High‑Performance Nanocrystalline Fe₂O₃ Thin‑Film Anodes via Pulsed Laser Deposition for Lithium‑Ion Batteries
Abstract
We present a scalable pulsed laser deposition (PLD) route to fabricate nanocrystalline Fe₂O₃ thin films directly on conductive substrates for use as lithium‑ion battery anodes. The films exhibit an outstanding high‑rate capacity of 510 mAh g⁻¹ at 15,000 mA g⁻¹ and a reversible capacity of 905 mAh g⁻¹ after 200 cycles at 100 mA g⁻¹—among the best performances reported for Fe₂O₃ anodes. The superior lithium storage is attributed to the ultrafine nanocrystalline architecture, which shortens lithium‑ion diffusion paths and mitigates particle agglomeration, thereby enhancing cycling stability. These findings position the Fe₂O₃ thin film as a promising anode for all‑solid‑state thin‑film batteries.
Background
As lithium‑ion batteries (LIBs) become ubiquitous in portable electronics and electric vehicles, the demand for electrodes with higher energy and power densities has spurred extensive research into transition‑metal oxides (TMOs). TMOs such as Co₃O₄, NiO, Fe₂O₃, and CuO offer higher theoretical capacities and improved safety compared to conventional graphite anodes. Fe₂O₃, in particular, delivers a theoretical capacity of ~1,005 mAh g⁻¹, is inexpensive, abundant, and environmentally benign. However, the large volume change during lithiation/delithiation typically leads to pulverization, loss of electrical contact, and rapid capacity fade. To address these challenges, nanostructured Fe₂O₃ forms—nanorods, nanoflakes, hollow spheres, core‑shell arrays, and micro‑flowers—have been explored.
Nanocrystalline thin‑film anodes fabricated by PLD or sputtering (e.g., NiO, MnO, Cr₂O₃, CoFe₂O₄, Si, Ni₂N) offer superior electrochemical performance due to excellent electrical contact, shortened ion diffusion lengths, and structural robustness. Thin‑film TMOs also enable integration into all‑solid‑state microbatteries, potentially replacing the reactive lithium metal anode. Yet, Fe₂O₃ thin‑film anodes prepared by PLD or sputtering have been limited, with reported capacities far below the theoretical value.
In this study, we deposit nanocrystalline Fe₂O₃ films by PLD at room temperature. The resulting films, with grain sizes of several tens of nanometers, deliver a reversible capacity of 905 mAh g⁻¹ at 100 mA g⁻¹ and a high‑rate capacity of 510 mAh g⁻¹ at 15,000 mA g⁻¹, highlighting their potential for high‑performance LIBs and all‑solid‑state thin‑film batteries.
Experimental
Synthesis of Nanocrystalline Fe₂O₃ Films
Fe₂O₃ films were deposited directly onto copper foils or stainless‑steel substrates by PLD in an oxygen atmosphere. A KrF excimer laser (λ = 248 nm) was focused on a rotating iron target at 5 Hz and 500 mJ pulse energy. The target–substrate distance was 40 mm. Room‑temperature deposition under an oxygen pressure of 0.3 Pa yielded uniform, ~200 nm thick films, as measured by AFM (Park Systems XE7). The film mass (0.121 mg) was determined by weight change using an electrobalance (Mettler Toledo).
Material Characterization
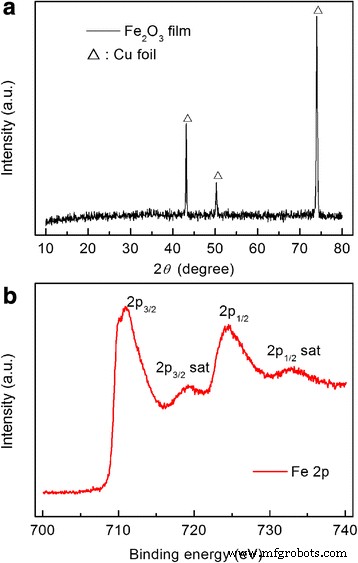
Phase composition was confirmed by XRD (Rigaku D/Max, Cu Kα, λ = 1.5406 Å). TEM and SAED (JEOL 100CX) revealed nanocrystalline grains with an average size of 10–30 nm and lattice fringes corresponding to α‑Fe₂O₃ (110) d = 0.251 nm. SEM (Hitachi SU8010) showed a uniform nanometer‑scale particle distribution. XPS (Thermo Scientific ESCALAB 250XI) confirmed the Fe³⁺ oxidation state, with Fe 2p₃/₂ at 710.9 eV and Fe 2p₁/₂ at 724.5 eV.
