High‑Performance Quasi‑Solid‑State Asymmetric Supercapacitors Based on MoS₂/MWCNT and PANI/MWCNT Composite Electrodes
Abstract
In this study, we report the synthesis of MoS₂ and polyaniline (PANI) electrodes decorated with multi‑walled carbon nanotubes (MWCNTs) via a facile hydrothermal route and in‑situ polymerization, respectively. The resulting MoS₂|MWCNTs electrode with a 1:1 molar ratio delivers a specific capacitance of 255.8 F g⁻¹ at 1 A g⁻¹, while exhibiting a low internal resistance and 91.6 % capacitance retention after 1,000 cycles. The PANI|MWCNTs electrode achieves 267.5 F g⁻¹ at the same current density and retains 97.9 % after 1,000 cycles. When assembled into a quasi‑solid‑state asymmetric supercapacitor (ASC) using a PVA‑Na₂SO₄ gel electrolyte, the device delivers a specific capacitance of 138.1 F g⁻¹ at 1 A g⁻¹, an energy density of 15.09 Wh kg⁻¹ at 2,217.95 W kg⁻¹, and 38.9 Wh kg⁻¹ at 382.61 W kg⁻¹. These results demonstrate that the MoS₂|MWCNTs/PANI|MWCNTs ASC offers a compelling combination of high energy density, power output, and cycling stability, underscoring its potential for next‑generation energy storage applications.
Background
The growing demand for sustainable energy solutions has spotlighted the need for reliable, high‑performance storage devices capable of handling intermittent renewable sources such as solar and wind power. Supercapacitors (SCs), known for their rapid charge–discharge capabilities and extended cycle life, are ideal candidates to bridge this gap. Their performance, however, hinges on the quality of electrode materials. Among the plethora of candidates—graphene, carbon nanotubes, conducting polymers, and metal oxides—transition‑metal dichalcogenides (TMDs) like MoS₂ and conducting polymers such as polyaniline (PANI) have emerged as front‑line contenders due to their pseudocapacitive behavior and high theoretical capacitances.
MoS₂ offers superior ionic conductivity and a higher theoretical capacity than conventional graphite, yet its two‑dimensional layers tend to restack during cycling, limiting usable surface area. Introducing a conductive, high‑surface‑area scaffold such as MWCNTs can mitigate this issue, preventing aggregation and providing continuous electron pathways. Meanwhile, PANI provides a high specific capacitance (500–2,200 F g⁻¹) and tunable redox properties, but suffers from poor conductivity and cycling stability unless combined with conductive carbon frameworks.
Recent advances in quasi‑solid‑state electrolytes, particularly polyvinyl alcohol (PVA)‑based gels, have further enhanced the practicality of SCs by eliminating leakage risks and enabling flexible device architectures. Building on these developments, we fabricated MoS₂|MWCNTs and PANI|MWCNTs composites, evaluated their electrochemical behavior, and integrated them into a quasi‑solid‑state ASC to assess energy and power performance.
Methods
Synthesis of MoS₂|MWCNTs
MoS₂ was prepared by a one‑step hydrothermal approach. Sodium molybdate (0.726 g) and thiourea (0.684 g) were dissolved in 35 mL deionized water, stirred and sonicated for 30 min. MWCNTs (varying loadings) were added and sonicated another 30 min. The mixture was acidified to pH < 1 with 12 M HCl, then transferred to a 50 mL Teflon‑lined autoclave and heated at 200 °C for 24 h. After cooling, the precipitate was filtered, washed with ethanol and water, and dried at 60 °C under vacuum. Three molar ratios—MoS₂:MWCNTs of 2:1, 1:1, and 1:2—were examined, labeled MS2MWCNT1, MS1MWCNT1, and MS1MWCNT2. Pure MoS₂ and MWCNTs were denoted MS1MWCNT0 and MS0MWCNT1, respectively.
Synthesis of PANI|MWCNTs
PANI|MWCNTs were produced by in‑situ oxidative polymerization. 18 mg MWCNTs were dispersed in 10 mL water and sonicated for 30 min (solution A). 0.3 mL aniline monomer was dissolved in 10 mL 1 M HCl (solution B). Potassium persulfate (0.21 g) was dissolved in 10 mL 1 M HCl (solution C). Solution B was added to A under stirring, followed by slow dropwise addition of C until the mixture turned dark green. The reaction proceeded at room temperature for >5 h, then the product was collected by centrifugation, washed with water and ethanol, and labeled PANI|MWCNTs.
