Biocompatibility of Graded Nano‑Glass/Zirconia Dental Materials After Aging
Abstract
A graded nano‑glass/zirconia (G/Z) system was created by infiltrating nano‑glass into a nano‑zirconia surface, enhancing core‑veneer bond strength. Aging is a critical concern for yttrium‑stabilized tetragonal zirconia polycrystals (Y‑TZP), so it is essential to assess how aging affects the biocompatibility of G/Z systems before clinical use. Human gingival fibroblasts (HGFs) were seeded onto unaged and aged G/Z and Y‑TZP specimens for 2–72 h. Tests included oral mucous membrane irritation, cell viability, adhesion, and oxidative stress. Metabolic activity decreased significantly in aged G/Z and Y‑TZP at 72 h (P < 0.00001). No significant difference in viability was observed between G/Z and Y‑TZP across all time points, before or after aging. ROS production increased at 72 h for aged G/Z and Y‑TZP, but differences were not significant (P > 0.05). Adhesion rates rose after aging for both materials, yet G/Z and Y‑TZP showed comparable adhesion (P > 0.05). Macroscopic and microscopic irritation scores for both aged and unaged G/Z were 0, indicating no irritation.
Conclusions
The G/Z system demonstrates excellent biocompatibility and holds promise for future clinical applications.
Background
Dental zirconia ceramics, particularly 3 mol% yttrium‑stabilized tetragonal zirconia polycrystals (3Y‑TZP), are prized for their high strength and fracture resistance due to transformation toughening mechanisms, making them common in prosthetic devices [1]. However, veneering porcelain applied to zirconia cores often fails via chipping or delamination, primarily because of mismatched thermal expansion coefficients and elastic moduli between core and veneer [2, 3, 4]. To address this, we previously introduced a graded nano‑glass/zirconia (G/Z) system that infiltrates low‑modulus nano‑glass into a sintered nano‑zirconia surface, achieving bond strengths three times greater than conventional systems [4].
Aging of Y‑TZP—triggered by humidity, mechanical loading, and low temperature—induces surface roughening, microcracks, and a tetragonal‑to‑monoclinic (t‑m) phase transformation, which can compromise mechanical properties [5–9]. Since surface chemistry and roughness influence biocompatibility, it is crucial to evaluate how aging affects the G/Z system’s interaction with biological tissues.
Previous work by Zhang et al. demonstrated the mechanical advantages of a glass‑infiltrated zirconia composite [10, 11], yet the biocompatibility of this graded composite, especially post‑aging, remains unknown. This study addresses that gap by comparing G/Z to the clinically accepted Y‑TZP, evaluating cell viability, morphology, adhesion, oxidative stress, and mucosal irritation both before and after aging.
Methods
Preparation of Specimens
Y‑TZP specimens, approved for clinical use, served as the control. All samples were uniform plates (1.5 × 1.5 × 0.2 cm) following ISO 13356 standards for bending bars with polished surfaces.
Preparation of G/Z Specimens
Glass powders were milled to nanometer size using an Emax grinder. The infiltrating glass composition is detailed in Table 1 [4]. Y‑zirconia powders (5.18 wt% Y₂O₃, TZ‑3Y‑E grade, Tosoh) were uniaxially pressed at 150 MPa for 2 min, partially sintered at 1350 °C for 2 h, and ball‑milled to 200‑mesh. Substrate specimens were presintered at 1200 °C for 2 h to form a porous network. Melted glass slurries were applied to the top surface and infiltrated at 1350 °C for 2 h, simultaneously achieving infiltration and densification.
Preparation of Y‑TZP Specimens
Y‑TZP blanks were milled and sintered to full density using a CAD/CAM system (Zenostar, Weiland Dental).
Cell Culture
HGFs were cultured in DMEM/F‑12 supplemented with 10% fetal bovine serum, 1% penicillin/streptomycin, 1% L‑glutamine, and 1% non‑essential amino acids at 37 °C, 5% CO₂. Media were refreshed every 3 days. Cells were detached with PBS and trypsin‑EDTA, then seeded on each substrate at 1 × 10⁵ cells/mL for all assays.
