Pd‑Decorated Few‑Layer MoS₂/SiO₂/Si Heterojunctions Deliver Record Room‑Temperature Hydrogen Detection
Abstract
We report a scalable fabrication of wafer‑scale, few‑layer MoS₂/SiO₂/Si heterojunctions by DC magnetron sputtering, followed by in‑situ decoration with Pd nanoparticles. The resulting devices exhibit a dramatic boost in hydrogen response at room temperature, achieving an outstanding sensitivity of 9.2 × 10³ % to H₂, far exceeding that of either Pd/SiO₂/Si or MoS₂/SiO₂/Si alone. The sensing performance is strongly governed by the Pd layer thickness, with an optimal thickness around 5 nm. Microstructural analysis and electrical measurements reveal the underlying mechanisms, confirming that Pd catalysis and interfacial charge transfer are responsible for the enhanced response. These findings establish Pd‑decorated MoS₂/SiO₂/Si heterojunctions as a promising platform for high‑performance, room‑temperature hydrogen sensors.
Background
Hydrogen (H₂) is a clean, abundant energy carrier widely used in fuel cells. Its tasteless, colorless, and highly explosive nature, however, poses significant safety risks, underscoring the need for reliable, real‑time H₂ leak detection [1]. Conventional metal‑oxide sensors can detect H₂, but they typically operate at ~150 °C, raising safety concerns and limiting practical deployment. A key research direction is thus the development of novel sensing materials capable of operating at room temperature (RT).
Transition‑metal dichalcogenide MoS₂ has emerged as a compelling two‑dimensional material, offering a high surface‑to‑volume ratio, tunable bandgap, and excellent charge transport [6–10]. While mechanical exfoliation yields high‑quality flakes, it is unsuitable for large‑area device fabrication. Chemical vapor deposition produces wafer‑scale films but requires 800–1000 °C, which can degrade sulfur stoichiometry and cause interfacial diffusion. Physical vapor deposition, especially DC magnetron sputtering, has proven capable of depositing few‑layer MoS₂ at temperatures as low as 300 °C, delivering high‑mobility (≈181 cm² V⁻¹ s⁻¹) and a current on/off ratio of ~10⁴ [20].
MoS₂ films have shown sensitivity to polar gases such as NH₃, NO, and NO₂, thanks to facile charge transfer at the MoS₂ surface. However, H₂ is a nonpolar molecule, and pristine MoS₂ responds poorly to it. Decorating MoS₂ with palladium nanoparticles enhances H₂ sensitivity through catalytic hydrogenation, forming PdHₓ and releasing electrons into MoS₂ [31, 32]. Prior Pd‑decorated MoS₂ sensors, however, have reported modest sensitivities. In earlier work, we integrated MoS₂ films with Si to form MoS₂/SiO₂/Si heterojunctions, achieving ~10⁴ % sensitivity but with sluggish response times (~443 s) due to hydrogen diffusion limitations in thick films. There is thus a clear need for a design that combines the high surface area of few‑layer MoS₂ with efficient charge transfer and rapid kinetics.
Here we present a novel architecture: few‑layer MoS₂ films grown on SiO₂/Si by DC sputtering, subsequently decorated with Pd nanoparticles. This configuration yields a highly responsive, fast‑recovery H₂ sensor that operates at RT, with performance tuned by Pd thickness. The sensing mechanism is elucidated through detailed microstructural and electrical analyses, and energy‑band alignment is constructed to rationalize the observed behavior.
Methods
Few‑layer MoS₂ films were deposited on (100)‑oriented n‑type Si wafers (resistivity 1–6 Ω cm) by DC magnetron sputtering from a 99 % pure MoS₂ target. The substrates were diced into 12.5 mm × 12.5 mm pieces, sequentially cleaned in acetone, isopropanol, and de‑ionized water, then etched in 5 % HF for 60 s to remove native oxide. A 40 % H₂O₂ treatment at 100 °C for 20 min formed a ~300 nm SiO₂ passivation layer, which both smoothens the surface for 2‑D growth and suppresses MoS₂–Si interdiffusion. MoS₂ deposition proceeded at 450 °C under 1.0 Pa Ar and 10 W power. After growth, Pd layers of 1.0–30.0 nm were sputtered in situ at RT, 3.0 Pa, 10 W. Indium pads (300 µm thick, 0.5 mm diameter) served as electrodes on the Pd‑decorated MoS₂ side and the Si backside.
