Engineering Band Alignment in MoS₂/HfO₂ Heterojunctions via Controlled Nitridation
Abstract
We examined how a nitrogen plasma treatment alters the band alignment between few‑layer MoS₂ and HfO₂ by X‑ray photoelectron spectroscopy (XPS). The valence‑band offset (VBO) and conduction‑band offset (CBO) of the MoS₂/HfO₂ heterojunction were found to be 2.09 ± 0.1 eV and 2.41 ± 0.1 eV without nitridation, and 2.34 ± 0.1 eV and 2.16 ± 0.1 eV with nitridation, respectively. The tunability originates from Mo‑N bond formation and surface band bending in HfO₂ induced by nitrogen incorporation. This study demonstrates that strategic nitridation can be used to engineer the energy landscape of MoS₂/HfO₂ interfaces, offering a pathway to optimize 2D‑material based electronic devices.
Background
Layered transition‑metal dichalcogenides (TMDCs) have attracted intense research interest due to their unique electronic and optical properties that are highly tunable with layer thickness [1, 2]. Molybdenum disulfide (MoS₂) is especially promising as a channel material for post‑7‑nm scaling, offering a direct bandgap of 1.8 eV in the monolayer and an indirect bandgap of 1.2 eV in bulk form [7]. Unlike graphene’s zero bandgap, MoS₂’s thickness‑dependent bandgap enables both optical and electrical device applications [3, 8]. The density of states in few‑layer MoS₂ is roughly three times that of the monolayer, which translates to higher ballistic drive currents [8]. Consequently, few‑layer MoS₂ can provide significant advantages for transistor design compared with its monolayer counterpart [3].
Meanwhile, silicon‑oxide‑based dielectrics are approaching their physical limits due to their low dielectric constant. To achieve thin equivalent‑oxide thicknesses (EOT) and high‑performance devices, high‑k dielectrics such as Al₂O₃, ZrO₂, HfO₂, and h‑BN have been integrated with MoS₂ [10–14]. For example, DiStefano et al. reported CBO and VBO values of 3.3 ± 0.2 eV and 1.4 ± 0.2 eV for MoS₂ on amorphous BN [13], while Tao et al. measured CBOs of 3.56 eV (Al₂O₃) and 1.22 eV (ZrO₂) for monolayer MoS₂ [15]. McDonnell et al. found a CBO of 2.09 ± 0.35 eV and a VBO of 2.67 ± 0.11 eV for the MoS₂/HfO₂ interface [12]. HfO₂ is particularly attractive because of its high dielectric constant (~20) and compatibility with poly‑SiGe, TaN, and poly‑crystalline silicon gates [16]. However, HfO₂ suffers from poor thermal stability, high leakage, and significant trap densities [17], motivating passivation strategies such as interface nitridation or fluorination [18, 19].
In this work, we investigated the band alignment of few‑layer MoS₂ on HfO₂ dielectrics with and without nitrogen plasma treatment, characterizing the interfacial chemistry by XPS.
Methods
SiO₂/Si wafers (280 nm SiO₂) were cleaned sequentially in acetone and isopropanol (10 min each) by ultrasonic agitation, followed by DI water rinse and N₂ dry. Few‑layer MoS₂ films were grown on SiO₂/Si by chemical vapor deposition (CVD) using MoO₃ (0.08 mg, 99 % Alfa Aesar) and S powder (1 g, 99 %) [20, 21]. After growth, the MoS₂ films were transferred onto HfO₂/Si substrates using a PMMA‑assisted wet transfer method (Figure 1a). PMMA was spin‑coated onto MoS₂/SiO₂/Si, the SiO₂ layer was etched in KOH, and the floating MoS₂/PMMA stack was scooped onto HfO₂/Si before dissolving PMMA in acetone.