Preparation of Quasi‑Solid‑State ASC
The ASC was constructed as a sandwich cell: a MoS₂|MWCNTs cathode and a PANI|MWCNTs anode separated by a PVA‑Na₂SO₄ gel. Electrode slurries (75 % active material, 15 % nano‑graphite, 10 % PVDF) in NMP were cast on foam nickel via a doctor blade, dried at 60 °C for 12 h, then soaked in 0.5 M Na₂SO₄ for 12 h. The gel electrolyte was prepared by dissolving 2 g PVA in water at 90 °C, then adding 0.5 M Na₂SO₄ under vigorous stirring until clear. The gel was cooled and used to fill the cell.
Characterization and Electrochemical Measurement
SEM (JSM‑7001F) provided surface morphology. XRD (X‘Pert Pro) and Raman (Renishaw RW1000) characterized crystallinity. Electrochemical tests were performed in a three‑electrode setup: Pt counter, Ag/AgCl reference, and 6 M KOH electrolyte. CV, GCD, and EIS (CHI660E) were carried out at 20 °C, 20 mV AC amplitude, 0.1–10⁵ Hz. Specific capacitance was calculated using Cₛ = 4Δt/(ΔV m_ac).
Results and Discussion
Cathode Material: MoS₂|MWCNTs
SEM images (Fig. 1) reveal uniform honeycomb‑structured MoS₂ nanospheres (~200 nm) bridged by MWCNTs in the MS1MWCNT1 composite. The MWCNT network enhances electrical conductivity and provides additional active sites, mitigating the restacking tendency of MoS₂ sheets.

SEM images of MoS₂ nanospheres (a, b) and the MS1MWCNT1 composite (c, d).
Raman spectra (Fig. 2a) show characteristic MoS₂ peaks at 375 cm⁻¹ (E₂g) and 408 cm⁻¹ (A₁g). MWCNTs display D and G bands at 1350 cm⁻¹ and 1580 cm⁻¹, respectively. The composite retains both sets of peaks with a slight red shift, indicating intimate mixing without new phase formation. XRD patterns (Fig. 2b) confirm the (002) peak at 14.46°, evidencing well‑ordered layered MoS₂ within the composite.
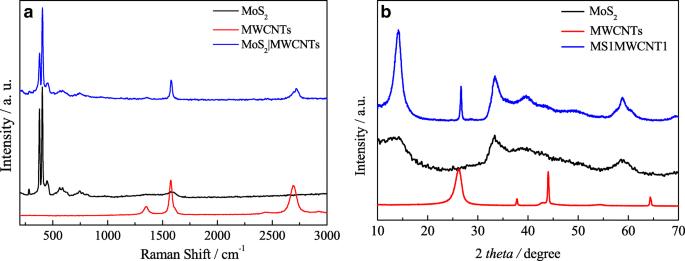
Raman spectrum (a) and XRD patterns (b) of MWCNTs, MoS₂, and MS1MWCNT1.
Electrochemical testing (Fig. 3) shows that the MS1MWCNT1 electrode achieves the highest specific capacitance of 255.8 F g⁻¹ at 1 A g⁻¹, outperforming pure MoS₂ (90.6 F g⁻¹) and MWCNTs (30.4 F g⁻¹). The 1:1 molar ratio optimizes the synergistic interaction between MoS₂’s pseudocapacitance and MWCNTs’ conductivity. At 0.5 A g⁻¹, the capacitance rises to 266.9 F g⁻¹, while at 5 A g⁻¹ it remains 203.5 F g⁻¹, demonstrating excellent rate capability. EIS (Fig. 3e) reveals a charge‑transfer resistance of 0.59 Ω, the lowest among the compared materials, confirming efficient electron transport. After 1,000 charge–discharge cycles, the capacitance retention is 91.6 %, indicating robust cycling stability.