Structural characterization employed Raman spectroscopy (488 nm excitation), atomic force microscopy (AFM), X‑ray photoelectron spectroscopy (Kratos Axis ULTRA, Al Kα 1486.6 eV), high‑resolution TEM (JEOL JEM‑2100F), UV‑vis spectroscopy (Shimadzu UV‑3150), and UPS (He‑I 21.22 eV). Electrical measurements were carried out in a sealed chamber using a Keithley 2400 source meter; sensors were exposed to H₂ concentrations (0.5–5.0 %) in dry air at RT. Recovery was achieved by purging with ambient air.
Results and Discussion
XPS confirms the successful synthesis of MoS₂ with negligible contamination. The survey spectrum (Fig. 1a) shows Mo, S, Pd, and O peaks, while high‑resolution scans reveal S 2p (162.8/163.9 eV), Mo 3d (229.9/233.1 eV), and Pd 3d (335.5/340.9 eV) binding energies consistent with stoichiometric MoS₂ and metallic Pd, indicating no significant Pd‑Mo substitution (Fig. 1b–d).
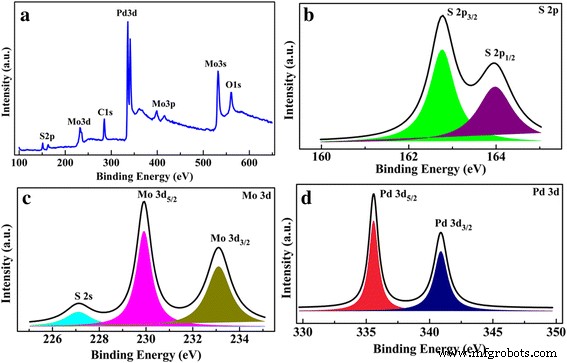
a XPS survey, b S 2p, c Mo 3d, and d Pd 3d core‑level spectra of the 5‑nm Pd‑decorated MoS₂ layers on Si substrate
AFM images (Fig. 2a) show a smooth MoS₂ surface (RMS ≈ 0.78 nm). After 5‑nm Pd deposition, nanoparticle islands appear (Fig. 2b) with an average diameter of 47.7 nm and a modest RMS increase to 0.89 nm. TEM cross‑section (Fig. 2c) reveals a ~7.2 nm gap in the Pd layer, confirming its discontinuous, island‑like nature. The MoS₂ film exhibits 2–3 atomic S–Mo–S layers with an interlayer spacing of ~0.65 nm (Fig. 2d). Raman spectra (Fig. 2e) consistently display E¹₂g (≈381.9 cm⁻¹) and A₁g (≈405.1 cm⁻¹) peaks across four regions; the 23.2 cm⁻¹ separation confirms few‑layer MoS₂.
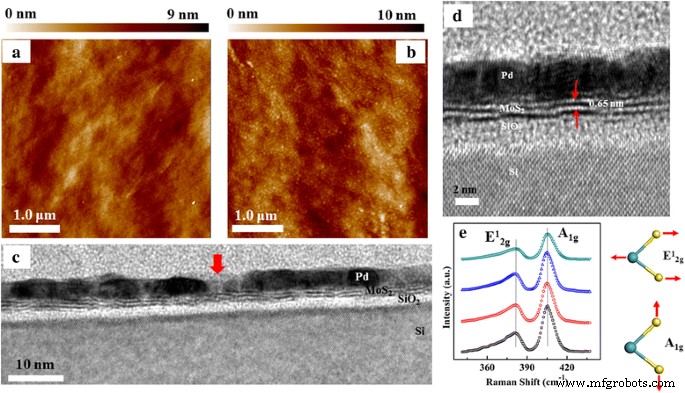
AFM images of the MoS₂ layers a without Pd decoration, and b with 5‑nm Pd decoration. c HRTEM image of the Pd‑decorated MoS₂ layers on the Si substrate. The red arrow denotes the gap between two Pd nanoparticles. d Enlarged HRTEM image. e Typical Raman spectra of the as‑grown MoS₂ layers on Si from different regions of the sample
Temperature‑dependent resistivity (ρ–T) measurements (Fig. 3) show semiconductor behavior for MoS₂ and the 5‑nm Pd layer, while the 10‑nm Pd layer displays metallic conductivity, indicating a percolation threshold near 5–10 nm. Pd‑decorated MoS₂ retains semiconducting characteristics but with a dramatically reduced resistivity (~1.1 Ω cm) due to effective Pd–MoS₂ interfacial coupling. The resistivity decreases sharply with Pd thickness above 5 nm, reflecting enhanced Pd nanoparticle coverage.