Aging
Mechanical aging simulated chewing conditions: specimens were placed in artificial saliva at 37 °C, subjected to a three‑point flexure load of 80 N at 2 Hz for 10⁵ cycles, following protocols [12, 13].
Cell Viability
Viability was assessed at 2, 24, 48, and 72 h using the alamarBlue® assay (10% in DMEM). After incubation, 500 µL of dye was added, incubated for 4 h, and fluorescence was measured (excitation 530 nm, emission 580 nm) on a Synergy™ H4 Microplate Spectrophotometer (BioTek). Experiments were performed in triplicate across three runs. Viability (%) = (absorbance of treated wells) / (absorbance of control wells).
Oxidative Stress
Reactive oxygen species (ROS) levels were quantified via chemiluminescence using the Reactive Oxygen Species Assay Kit (Nanjing Jiancheng Bioengineering Institute).
Cell Adhesion
HGFs were cultured for 2 h on specimen surfaces pre‑ and post‑aging. After fixation, nuclei were stained with DAPI (Yeasen), and images were captured with a Carl Zeiss LSM 510 inverted fluorescence microscope. Adhesion rates were calculated as adhered cells / total seeded cells across five random 450 µm × 450 µm fields at ×200 magnification.
Cell Morphology
After 2 h incubation, cells on G/Z surfaces were fixed and stained for F‑actin with rhodamine phalloidin (1:100 in 3% BSA). Images were taken with a Zeiss LSM 510. Morphology was also examined by SEM (Philips XL‑30 ESEM). Samples were mounted on glass coverslips and counterstained with DAPI.
Oral Mucous Membrane Irritation Test
Ten Wistar male mice received aged G/Z in one cheek pouch and unaged G/Z in the contralateral side, following YY/T 0127.13‑2009 standards. After 2 weeks, pouches were inspected macroscopically, and buccal mucosa was histologically examined via H&E staining. Irritation indices were calculated by subtracting control scores from test scores.
Statistical Analyses
One‑way ANOVA (SPSS 22.0) evaluated pooled data for viability, oxidative stress, and adhesion. Significance was set at P < 0.05.
Results
Graded Layer Structure
The graded layer thickness averaged 0.9–1.0 mm. SEM images (Fig. 1a, b) reveal a surface composed of residual glass, glass‑coated zirconia grains, and intergranular voids—features that facilitate strong core‑veneer adhesion. EDS mapping (Fig. 1c) shows a gradient: Zr concentration increases deeper into the material, while Si, Al, and La decrease, confirming the intended compositional gradient [4].
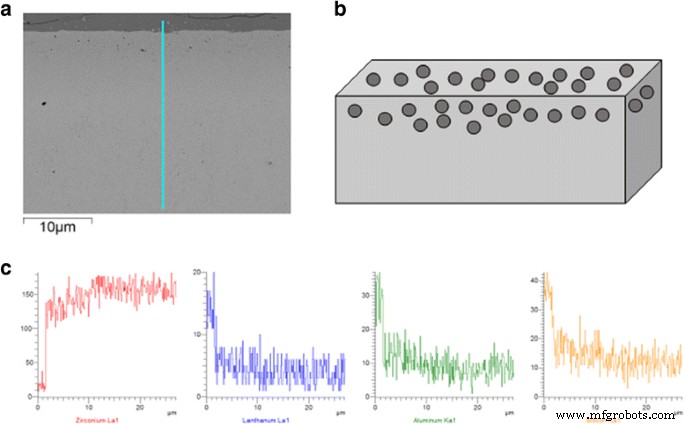
Physical and chemical properties of G/Z. a Structural diagram. b SEM image. c EDS analysis of the functionally graded layer
Cell Viability
Metabolic activity decreased significantly in aged G/Z and Y‑TZP at 72 h (P < 0.00001) (Fig. 2a). No significant drop was observed at 2, 24, or 48 h for either material. Viability of G/Z and Y‑TZP did not differ at any time point, pre‑ or post‑aging (P > 0.05). The data are summarized in Figure 2a.