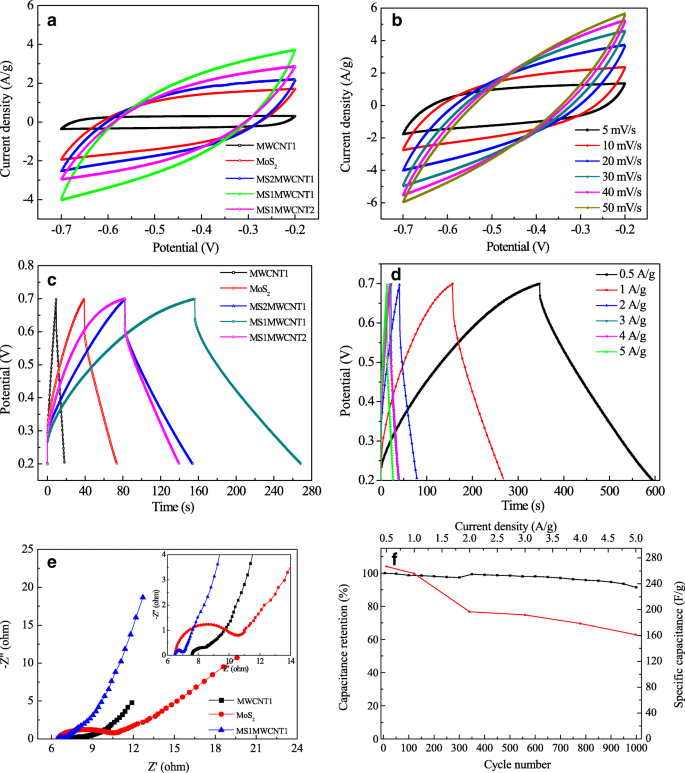
CV curves for MoS₂|MWCNTs electrodes with different molar ratios (a); CV curves of MS1MWCNT1 at varying scan rates (b); GCD curves at 1 A g⁻¹ (c); GCD at different current densities (d); EIS (e); stability after 1,000 cycles (f).
Anode Material: PANI|MWCNTs Composite
SEM images (Fig. 4) show PANI uniformly coating the MWCNT framework, forming a robust hybrid that combines PANI’s redox activity with MWCNTs’ conductivity. The MWCNT skeleton prevents PANI from dissolving and enhances ion diffusion.

SEM images of PANI|MWCNTs (a, b).
Raman spectra (Fig. 5) display PANI peaks at 1165, 1308–1347, 1468, and 1593 cm⁻¹, confirming successful polymerization. The composite retains both PANI and MWCNT signatures, indicating intimate contact.
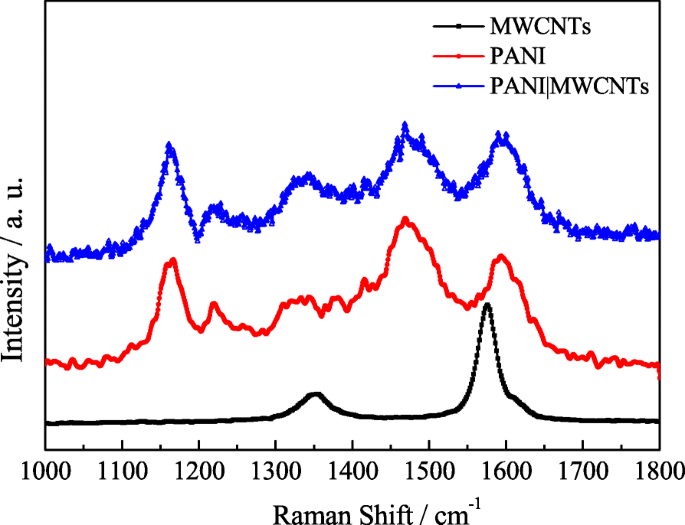
Raman spectrum of MWCNTs, PANI, and PANI|MWCNTs.
Electrochemical evaluation (Fig. 6) shows a broadened voltage window for the composite relative to the individual components, reflecting enhanced electronic pathways. At 1 A g⁻¹, the composite delivers 267.5 F g⁻¹, with capacitance decreasing to 150.8 F g⁻¹ at 5 A g⁻¹. EIS indicates a lower charge‑transfer resistance (≈0.6 Ω) compared to PANI (≈2.5 Ω) and MWCNTs (≈1.0 Ω). Cycling tests demonstrate 97.9 % capacitance retention after 1,000 cycles, a testament to the structural stability conferred by the MWCNT network.