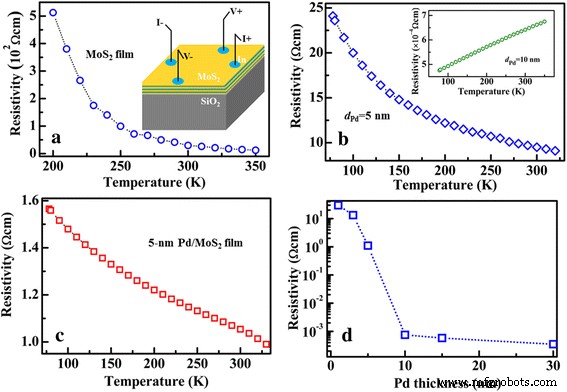
Resistivity–temperature curve of different samples grown on 300‑nm SiO₂/Si substrate. a Few‑layer MoS₂. The inset shows the schematic illustration for the measurements. b 5‑nm Pd layer. The inset shows the ρ–T curve for the 10‑nm Pd layer. c 5‑nm Pd‑decorated MoS₂ layers. d Resistivity of the Pd‑decorated MoS₂ layers as a function of Pd thickness
Electrical characterization of the Pd‑decorated MoS₂/SiO₂/Si junction (Fig. 4a) reveals pronounced rectification, confirming a p–n heterojunction. UPS measurements (Fig. 4b) yield a MoS₂ work function of 5.53 eV and a valence‑band offset of 0.48 eV. Optical absorption (Fig. 4c) indicates a bandgap of 1.48 eV, while Hall measurements confirm p‑type behavior (hole density 4.38 × 10¹⁵ cm⁻³, mobility 11.3 cm² V⁻¹ s⁻¹). Energy‑band diagrams (Fig. 4d) illustrate electron transfer from n‑Si to MoS₂, establishing a built‑in field (V_bi) directed from Si to MoS₂, responsible for the observed I–V asymmetry.
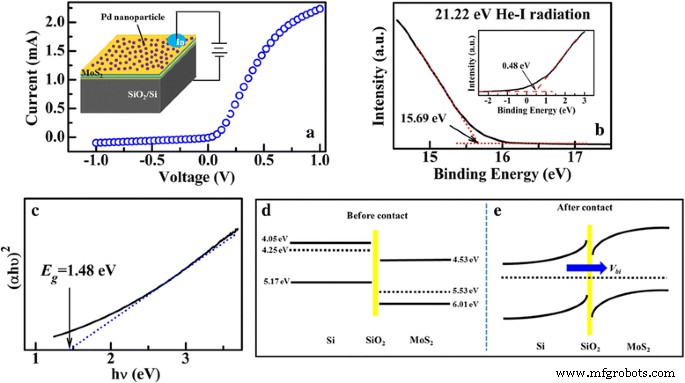
a I–V characteristics of Pd‑decorated MoS₂/Si heterojunction. The inset shows the schematic illustration for the measurements. b UPS spectra of the MoS₂ layers on Si substrate. c Curve of (αhν)² vs hν of the MoS₂ layers. The energy band diagram at the MoS₂/Si interface before contact (d) and after contact (e)
Hydrogen sensing performance (Fig. 5a) demonstrates a remarkable sensitivity of 9.2 × 10³ % at −1.0 V bias, surpassing single‑layer MoS₂ or Pd‑decorated MoS₂ devices by an order of magnitude. The sensor responds within 10.7 s and recovers in 8.3 s, outperforming many RT H₂ sensors [2–5]. The response originates from Pd‑mediated hydrogenation forming PdHₓ, injecting electrons into MoS₂, reducing the p‑type carrier concentration, shifting the Fermi level toward the conduction band, and lowering the built‑in barrier. Under positive bias, the effect is muted due to electron injection from Si dominating the channel conductivity. Repeated exposure cycles confirm reproducibility and stability.
