (c) high‑resolution SEM showing CNT bridges between nanosheets.
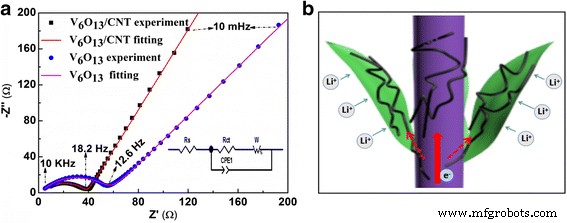
(a) Nyquist plots of V6O13 electrode with/without CNT coating; (b) schematic of electron transport pathways in CNT‑coated electrode.
Conclusions
We have demonstrated a facile seed‑assisted hydrothermal route to grow 3‑D, free‑standing V6O13 nanosheets directly on a flexible carbonized textile, producing a high‑performance, flexible cathode for LIBs. The electrode delivers robust capacity and excellent cycle life, further enhanced by CNT coating, which establishes a hierarchical conductive network and improves both electronic and ionic transport. This strategy—combining 3‑D nanostructured building blocks with a hierarchical conductive scaffold—can be extended to other electrode chemistries to achieve superior energy‑storage performance.
Abbreviations
- 3D:
Three dimensional
- CE:
Coulombic efficiency
- CNT:
Carbon nanotube
- c-textile:
Carbonized textile
- CV:
Cyclic voltammetry
- DEC:
Diethyl carbonate
- DMC:
Dimethyl carbonate
- EC:
Ethylene carbonate
- EIS:
Electrochemical impedance spectroscopy
- LIB:
Lithium‑ion battery
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
Nanomaterials
- High‑Performance Nanocrystalline Fe₂O₃ Thin‑Film Anodes via Pulsed Laser Deposition for Lithium‑Ion Batteries
- Carbonized Leaf Cathodes with Melt‑Diffusion Selenium for High‑Performance Sodium‑Selenium Batteries
- Sea Urchin‑Shaped NiCo₂O₄ via Charge‑Driven Self‑Assembly for Superior Li‑Ion Battery Performance
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- Penta‑Siligraphene: A Thermally Stable, High‑Capacity Anode for Li‑Ion Batteries – A First‑Principles Study
- High‑Performance CoSe/NC Anodes from ZIF‑67 Precursors for Lithium‑Ion Batteries
- Molecular‑Sieve‑Enhanced Separator Boosts Lithium‑Ion Battery Performance & Safety
- Enhancing 2D Perovskite Solar Cell Performance Through Water-Assisted Crystallization
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries
- Engineered Nickel‑Cobalt Layered Double Hydroxides: Tunable Thin‑Layer Nanosheets for Superior Supercapacitor Performance



