Gold Nanoparticles Boost Electron Transport in PF‑NR2 Cathode Interface for High‑Performance iPLEDs
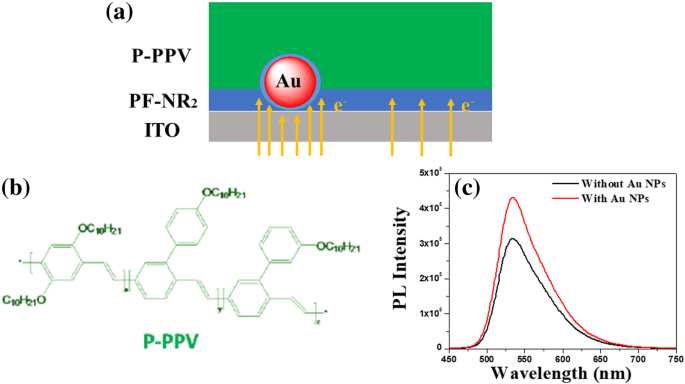
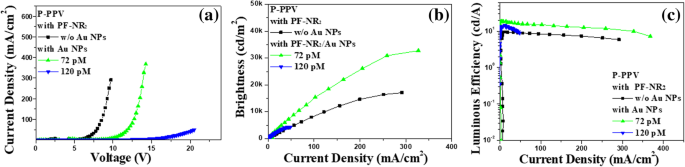
To broaden the commercial impact of organic light‑emitting diodes (OLEDs), solution‑processed inverted polymer LEDs (iPLEDs) are emerging as a promising platform. However, the electron‑transport efficiency of the cathode interface layer—specifically PF‑NR2—remains a bottleneck. In this study, we incorporated 20‑nm gold nanoparticles (Au NPs) into the PF‑NR2 layer at an optimized ratio. Conductive atomic‑force microscopy (c‑AFM) demonstrated a substantial increase in electron conductivity, attributable to the excellent metallic conductivity of Au NPs. Devices fabricated with the Au‑NP‑doped PF‑NR2 layer exhibited a 94 % rise in brightness (from 17 k cd m⁻² to 33 k cd m⁻²) and a 101 % boost in luminous efficiency (from 9.4 cd A⁻¹ to 18.9 cd A⁻¹). These results provide a practical, scalable strategy for enhancing the performance of high‑efficiency iPLEDs. Over the past two decades, OLEDs have attracted significant attention due to their flexibility, lightweight, and cost‑effective fabrication. Solution‑processing offers a route to low‑cost, scalable production, yet commercial deployment of iPLEDs remains limited by sub‑optimal charge‑transport layers. The cathode interface, particularly the PF‑NR2 polymer, is critical for balancing electron and hole injection. Prior work has shown that side‑chain modifications of PF‑NR2 can enhance electron injection, but the intrinsic electron mobility of the polymer remains modest. Metal nanoparticles—especially gold—exhibit exceptional electrical conductivity and plasmonic properties that can be harnessed to improve charge transport in organic layers. This study explores the incorporation of 20‑nm Au NPs into the PF‑NR2 interface layer and evaluates their impact on electron transport and device performance. PF‑NR2 was synthesized via a Pd‑catalyzed Stille coupling of 2,7‑dibromo‑9,9‑bis(3‑(N,N‑dimethylamino)propyl)fluorene with 2,7‑bis(4,4,5,5‑tetramethyl‑1,3,2‑dioxaborolan‑2‑yl)‑9,9‑dioctylfluorene in toluene/Na₂CO₃ under argon. After reflux, the product was precipitated in methanol, filtered, and washed with acetone to remove oligomers, yielding 0.28 g (77 % isolated yield). P‑PPV, ITO glass, and PEDOT:PSS were sourced from commercial suppliers. Gold nanoparticles (20 nm) were synthesized by the Frens citrate reduction method. Zinc acetate dihydrate (1 g) and ethanolamine (0.28 g) were dissolved in 2‑methoxyethanol (10 mL) and stirred for 12 h to form the ZnO precursor solution. HAuCl₄ (0.25 mM) was boiled under vigorous stirring, followed by the addition of 1 mL 5 % trisodium citrate. The reaction proceeded for 15 min until a wine‑red color appeared, indicating 20‑nm Au NPs. ZnO precursor was spin‑coated on ITO at 4000 rpm and annealed at 200 °C for 1 h, yielding ~30 nm films. The PF‑NR2 interlayer (2 mg mL⁻¹ in methanol + 10 µL mL⁻¹ acetic acid) was spin‑coated at 2000 rpm, forming a ~5 nm layer. P‑PPV (6 mg mL⁻¹) was spin‑coated at 1400 rpm to give ~80 nm. After evacuation to 3 × 10⁻⁴ Pa, 10 nm MoO₃ and 120 nm Al were thermally evaporated, defining a 16 mm² pixel area. All steps, except ZnO deposition, were performed in a nitrogen glovebox (<10 ppm O₂/H₂O). c‑AFM was performed with a 3 N m⁻¹ Pt/Ir‑coated silicon cantilever, setpoint 1 V, and current amplification (10⁷ V A⁻¹). I‑V‑B curves were measured in a glovebox using a Keithley 236 SMU and a calibrated silicon photodiode. UV‑Vis spectra (Shimadzu UV‑3600), film thickness (Dektak 150), and AFM (Seiko SPA 400) were also recorded. Transmission electron microscopy confirmed monodisperse 20‑nm Au NPs (Fig. 1a). Their LSPR peak at 520 nm (Fig. 1b) validates the nanoparticle size. The PF‑NR2/ Au NP blend (PF‑NR2/30 pM Au NPs) was spin‑coated to produce ~5 nm thick films; absorbance measurements (Lambert‑Beer law) corroborated the thickness (Fig. 2b). a TEM image. b Absorption spectra of Au NPs a Molecular structure of PF‑NR2. b Thickness variation of PF‑NR2 under different fabrication conditions measured by UV‑Vis spectroscopy c‑AFM I‑V mapping revealed a pronounced increase in current density for PF‑NR2 doped with 36 pM Au NPs, confirming enhanced electron injection. At 120 pM, conductivity decreased due to nanoparticle aggregation, as shown by SEM (Additional file 1: Fig. S1). The optimal Au NP loading (36 pM) provided the highest electron transport without compromising film morphology (RMS roughness increased from 0.56 nm to 1.59 nm; Fig. 3). PF‑NR2 AFM surface morphology a, b height images without and with Au NPs and c phase image with Au NPs (scan area 1.0 µm × 1.0 µm) a Schematic of c‑AFM testing. b, c I‑V characteristics near a single Au NP and the depiction of the height of a single Au NP in PF‑NR2 layer. The locations of the colored numbers in the inset image correspond to the color of the I‑V curve The electron‑only devices ITO/ZnO (30 nm)/PF‑NR2 (5 nm, with and without Au NPs)/P‑PPV (80 nm)/CsF (1.5 nm)/Al (120 nm) a Schematics of the proposed enhanced electron transportation of the hybrid layer with an inverted structure. b Molecular structure of PF‑NR2. c PL spectroscopy of P‑PPV with and without Au NPs Photoluminescence measurements (Fig. 6c) confirmed that Au NPs, confined within the PF‑NR2 layer, do not quench the emission of P‑PPV, ensuring that the observed performance gains stem from improved charge transport rather than optical effects. a Current density vs. applied voltage (I‑V). b Brightness vs. current density (B‑I), and c Luminous efficiency vs. current density (LE‑I) curves under different conditions when P‑PPV was used as emitting layer in iPLEDs, respectively Integrating the Au‑NP‑doped PF‑NR2 layer into a full iPLED