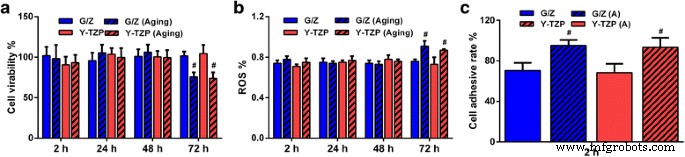
Biocompatibility of G/Z and Y‑TZP before and after aging. Data represent the mean ± SD, n = 5. a Cell viability of aged and unaged specimen‑treated HGF. b ROS production of aged and unaged specimen‑treated HGF. c Cell adhesion rates of aged and unaged specimen‑treated HGF. Significance versus control group: #P < 0.01; *P < 0.05
Oxidative Stress
Aged G/Z and Y‑TZP showed a significant rise in ROS at 72 h (P < 0.00001). No significant differences were noted at earlier time points (P > 0.05). G/Z and Y‑TZP produced comparable ROS levels throughout the study, both before and after aging (P > 0.05).
Cell Adhesion
Adhesion rates increased for both G/Z and Y‑TZP after aging (Fig. 2c). No significant difference existed between the two materials pre‑ or post‑aging (P > 0.05). Representative images of cell adhesion on Y‑TZP and G/Z before and after aging are shown in Figure 3.
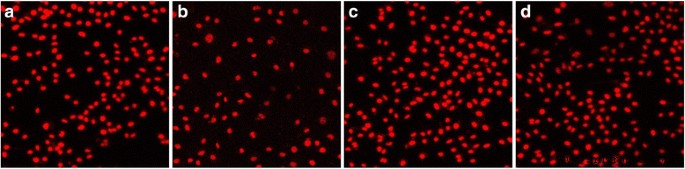
Cell adhesion to G/Z and Y‑TZP before and after aging. a Aged G/Z. b Unaged G/Z. c Aged Y‑TZP. d Unaged Y‑TZP
Cell Morphology
Fluorescence imaging revealed that cells adhered to both aged and unaged G/Z, with enhanced spreading and flattening on aged surfaces (Fig. 4a–c). SEM confirmed well‑spread, polygonal cells with extensive microvilli on both surfaces (Fig. 5a, b).
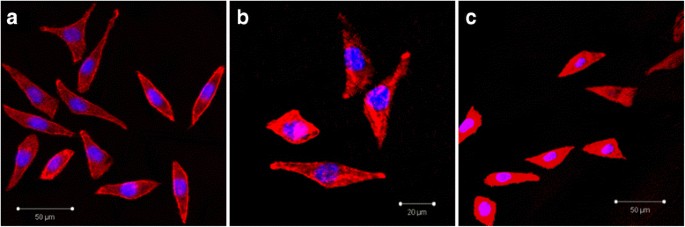
Attachment, spreading, and morphology of HGF on G/Z before and after aging observed with fluorescence microscopy. a, b Aged G/Z. c Unaged G/Z. Cells were cultured for 72 h on substrates and then fixed and stained for filamentous actin (F‑actin, red) and nuclei (blue)
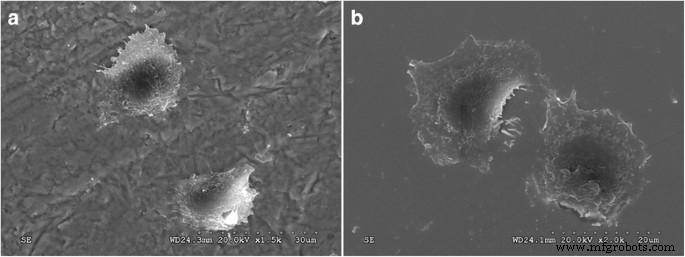
SEM micrographs of the HGF morphology on G/Z before and after aging 72 h post culture. a Aged G/Z. b Unaged G/Z. Original magnification: ×2000
Oral Mucous Membrane Irritation Test
Both macroscopic and microscopic irritation scores were 0 for aged and unaged G/Z, indicating no irritation. Histopathology confirmed the absence of mucosal changes (Fig. 6a, b).