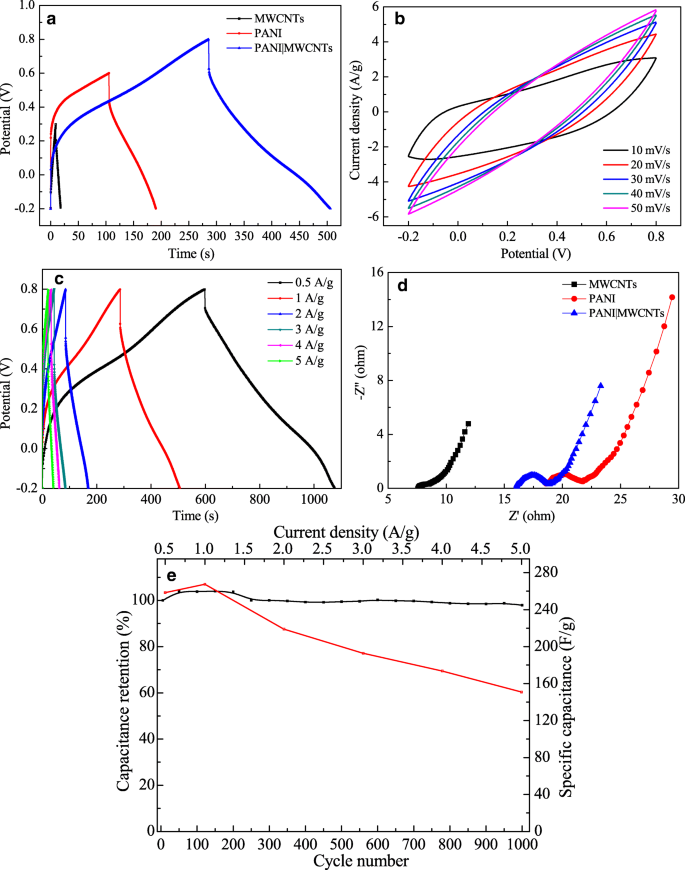
GCD curves of PANI, MWCNTs, and PANI|MWCNTs at 1 A g⁻¹ (a); CV curves at varying scan rates (b); GCD at different current densities (c); EIS (d); stability after 1,000 cycles (e).
Quasi‑Solid‑State ASC: MM//PM
The assembled ASC (Fig. 7a) operates up to 1.5 V while maintaining quasi‑rectangular CV profiles, indicative of stable charge storage. GCD tests (Fig. 7b) confirm high coulombic efficiency across 0.5–4 A g⁻¹. EIS (Fig. 7c) shows a low solution resistance (20.86 Ω) and a charge‑transfer resistance of 15.49 Ω, attributed to the MWCNT network facilitating ion/electron transport. After 1,000 cycles, the device retains 65.2 % of its initial capacitance (Fig. 7d), a satisfactory figure for quasi‑solid‑state configurations.
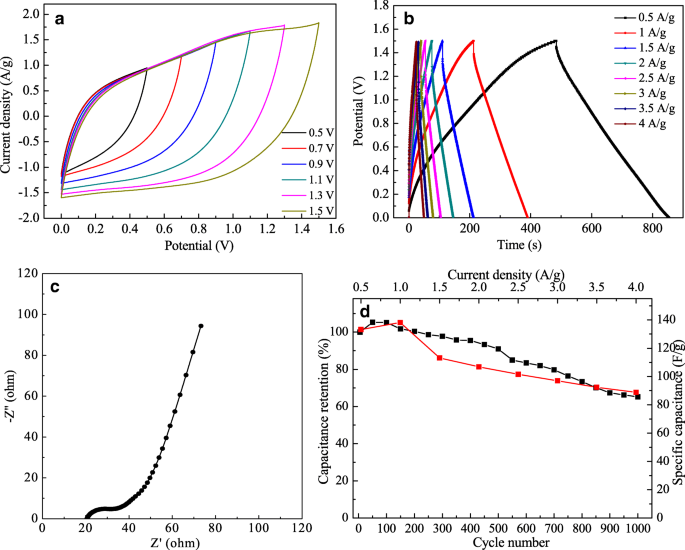
CVs at different voltage windows (a); GCD at various current densities (b); EIS (c); stability after 1,000 cycles (d).