Electrochemical Measurements
Coin cells (CR2032) were assembled in an Ar glove box (O₂/H₂O < 0.1 ppm) with Li metal counter electrode and 1:1:1 EC/DMC/LiPF₆ electrolyte. Galvanostatic cycling was performed at 25 °C using a LAND‑CT2001A system across 0.01–3 V. CV and AC impedance were measured with a CHI660E workstation at 0.1 mV s⁻¹ scan rate.
Results and Discussion
Figure 1a shows the XRD pattern: only Cu substrate peaks are visible, indicating amorphous or ultrafine crystalline Fe₂O₃. XPS spectra (Figure 1b) confirm Fe³⁺ oxidation states. TEM (Figure 2a) and SAED (Figure 2b) illustrate polycrystalline Fe₂O₃ nanograins; SEM (Figure 2c) confirms uniform nanometer‑scale morphology. Thus, the room‑temperature PLD process yields ultrafine nanocrystalline Fe₂O₃ films.

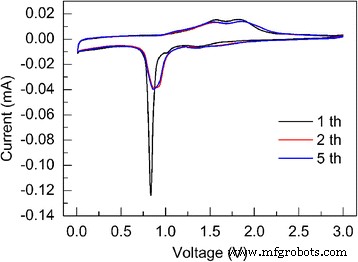
CV curves (Figure 3) display the expected three cathodic peaks at 1.38, 1.02, and 0.84 V, corresponding to lithium insertion, phase transition, and full reduction to Fe⁰, respectively. Two anodic peaks at 1.57 and 1.85 V represent Fe⁰ → Fe²⁺ and Fe²⁺ → Fe³⁺ oxidation. The overlapping CVs after the second cycle confirm electrochemical reversibility.
Galvanostatic charge–discharge profiles (Figure 4a) exhibit clear voltage plateaus matching the CV peaks. The initial discharge capacity is 1,183 mAh g⁻¹, while the first charge is 840 mAh g⁻¹, yielding a Coulombic efficiency of 71 %—typical for conversion‑type anodes due to SEI formation.
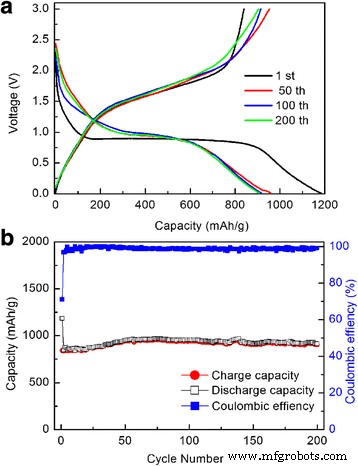
After 200 cycles at 100 mA g⁻¹, the reversible capacity stabilizes at 905 mAh g⁻¹ with nearly 100 % Coulombic efficiency, surpassing most reported Fe₂O₃ thin‑film anodes. The capacity increase in the first 70 cycles likely stems from electrode activation and the formation of conductive polymer/gel‑like layers.
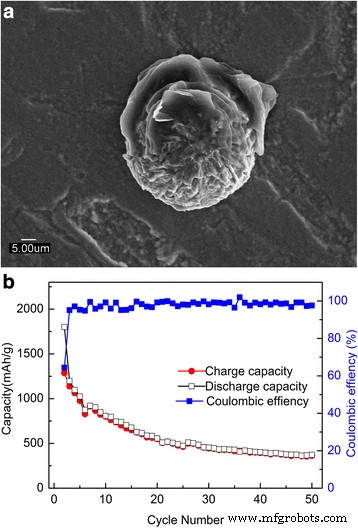
To elucidate the role of particle size, we annealed films at 400 °C on stainless steel. SEM (Figure 5a) shows grain growth, and cycling (Figure 5b) yields only 263 mAh g⁻¹ after 100 cycles—dramatically lower than the as‑deposited films. Similar results were obtained for 400 °C films on stainless steel (Figure 6), where the capacity dropped to 361 mAh g⁻¹ after 50 cycles. These results confirm that the ultrafine nanocrystalline structure is essential for high reversible capacity and cycling stability.