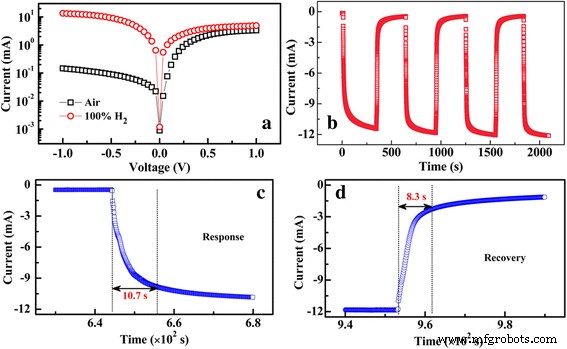
a Log I–V curves of the Pd‑decorated MoS₂/Si heterojunction in air and pure H₂ at RT. b I–t graphs of the Pd‑decorated MoS₂/Si heterojunction exposed to pure H₂ at −1.0 V and RT. c, d Enlarged response and recovery edges, respectively, of the sensing curve. The response time (t_res) is the interval for the current to rise from 10 % to 90 % of the total change. The recovery time (t_rec) is the interval for the current to decay from 90 % to 10 % of the total change
Dynamic response to varying H₂ concentrations (Fig. 6a) shows linear sensitivity from 0.5 % to 5.0 %, with sensitivities ranging from 5.7 × 10² % to 4.3 × 10³ %. Both t_res and t_rec increase as H₂ concentration decreases (Fig. 6b), reflecting slower adsorption kinetics at lower partial pressures. The linearity suggests that the sensor can be calibrated for quantitative H₂ detection.
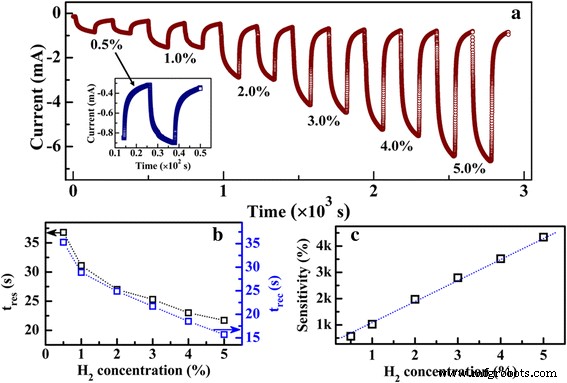
a Dynamic responses of the Pd‑decorated MoS₂/Si heterojunction upon consequent H₂ at varying concentrations from 0.5 to 5 % at −1.0 V. The inset shows the enlarged image of the sensing characteristics of the heterojunction under H₂ of 0.5 %. b Dependence of t_res and t_rec on H₂ concentration. c Dependence of the sensing response of the heterojunction on H₂ concentration
Pd thickness critically influences sensing performance (Fig. 7). Sensors with 1.0 nm Pd show modest sensitivity (~120 %) and slow response (~58 s) due to sparse nanoparticle coverage. As Pd thickness increases, coverage improves, electron transfer upon H₂ exposure is enhanced, and sensitivity rises, peaking at 5.0 nm with 9.2 × 10³ % sensitivity and 10.7 s response. Further increase to 30 nm results in a continuous Pd film, reducing nanoparticle density and thus lowering sensitivity to 1.5 × 10³ %. Additionally, thicker Pd layers exhibit charge accumulation, slowing response times. Therefore, 5.0 nm represents the optimal Pd thickness for maximum sensitivity and fastest kinetics.