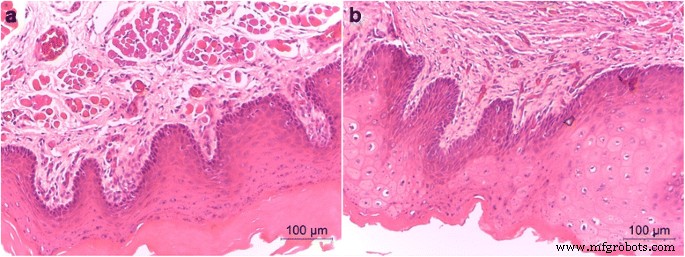
Pathological examination of the mucosa treated with aged G/Z (a) and unaged G/Z (b)
Discussion
Metal‑ceramic alloys have increasingly given way to metal‑free alternatives due to concerns over metal ion release [14–18]. While much research focuses on ceramic improvements, the G/Z system offers a promising route to enhance zirconia’s performance without adding metal components. This study demonstrates that G/Z maintains biocompatibility comparable to Y‑TZP, even after aging, which simulates realistic oral conditions.
Aging diminished cell proliferation at 72 h for both materials, aligning with some literature that reports reduced biocompatibility post‑aging [20] and contrasting others that note improvement [21]. Variations likely stem from differing aging protocols. Our protocol—80 N load, 2 Hz, 10⁵ cycles in artificial saliva at 37 °C—mirrors clinical bite forces and temperature, providing a realistic assessment of aging effects [22].
The observed decline in viability and rise in ROS may reflect ion diffusion and local stress from the t‑m transformation, which can compromise cell integrity [6, 23].
Interestingly, aging increased adhesion for both G/Z and Y‑TZP without altering relative adhesion rates. The roughened, hydrophilic surfaces post‑aging likely enhance protein adsorption, fostering better cell attachment [27–29]. The comparable adhesion suggests G/Z’s surface properties remain favorable after aging.
In vivo irritation testing revealed no histopathological changes, supporting the safety of G/Z for mucosal contact. The modest amount of monoclinic phase introduced during infiltration likely preserves mechanical integrity while keeping the material biocompatible, as shown in prior work on Al₂O₃‑infiltrated Y‑TZP [30].
Other glass‑zirconia composites have shown good biocompatibility and bioactivity in vitro and in vivo [31–33]. Our findings extend this evidence to the graded nano‑glass/zirconia system, offering a material that balances mechanical strength, esthetics, and biological safety.
Conclusions
Biocompatibility assessments—including cell viability, adhesion, morphology, oxidative stress, and mucosal irritation—demonstrate that G/Z performs on par with Y‑TZP before and after aging. These results position G/Z as a promising candidate for dental prostheses. Further comprehensive in vitro and in vivo studies are warranted to confirm these preliminary findings.
Abbreviations
- DAPI:
4′,6‑Diamidino‑2‑phenylindole dihydrochloride
- F‑actin:
Filamentous actin
- FGM:
HAp‑Al₂O₃‑ZrO₂
- G/Z:
Graded nano‑glass/zirconia
- SEM:
Scanning electron microscopy
- t‑m:
Tetragonal to monoclinic
- Y‑TZP:
Yttrium‑stabilized tetragonal zirconia polycrystal
Nanomaterials
- How Extrusion Drives Precision in 3D Printing
- Vespel® Polyimide: The Aerospace Material of Choice
- Materials Used in Boiler Tubes: Choosing the Right Steel, Stainless, Copper and More
- Assessing the Safety of Liposome Nanocarriers Delivered Intratympanically to the Rat Inner Ear
- Gradient Nanomechanical Properties of Fluorosed Enamel: Implications for Restorative Material Selection
- Choosing the Optimal CNC Material: A Guide to Performance, Cost, and Application
- Exploring Material Flexibility: How Elasticity Determines Performance
- Why Material Removal Robots are Essential for Modern Manufacturing
- Palletizing, Packaging, and Part Transfer: The Three Pillars of Material Handling Robotics
- Maintaining Biocompatibility During Material Processing for Medical Applications