HfO₂ layers were deposited by atomic layer deposition (ALD) at 200 °C using tetrakis(ethylmethylamido)hafnium (TEMAH) and H₂O as precursors [23, 24]. To optimize the nitrogen incorporation, secondary ion mass spectrometry (SIMS) indicated that 70 s of N₂ plasma introduced excessive nitrogen into the oxide, degrading its quality, whereas 30 s produced no detectable surface nitrogen. For the nitridated samples, a 50 s N₂ plasma treatment (3 Pa) was applied to HfO₂/Si prior to MoS₂ transfer. This condition delivered a nitrogen dose of ~8.4 × 10¹⁴ atoms cm⁻², corresponding to ~1.5 % nitrogen concentration as confirmed by XPS. Four samples were prepared for XPS analysis: (1) MoS₂ on SiO₂/Si, (2) bulk HfO₂ on Si, (3) MoS₂ on as‑grown HfO₂, and (4) MoS₂ on N₂‑plasma‑treated HfO₂.
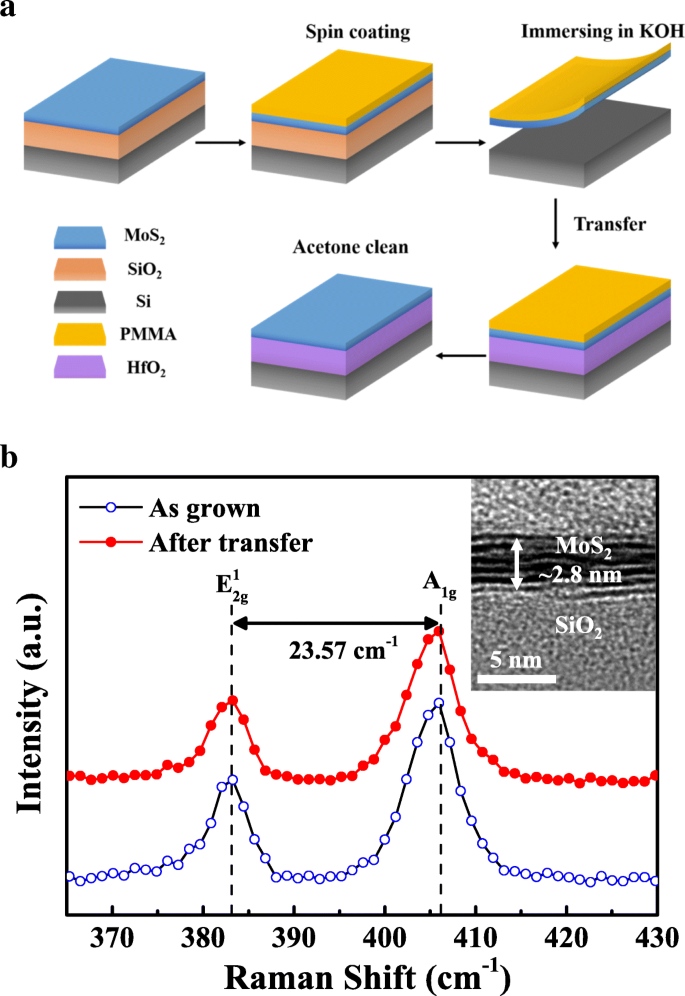
a Process flow of PMMA‑assisted wet transfer for the MoS₂/ALD‑HfO₂ heterojunction. b Raman spectra of as‑grown and transferred MoS₂. Inset: cross‑section TEM of MoS₂ on SiO₂/Si.
Results and Discussions
Raman spectroscopy (RENISHAW InVia) confirmed that the transfer process preserved the crystal quality of few‑layer MoS₂. Two prominent peaks at 382.86 cm⁻¹ (E₂g¹) and 406.43 cm⁻¹ (A₁g) showed no significant shift after transfer, indicating minimal strain or defect introduction. The frequency separation (Δk ≈ 23.57 cm⁻¹) corresponds to four‑to‑five layers, corroborated by HRTEM measurements of ~2.8 nm thickness (Figure 1b).
SIMS depth profiling of the nitrided sample (Figure 2a) revealed a nitrogen concentration gradient, with nitrogen diffusing into the HfO₂ layer and accumulating near the HfO₂/Si interface. The N 1s XPS spectra (Figure 2b) showed a low‑intensity peak at ~395.8 eV in the nitrided heterojunction, indicative of Mo‑N bond formation [31].