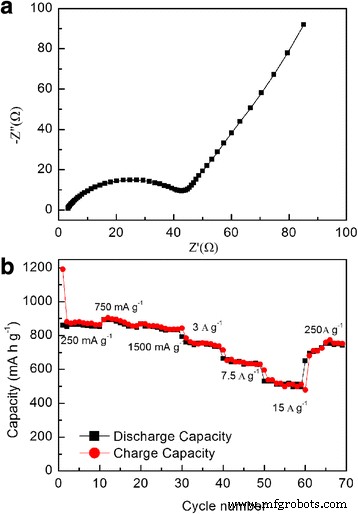
Electrochemical impedance spectroscopy (Figure 7a) shows a low charge‑transfer resistance (~50 Ω), reflecting excellent electronic conductivity from the direct contact between the film and the substrate and the nanocrystalline architecture. The rate capability test (Figure 7b) reveals capacities of 855, 843, 753, 646, and 510 mAh g⁻¹ at 750, 1,500, 3,000, 7,500, and 15,000 mA g⁻¹, respectively—over 59 % of the capacity at 250 mA g⁻¹. When the current is reduced back to 250 mA g⁻¹, the capacity recovers to 753 mAh g⁻¹, confirming excellent rate performance and structural resilience.
Conclusions
We have demonstrated that nanocrystalline Fe₂O₃ films deposited by PLD at room temperature deliver exceptional electrochemical performance: a reversible capacity of 905 mAh g⁻¹ after 200 cycles at 100 mA g⁻¹ and a high‑rate capacity of 510 mAh g⁻¹ at 15,000 mA g⁻¹. The ultrafine nanocrystalline structure shortens lithium‑ion diffusion paths, accommodates volume expansion, and maintains structural integrity, leading to superior cycling stability and rate capability. These attributes make nanocrystalline Fe₂O₃ thin‑film anodes attractive for high‑performance LIBs and all‑solid‑state thin‑film batteries.
Abbreviations
- AFM:
Atomic force microscope
- CV:
Cyclic voltammetry
- DMC:
Dimethyl carbonate
- EC:
Ethylene carbonate
- LIB:
Lithium‑ion battery
- PLD:
Pulsed laser deposition
- SEAD:
Selected area electron diffraction
- SEI:
Solid electrolyte interface
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
- TMOs:
Transition metal oxides
- XPS:
X-ray photoelectron spectroscopy
- XRD:
X-ray diffraction
Nanomaterials
- Arkema Unveils 100% Renewable PVDF Grades for Lithium‑Ion Batteries
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Enhanced Lithium‑Ion Battery Anodes: Polypyrrole‑Coated MnO₂ Core–Shell Micromaterials Deliver Superior Cyclic Stability
- Binder Selection for Copper Oxide Anodes: Why SBR+CMC and LA133 Outperform PVDF in Lithium‑Ion Batteries
- High‑Capacity Si/Graphene Composite Anode Fabricated via Magnesium‑Thermal Reduction for Li‑Ion Batteries
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- High‑Performance CoSe/NC Anodes from ZIF‑67 Precursors for Lithium‑Ion Batteries
- Molecular‑Sieve‑Enhanced Separator Boosts Lithium‑Ion Battery Performance & Safety
- Enhancing Lithium‑Ion Battery Anodes: Deep Cryogenic Activation of Hemp‑Stem Derived Carbon
- NiCo2S4 Nanocrystals on N‑Doped CNTs: A High‑Performance Anode for Next‑Generation Lithium‑Ion Batteries