stack yielded a 94 % increase in brightness (from 17 k cd m⁻² to 33 k cd m⁻²) and a 101 % improvement in luminous efficiency (from 9.4 cd A⁻¹ to 18.9 cd A⁻¹). These gains demonstrate that Au NPs provide an effective, scalable route to enhance electron injection and overall device efficiency. We successfully incorporated 20‑nm Au NPs into the PF‑NR2 cathode interface layer, achieving a two‑fold enhancement in electron transport without compromising optical performance. The Au‑NP‑doped PF‑NR2 films exhibited superior electron injection, leading to a 94 % brightness increase and a 101 % rise in luminous efficiency in iPLEDs. This straightforward, scalable doping strategy offers a practical path toward high‑efficiency, solution‑processed OLEDs. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. Atomic force microscopy Gold nanoparticles Brightness vs. current density Conductive atomic force microscopy Inverted polymer light‑emitting diode Indium tin oxide Current density vs. applied voltage Current density–voltage–brightness Luminous efficiency vs. current density Local surface plasmon resonance Organic light‑emitting diode Poly(3‑hexylthiophene): indene‑C₆₀ bisadduct Poly(3,4‑ethylenedioxythiophene):poly(styrene‑sulfonate) Poly[(9,9‑bis(3′-(N,N‑dimethylamino)propyl)-2,7‑fluorene)-alt‑2,7‑(9,9‑dioctylfluorene)] Photoluminescence Polymer light‑emitting diode Polymer‑poly(2-(4-(3′,7′‑dimethyloctyloxyphenyl)-1,4‑phenylene‑vinylene) Transmission electron microscopeAbstract
Introduction
Materials and Methods
Materials
Preparation of the Zinc Oxide (ZnO) Precursor
Synthesis of Au NPs
iPLED Device Fabrication
Characterization of Devices and Thin Films
Conductive Atomic Force Microscopy
Results and Discussion
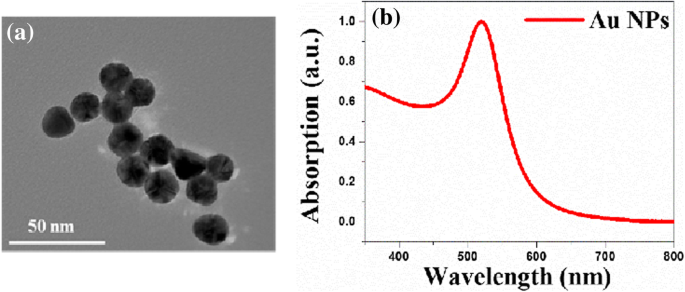
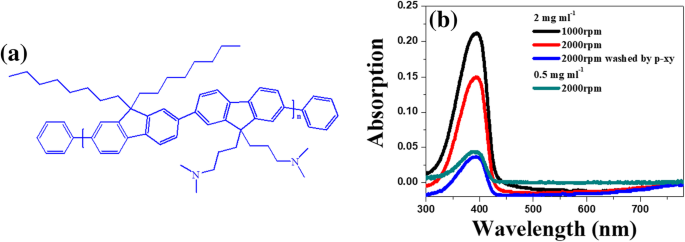
Characterization of Au NPs and PF‑NR2 Film
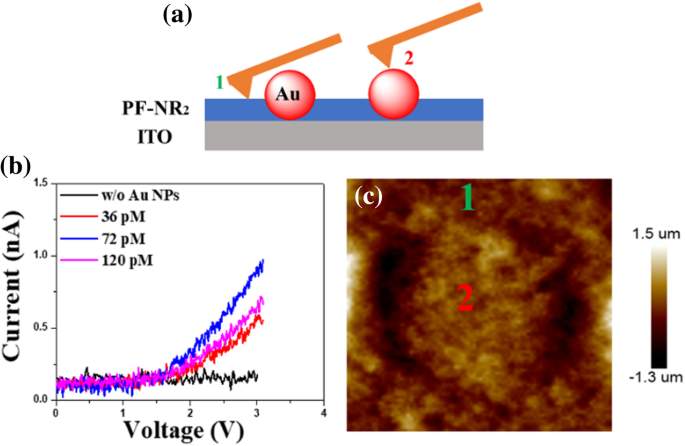
c‑AFM Characterization of PF‑NR2 Thin Films

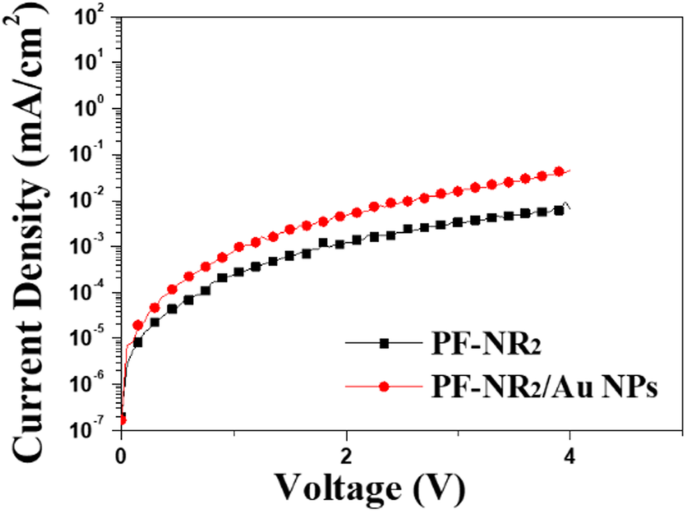
Conclusions
Availability of Data and Materials
Abbreviations
Nanomaterials
- Understanding Electron Tubes: Components and Functionality
- Plasmonic Nanoparticles: Harnessing Surface Plasmons for Advanced Photothermal Applications
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Optimized Al₂O₃/MgO Emission Layers for Ultra‑High Gain Electron Multipliers
- Optimizing Sb-Loaded Palygorskite Nanoparticles for Superior Catalytic Hydrogenation of p-Nitrophenol
- GoldMag: PSS‑MA Coating Enhances Stability & Optical Precision for Protein Detection
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Enhanced Gene Delivery Using Gold Nanoparticles Functionalized with Chitosan, N‑Acylated Chitosan, and Chitosan Oligosaccharide
- Boosting AlGaN Deep UV LED Efficiency with Chirped Superlattice Electron Deceleration Layers
- Plasmon‑Enhanced Fluorescence of Rhodamine 6G at Controlled Distances from Gold Nanoparticles