The Ragone plot (Fig. 8a) demonstrates an energy density of 38.9 Wh kg⁻¹ at 382.61 W kg⁻¹ and 15.09 Wh kg⁻¹ at 2,217.95 W kg⁻¹, confirming the ASC’s suitability for high‑power applications. A practical demonstration (Fig. 8b) shows two ASC cells in series lighting a red LED, underscoring real‑world viability.
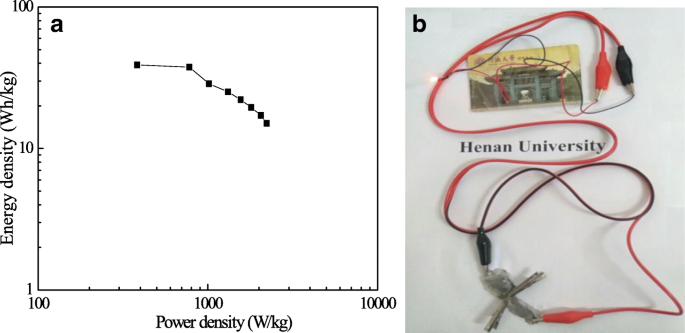
Energy density vs. power density (a); LED illumination demonstration (b).
Conclusion
We have successfully fabricated MoS₂|MWCNTs and PANI|MWCNTs composites via hydrothermal and in‑situ polymerization routes, achieving specific capacitances of 255.8 F g⁻¹ and 267.5 F g⁻¹ at 1 A g⁻¹, respectively. When assembled into a quasi‑solid‑state ASC with a PVA‑Na₂SO₄ gel, the device delivers a specific capacitance of 138.1 F g⁻¹ at 1 A g⁻¹, an energy density of 38.9 Wh kg⁻¹ at 382.61 W kg⁻¹, and retains 15.09 Wh kg⁻¹ at 2,217.95 W kg⁻¹. The ASC also demonstrates 65.2 % capacitance retention after 1,000 cycles and can light a red LED when two cells are connected in series. These findings confirm that the MoS₂|MWCNTs/PANI|MWCNTs architecture offers a highly efficient, durable, and practical platform for next‑generation energy storage devices.
Abbreviations
- ASC:
Asymmetric supercapacitor
- Cs:
Specific capacitance
- CV:
Cyclic voltammetry
- E:
Energy density
- EIS:
Electrochemical impedance spectroscopy
- GCD:
Galvanostatic charge‑discharge
- MoS₂:
Molybdenum disulfide
- MWCNTs:
Multi‑walled carbon nanotubes
- PANI:
Polyaniline
- Rct:
Charge‑transfer resistance
- Rs:
Solution resistance
- SEM:
Scanning electron microscopy
- η:
Power density
Nanomaterials
- High-Performance Flexible Supercapacitors Using Polyaniline-Coated Graphene Aerogel Electrodes
- Rapid Fabrication of Hierarchical Porous Polyaniline/Polyurethane Sponge Composites for Flexible Pressure and Tunable Gas Sensors
- High-Capacity Few-Layered MoS₂/Acetylene Black Anode for Li‑Ion Batteries
- Pd‑Decorated Few‑Layer MoS₂/SiO₂/Si Heterojunctions Deliver Record Room‑Temperature Hydrogen Detection
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- Carrier Transport and Gas‑Sensing Performance of Asymmetric MoS₂ Schottky Diodes via Charge‑Transfer‑Induced Barrier Modulation
- High‑Performance Dye‑Sensitized Solar Cells Using Screen‑Printed Multi‑Walled Carbon Nanotube Counter Electrodes
- How Interlayer Coupling and Van Hove Singularities Shape MoS₂ Multilayer Electronics and Optics
- High‑Yield Exfoliation of MoS₂ via Ultrasound Sonication in a Supercritical CO₂–NMP Complex Solvent
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures