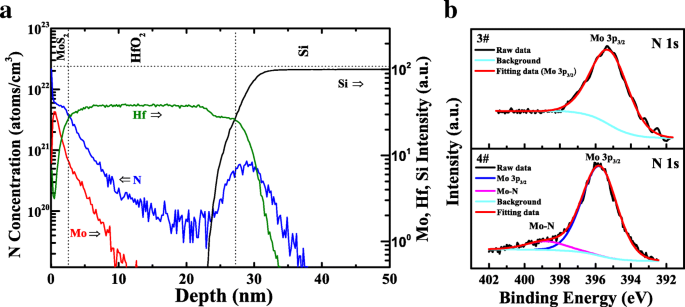
a SIMS depth profiles of Mo, N, Hf, and Si for MoS₂ on nitrided HfO₂/Si. b N 1s XPS spectra for MoS₂/HfO₂ heterojunctions with and without nitridation.
To extract the VBO, XPS spectra of Mo 3d and Hf 4f core levels were recorded with 0.05 eV step size using a VG ESCALAB 220i‑XL system (Al Kα, 1486.6 eV). The binding‑energy differences (BEDs) between core levels and the valence‑band maximum (VBM) were determined as 228.49 ± 0.1 eV for Mo 3d₅/₂ in MoS₂ (sample 1) and 14.10 ± 0.1 eV for Hf 4f₇/₂ in bulk HfO₂ (sample 2). For the heterojunctions, the Mo 3d₅/₂ peak shifted from 229.45 ± 0.05 eV (sample 3) to 229.90 ± 0.05 eV (sample 4) upon nitridation, reflecting charge transfer across the Mo‑N interface. Likewise, the Hf 4f₇/₂ peak moved from 17.40 ± 0.05 eV to 17.60 ± 0.05 eV, and the O 1s peak shifted by 0.20 eV to higher binding energy (Figure 3d). These shifts signify downward band bending in HfO₂, consistent with nitrogen‑induced donor‑like defects [34].
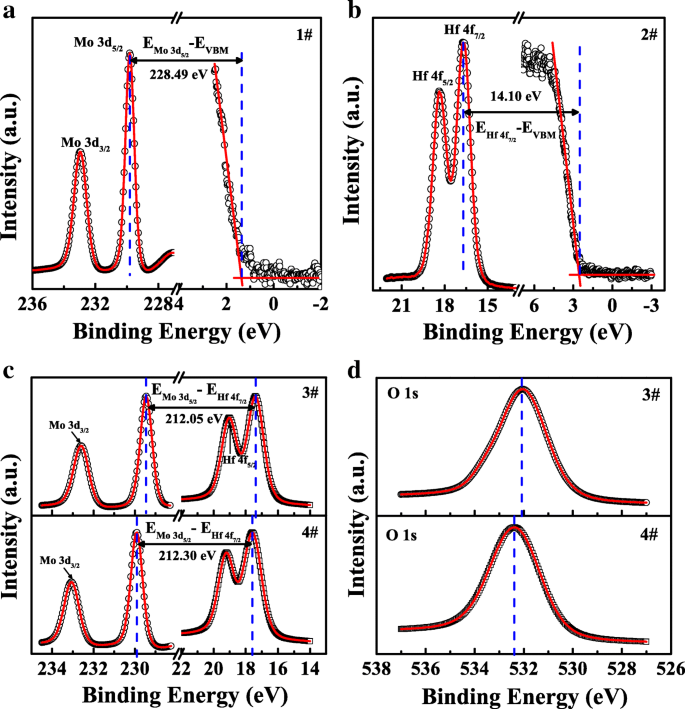
a XPS spectra of Mo 3d and valence band for few‑layer MoS₂. b XPS spectra of Hf 4f and valence band for bulk HfO₂. c XPS spectra of Mo 3d, Hf 4f, and d O 1s for MoS₂/HfO₂ heterojunctions with and without nitridation.
Using the Kraut method, the VBO (ΔE_V) was calculated as 2.09 ± 0.1 eV for the nitrided heterojunction and 2.34 ± 0.1 eV for the unnitrided one. The CBO (ΔE_C) was derived from the bandgap difference (HfO₂: 5.9 ± 0.1 eV; MoS₂: 1.4 ± 0.1 eV) and the VBO: 2.41 ± 0.1 eV (nitrided) versus 2.16 ± 0.1 eV (unnitrided) (Eq. 2). Band diagrams (Figure 4) illustrate that both heterojunctions provide strong confinement for electrons and holes, essential for MoS₂‑based FETs. The nitrided interface, with its higher CBO, is particularly favorable for n‑channel devices.
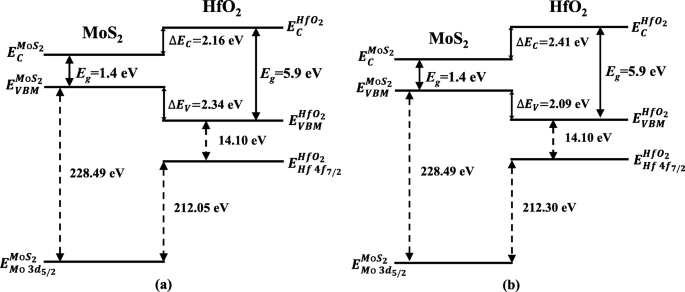
Band diagrams of MoS₂/HfO₂ heterojunctions: a without nitridation and b with nitridation.
Conclusions
Our XPS study demonstrates that a brief N₂ plasma treatment of HfO₂ before MoS₂ deposition can significantly modify the interfacial band alignment. The unnitrided heterojunction exhibits a VBO of 2.34 ± 0.1 eV and a CBO of 2.16 ± 0.1 eV, whereas nitridation reduces the VBO to 2.09 ± 0.1 eV and raises the CBO to 2.41 ± 0.1 eV. The observed Mo‑N bonding and nitrogen‑induced donor states account for the band bending and the improved electron confinement. These findings provide a clear route for band‑engineering of 2D/ high‑k interfaces, paving the way for more reliable and high‑performance MoS₂‑based electronics.
Availability of Data and Materials
The datasets supporting the conclusions of this manuscript are included within the manuscript.
Abbreviations
- ALD:
Atomic layer deposition
- BE:
Binding energy
- BED:
Binding energy difference
- CBO:
Conduction band offset
- CL:
Core level
- CVD:
Chemical vapor deposition
- FET:
Field‑effect transistor
- HfO₂:
Hafnium oxide
- HRTEM:
High‑resolution transmission electron microscope
- MoS₂:
Molybdenum disulfide
- PMMA:
Poly (methyl methacrylate)
- SIMS:
Secondary ion mass spectrometry
- SL:
Single‑layer
- TEMAH:
Tetrakis (ethylmethylamido) hafnium
- TMDC:
Transition metal dichalcogenide
- VBM:
Valence band maximum
- VBO:
Valence band offset
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- Terahertz Band: Fundamentals, Properties, and Emerging Applications
- Controlling Photoresponsivity in Multi‑Layer MoS₂ Phototransistors Through Applied Bias
- Gradient Nanomechanical Properties of Fluorosed Enamel: Implications for Restorative Material Selection
- Energy Band Alignment in Atomic‑Layer‑Deposited ZnO/β‑Ga₂O₃ (over 2 01) Heterojunctions Revealed by XPS
- Energy Band Alignment in MoS₂/ZrO₂ Heterojunctions: Impact of CHF₃ Plasma Treatment
- How Interlayer Coupling and Van Hove Singularities Shape MoS₂ Multilayer Electronics and Optics
- Precise Band Alignment of 2D MoS₂ on 3D β‑Ga₂O₃ Heterojunctions: Impact of Nitridation
- Polarization-Dependent Reflection Spectra of Single-Layer MoS₂ and ReS₂ on SiO₂/Si and Quartz
- Precision Biaxial Strain Sensor Leveraging a Single MoS₂ Grating
- Band‑Gap Engineering in Short‑Period CdO/MgO Superlattices: Experimental and Theoretical Insights