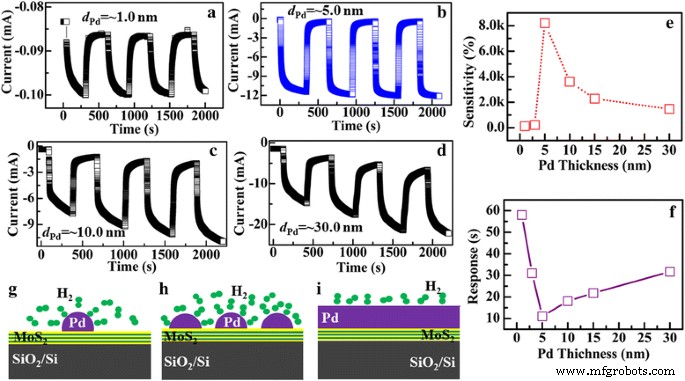
Sensing characteristics of the Pd‑decorated MoS₂/Si heterojunction with different Pd thickness, respectively. a d_Pd ≈ 1.0 nm, b d_Pd ≈ 5.0 nm, c d_Pd ≈ 10.0 nm, and d d_Pd ≈ 30.0 nm. e, f Dependence of the sensitivity and response time of the heterojunctions on Pd thickness, respectively. g–i Schematic illustration of the reaction of H₂ on the Pd‑decorated MoS₂/Si heterojunction with different Pd thickness
Conclusions
We have demonstrated that few‑layer MoS₂ films grown by DC sputtering and decorated with 5‑nm Pd nanoparticles form a robust p–n heterojunction on SiO₂/Si that delivers an unprecedented H₂ sensitivity of 9.2 × 10³ % at RT, with a rapid response of 10.7 s and recovery of 8.3 s. The performance is highly sensitive to Pd thickness, with ~5 nm providing the optimal nanoparticle density for maximal catalytic activity and charge transfer. These results confirm that sputtered, Pd‑decorated MoS₂ combined with conventional Si substrates offers a scalable route to high‑performance, room‑temperature H₂ sensors.
Abbreviations
- AFM:
Atomic force microscope
- dPd:
Thickness of the Pd layer
- EC:
Conduction band level
- EF:
Fermi energy level
- Eg:
Energy band gap
- Ep:
Distance between EV and EF
- EV:
Valence band level
- HRTEM:
High‑resolution transmission electron microscopy
- MoS₂:
Molybdenum disulfide
- RMS:
Root‑mean‑square roughness
- trec:
Recovery time for the sensor
- tres:
Response time for the sensor
- UPS:
Ultraviolet photoelectron spectroscopy
- Vbi:
Built‑in electrical field
- W:
Work function
- XPS:
X‑ray photoemission spectroscopy
Nanomaterials
- Controlling Photoresponsivity in Multi‑Layer MoS₂ Phototransistors Through Applied Bias
- Efficient Humate-Driven Co-Precipitation/Calcination Synthesis of MoS₂/C Nanocomposites for Superior Lithium‑Ion Battery Performance
- High‑Performance Refractive‑Index Sensing with Au/SiO₂ Triangle Arrays on Reflective Gold Substrates
- Optimizing Silver Nanoparticle Dispersion on PSA Microspheres via Carboxyl Group Dissociation: Enhanced Catalytic Performance
- High‑Performance Quasi‑Solid‑State Asymmetric Supercapacitors Based on MoS₂/MWCNT and PANI/MWCNT Composite Electrodes
- MoS₂/SiO₂ Composite Saturable Absorber Enables Sub‑Nanosecond Mode‑Locked Er‑Doped Fiber Laser
- Polarization-Dependent Reflection Spectra of Single-Layer MoS₂ and ReS₂ on SiO₂/Si and Quartz
- R‑Scheme Ti3C2 MXene/MoS2 2D/2D Heterojunctions: A Breakthrough in Photocatalytic Efficiency
- Hierarchically Porous MoS₂/C Nanospheres from Nanosheets: Exceptional Electrochemical Energy Storage
- Copper Nanoparticles on Porous Carbon Substrates Deliver High-Performance Glucose Sensors



